A Kidney-Targeted Nanoparticle To Augment Renal Lymphatic Density Decreases Blood Pressure in Hypertensive Mice
Nov 10, 2023
1. Introduction
Identifying food ingredients with immunomodulatory properties together with their respective molecular targets has attracted increased interest in recent years. Due to their high ligand diversity, the transient receptor potential superfamily of ion channels (TRP channels) represents a very interesting class of potential target structures.
The mammalian TRP channel superfamily includes six related protein families, namely the ankyrin (TRPA), the canonical (TRPC), the melastatin (TRPM), the mucolipin (TRPML), the polycystin (TRPP), and the vanilloid (TRPV) family, all of which typically share six transmembrane segments that assemble as tetramers to form cation-permeable pores with varying cation selectivity.[1] The TRPV1 channel may be, by far, the most intensively investigated member of the TRP superfamily. TRPV1 was initially identified as the receptor for capsaicin, the pungent component of chili peppers.[2] Later, also the pungent ingredients from ginger, the gingerols, were shown to activate TRPV1.[3]

CLICK HERE TO GET HERBAL CISTANCHE FOR KIDNEYS
The biological role of TRPV1 in non-neuronal cell types is still under extensive investigation, yet the results available imply functions far beyond sensory and thermal perception. For example, a 12-week intervention with a daily dose of 0.15 mg of the TRPV1 agonist nonivamide, a structural capsaicin analogue, prevented a dietary-induced gain in body fat mass, and increased plasma serotonin levels in healthy overweight subjects.[4]
In 3T3-L1 adipocytes activation of TRPV1 by nonivamide decreased lipid accumulation during differentiation and maturation by suppressing PPAR𝛾 expression.[5] In macrophages, capsaicin and nonivamide attenuated an LPS-induced release of pro-inflammatory cytokines like IL6, CXCL8, and TNF-alpha, in a TRPV1-dependent manner.[6] Yet, the particular functions of TRPV1 in human blood leukocytes remain vague. In human NK cells, 10 and 50 μM capsaicin induced a rise in intracellular Ca2+ concentrations, indicating a functional TRPV1 channel.[7]
However, dampened NK cell effector functions such as cytotoxic degranulation and cytokine secretion, induced by pre-treatment of the cells with capsaicin for 1 h in a concentration range of 10–100 μM, were largely TRPV1 independent.[7] In T cells, TRPV1 was documented to be involved in the processes of T cell receptor signaling, T cell proliferation and differentiation, as well as cytokine production.[8] Previous work from our group also demonstrated functional expression of TRPV1 in human primary T cells.[9] Furthermore, dose response analyses in concentrations ranging from 0.03 to 300 μmol L−1 revealed that [6]-gingerol inhibits cytokine secretion by primary human leukocytes with an IC50 value of 82.2 μmol L−1.

However, quantitation of [6]-gingerol in plasma samples of healthy subjects revealed a mean maximum plasma concentration of only 42.0 ± 16.3 nmol L−1 after the intake of 1 L of ginger tea. Since these [6]- gingerol concentrations had no significant impact on cytokine secretion in previous studies,[9] it is unclear, whether a dietary relevant concentration of 50 nmol L−1, being reached in blood plasma after consumption of 1 L of ginger tea, is sufficient to modulate cellular immune responses in other human primary leukocytes.
For human neutrophils, knowledge about the functional role of TRPV1 is limited. Whereas Köse and Nazıroglu˘ [10] showed Ca2+-fluxes in neutrophils in response to 10 μM capsaicin to be decreased by the TRPV1 antagonist capsazepine, other results did not demonstrate capsaicin to induce a Ca2+ influx when tested in a concentration range of 1–100 μM, despite a detectable TRPV1 RNA expression.[11]
Neutrophils are the most abundant leukocytes in human blood, accounting for 60–70 % of all circulating white blood cells. They are the first immune cells that are recruited to the sites of infection; they are therefore often referred to as the first line of defense.[12] Recruitment of neutrophils is triggered by, among others, the bacterial or mitochondria-derived peptide N-Formylmethionine-leucyl-phenylalanine (fMLF) or chemokines such as CXCL8 (IL-8).[13]
Defense mechanisms of neutrophils include phagocytosis,[14] anti-microbial enzyme release via degranulation,[15] generation of reactive oxygen species (ROS),[16] and the formation of neutrophil extracellular traps.[17] Besides these direct defense mechanisms, neutrophils further contribute to subsequent immune responses via the release of various cytokines and chemokines.[18] Also, neutrophils can undergo a priming process that enables them to respond more strongly to subsequent full activation.[16b]
In recent years, evidence has grown that ingredients from food and or medicinal plants can modify one or more of the mentioned defense responses of human neutrophils. These modifications include increased phagocytotic activity[19] and ROS generation,[20] augmented chemotaxis towards fMLF,[21] and formation of neutrophil extracellular traps.[20] However, the active compound(s) were not identified. Constituents from Ferula akitschkensis (𝛽-pinene, sabinene, 𝛾-terpinene, geranylacetone, and isobornylacetate) desensitized neutrophils to fMLF- and CXCL8- induced Ca2+ influx and inhibited fMLF induced chemotaxis, wherein the geranylacetone-induced effects were mediated via TRPV1.[22]

Based on the available data, we hypothesized that ligand induced activation of TRPV1 by [6]-gingerol can affect general neutrophil functions, either directly or via enhancing their responses to activating stimuli. Within the scope of this hypothesis, we particularly aimed at elucidating, whether a verified nutritionally relevant concentration is sufficient to modulate cellular immune responses in human primary neutrophils as part of the leukocyte population. '
Furthermore, we sought to compare the RNA expression levels of all members of the mammalian TRP superfamily in five of the most prominent cell types in human blood in order to obtain a qualitatively and quantitatively comprehensive overview of TRP channel expression in human leukocytes.
2. Results
2.1. Abundance and Relative Transcript Levels of TRP Channels in Human Leukocytes
In order to evaluate TRP channel RNA expression in blood leukocytes, five of the most prominent leukocyte cell types were isolated from the blood of healthy donors and the RNA expression of TRP channels was analyzed via quantitative RT-PCR (Figure 1).
Specific transcripts of the TRPV as well as the TRPM family were detected with high frequencies of 75–100% in all cell types analyzed. The mean overall frequency considering all cell types was 96% for the TRPV family and 98% for the TRPM family. TRPC-specific transcripts were much less abundant, ranging from 0% in monocytes, NK cells, T cells, and B cells to 100% in NK cells, and T cells with an overall mean frequency of only 53% (Figure 1A). The TRPC5-specific transcript was only detected in neutrophils, with a frequency of 70%.
Also, the TRPA1-specific transcript generally revealed a rather low abundance in the cell types analyzed with a frequency of 100% in neutrophils, 25% in monocytes, 80% in NK cells, 100% in T cells, and 60% in B cells. Likewise, the TRPML3-specific transcript showed a low frequency in neutrophils (50%), monocytes (25%), NK cells (40%), and B cells (20%), but a frequency of 100% in T cells. The TRPV1-specific transcript was detected in all cell types, showing a frequency of 100% in monocytes, NK cells, and T cells, and a frequency of 90% in neutrophils and 80% in B cells.
Regarding relative RNA expression, as compared to the respective frequencies, the TRP channels revealed a more cell type-specific expression pattern, as evident by the comparison of the respective Δct values in the different cell types analyzed (Figure 1B). For example, TRPP3 was detected with a frequency of 100% in all cell types, but clearly revealed the highest expression level in monocytes.
In contrast, the TRPV2 channel was detected in all of the cell types investigated, with a comparably high RNA expression level as well as a high frequency. Likewise, transcript levels of TRPV1 were similar in all cell types examined (Figure 1B).
2.2. TRPV1 Surface Expression on Neutrophils
The current knowledge about the function of TRPV1 in human neutrophils is still unclear. To further explore the roles of TRPV1 in human neutrophils, we next investigated whether the TRPV1 channel is expressed on the surface of primary human neutrophils using live cell flow cytometry (Figure 2).
The isolated neutrophils were stained for CD15 as a surface marker for neutrophils (Figure 2A,B) and simultaneously either stained with an antibody raised against an epitope in the first extracellular loop of the TRPV1 protein (Figure 2B) or the respective isotype control, the latter serving as a surrogate for measuring unspecific binding (Figure 2A). Within the CD15+ population, the fluorescence intensity for FITC was analyzed. Staining of the neutrophils with the TRPV1 antibody led to a clearly distinguishable fluorescence signal compared to the isotype control, thereby confirming surface expression of TRPV1 in primary human neutrophils (Figure 2C). Analyzing neutrophils from four individual donors revealed a comparable surface expression of TRPV1 (Figure 2D).

2.3. [6]-Gingerol Induced Increase in Intracellular Ca2+
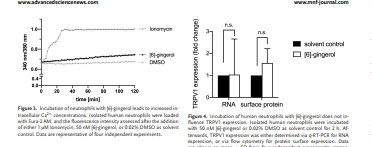
Since ligand-induced activation of TRPV1 will result in an influx of Ca2+, [23] intracellular Ca2+ concentrations of neutrophils were determined via the Ca2+-sensitive dye Fura-2. Based on our previous findings,[9] a concentration of 50 nM of the well-known TRPV1 ligand [6]-gingerol and an incubation time of 2 h were chosen. The analyses showed that incubation of neutrophils with 50 nM [6]-gingerol resulted in increased intracellular Ca2+ concentrations which were on average 18.4% ± 1.0% of the maximum value, as determined by applying 1 μM of the ionophore ionomycin. The increase induced by DMSO was at 4.7% ± 1.2% (Figure 3).

2.4. Impact of [6]-Gingerol on TRPV1 Expression
Next, we aimed at analyzing the impact of TRPV1 stimulation by [6]-gingerol on TRPV1 expression at the transcript level via q-RTPCR as well as at the surface protein level via live cell staining. For this purpose, human neutrophils were incubated with 50 nM [6]- gingerol for 2 h and the respective expression levels quantified.
This 2 h incubation impacted neither TRPV1 transcript nor protein levels (Figure 4), and also did not change, except for IL6, IL17A, IL24, C5, and GDF5, the RNA expression of common cytokine and chemokine genes investigated (Figure S1, Table S2, Supporting Information).
2.5. Impact of [6]-Gingerol on Expression of Neutrophil Surface Markers
Supportive Service Of Wecistanche-The largest cistanche exporter in the China:
Email:wallence.suen@wecistanche.com
Whatsapp/Tel:+86 15292862950
Shop For More Specifications Details:
https://www.xjcistanche.com/cistanche-shop
GET NATURAL ORGANIC CISTANCHE EXTRACT WITH 25% ECHINACOSIDE AND 9% ACTEOSIDE






