Cost Analysis Of Screening For IgA Nephropathy Using Novel Biomarkers
Jul 21, 2023
Abstract
1. Objectives
IgA nephropathy (IgAN) is the most common primary chronic glomerulonephritis and a major cause of end-stage kidney disease worldwide. Novel biomarkers, including the aberrantly glycosylated IgA1 and glycan-specific antibodies, could be useful in the diagnosis of IgAN. This study aimed to assess the cost analysis of IgAN screening using novel biomarkers in addition to conventional screening compared with conventional screening alone.
2. Methods
To estimate the medical expense of each strategy related to renal disease for 40 years, we developed an analytical decision model. The decision tree started at “40 years of age with first-time hematuria.” It simulated 2 clinical strategies: IgAN screening using the novel biomarkers (group N) and conventional screening (group C). The analysis results presented medical expenses from a societal perspective. Discounting was not conducted.
3. Results
The expected medical expense per person for 40 years was U31.2 million (~$291000) in group N and U33.4 million (~$312 000) in group C; hence, expense in group N was lower by U2.2 million (~$21000). In group N, the expected value of IgAN increased by 5.67% points (N 48.44%, C 42.77%) and that of dialysis introduction decreased by 0.85% points (N 19.06%, C 19.91%). In the sensitivity analysis, expenses could be reduced in almost all cases except when renal biopsy using conventional screening was performed at the rate of 73% or higher.
4. Conclusion
Screening for IgAN using novel biomarkers would reduce renal disease–related expenses.
5. Keywords
cost analysis, cost-effectiveness, IgA nephropathy, novel biomarkers, screening, chronic kidney disease, and end-stage kidney disease.

Click here to know what is the Cistanche
Introduction
The annual number of patients on dialysis has increased worldwide, and it is expected to rise sharply in the next decade.1 Regarding the incidence rate of treatable end-stage kidney disease (ESKD), many Asian countries including Japan, Taiwan, South Korea, Singapore, and Thailand have the highest grade rate according to the United States Renal Data System.2 In Japan, the number of patients on dialysis exceeded 330 000 in 2018.3 Chronic glomerulonephritis is the second most common cause of dialysis introduction after diabetes mellitus,3 and almost half of its cases are regarded as IgA nephropathy (IgAN). According to an epidemiologic study and genome-wide survey,4 IgAN is the most common primary chronic glomerulonephritis, and East Asia including Japan has a high prevalence of IgAN. IgAN causes ESKD in 20% to 40% of patients within 10 to 20 years from the onset,5-7 whereas a previous study showed that 90% of cases of IgAN could achieve clinical remission when tonsillectomy and steroid pulse therapy (TSP) was performed within 3 years from the onset in Japan.8 Therefore, early diagnosis and treatment of IgAN would reduce the number of patients on dialysis with IgAN by preventing progression to ESKD.
Because the annual health checkup is performed mandatorily for almost all Japanese citizens, 70% of cases of IgAN are diagnosed as asymptomatic hematuria by screening in Japan. Nonetheless, it is not easy to detect patients with IgAN in the early stages by annual checkups owing to the following 3 reasons: first, detection of the presence of hematuria through annual checkups is relatively common, occurring in around 5% and 10% of men and women, respectively9; second, hematuria also arises from urological diseases, such as urinary stones and urinary tract infection, many of which are not serious problems, and therefore, a detailed examination is not often performed to determine the cause9; third, the diagnosis of IgAN requires an invasive renal biopsy. Therefore, the presence of hematuria alone at annual checkups often does not lead to a diagnosis of IgAN, and in some cases, in which treatment is delayed, leading to ESKD because of missed diagnosis and subsequent treatment. Thus, an efficient screening for IgAN on the diagnosis of hematuria is needed.
Lately, novel biomarkers for IgAN including aberrantly glycosylated IgA1 and glycan-specific antibodies can be measured, and these biomarkers are reportedly associated with disease activity10 and prognosis.11-14 Yanagawa et al15 indicated that biomarkers might be useful to differentiate IgAN from other glomerular diseases based on the results of 230 cases. Screening for IgAN using these biomarkers in addition to conventional screening may help identify patients with early-stage IgAN and consequently allow us to offer appropriate therapies and prevent ESKD.
Cost-effectiveness analysis (CEA) has gained importance as an approach to evaluating new medical technologies along with highly specialized medical technology and rising medical expenses in Japan and worldwide.16 The expensive dialysis costs because of the increasing number of patients on dialysis are a big burden in Japan.17 To prevent the introduction of dialysis for patients with IgAN and consequently reduce the high costs, a useful screening method, and its CEA are needed. Nonetheless, little is known about the cost-effectiveness of this IgAN screening method using novel biomarkers from a societal perspective. The study aimed to evaluate and compare renal disease–related expenses for 40 years when adding novel biomarkers for IgAN screening with that of conventional IgAN screening alone in Japanese clinical settings.

Cistanche tubulosa
Methods
1. Model Overview
We developed an analytical decision model to estimate the renal disease–related medical expense of each strategy for 40 years (Fig. 1). The decision tree started at 40 years of age with first-time hematuria and compared 2 kinds of strategies with simulated clinical scenarios: the intervention strategy as screening with novel biomarkers (group N) and the control strategy as conventional screening (group C). Figures 2 and 3 show the clinical flow of diagnosed “IgAN” and “not-IgAN,” respectively. The screening strategy with novel biomarkers had 4 components, namely, the result of screening with novel biomarkers (positive or negative), whether renal biopsy was performed or not, the diagnosis by renal biopsy, and the consequent treatment (Figs. 2[A] and 3[A]). The conventional screening strategy had 3 components, namely, whether a renal biopsy was performed or not, the diagnosis by renal biopsy, and the consequent treatment (Figs. 2[B] and 3[B]).
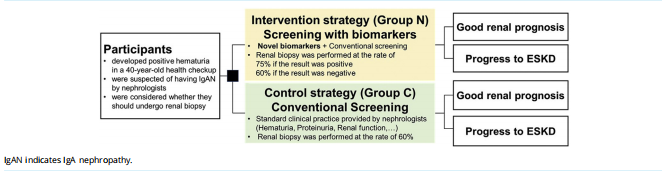
Figure 1. Analytical decision tree. The decision tree presents 2 options: the intervention strategy using screening with biomarkers (group N) or the control strategy using conventional screening (group C) for participants with suspected IgAN with first-time hematuria at the age of 40 years. The outcomes (the expected medical expenses, the prevalence of IgAN, and the proportion of dialysis introduction) of each strategy are compared.
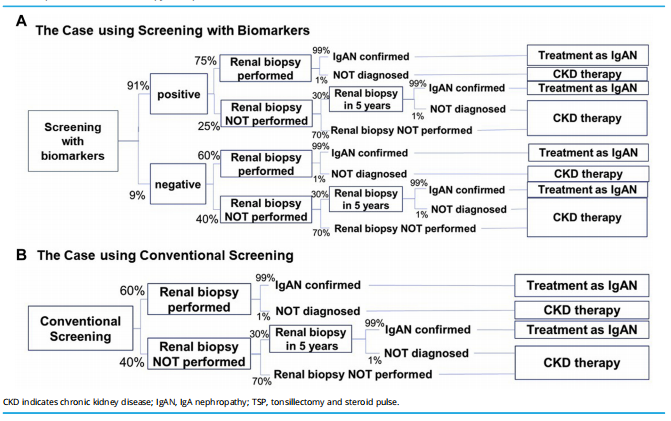
Figure 2. Clinical flow when the true diagnosis is IgAN. The proportion of patients with “true IgAN” was 60%. (A) The case using screening with biomarkers. The proportion of positive results with “true IgAN” of screening with biomarkers was 91%, which was defined as the value of sensitivity of screening with novel biomarkers. Renal biopsy was performed in 75% of patients with positive results and 60% of those with negative results, and 30% of patients who did not undergo renal biopsy at baseline received the procedure in 5 years. Definitive IgAN diagnosis was obtained in 99% of renal biopsies, which was defined as the value of sensitivity of renal biopsy. When the diagnosis was IgAN, TSP therapy or CKD therapy was performed. When the diagnosis of IgAN was not obtained by renal biopsy or when renal biopsy was not performed, CKD therapy was performed. (B) The case using conventional screening. Renal biopsy was performed at the rate of 60%, and 30% of those who did not undergo renal biopsy at baseline received the procedure in 5 years. Definitive IgAN diagnosis was obtained in 99% of renal biopsies, which was defined as the value of sensitivity of renal biopsy. When the diagnosis was IgAN, TSP therapy or CKD therapy was performed. When the diagnosis of IgAN was not obtained by renal biopsy or when renal biopsy was not performed, CKD therapy was performed.
2. Participants
We assumed that it was at the age of 40 years that Japanese people were informed about the first-time hematuria at the annual health checkup along with a diagnosis of suspected IgAN by nephrologists. Then, the nephrologists considered whether the participants should undergo renal biopsy or not, as shown in the details in Figures 2 and 3. The rate of normal renal function (chronic kidney disease [CKD] stage 1 or 2) was assumed to be 80% at baseline and 70% in 5 years, at the point of renal biopsy.

Figure 3. Clinical flow when the true diagnosis is “not-IgAN.” The proportion of patients with true “not IgAN” was 40%. (A) The case using screening with biomarkers. The proportion of negative results of screening with biomarkers in true “not IgAN” was 81%, which was defined as the value of specificity of screening with novel biomarkers. Renal biopsy was performed in 75% of patients with positive results and 60% of those with negative results, and 30% of those who did not undergo renal biopsy at baseline received this procedure in 5 years. Renal biopsy diagnosed that 25% had normal tissue and 75% had glomerular disease other than IgAN. The treatment was not performed because the renal prognosis was good in either case. (B) The case using conventional screening. Renal biopsy was performed at the rate of 60%, and 30% of patients who did not undergo renal biopsy at baseline received the procedure in 5 years. Renal biopsy diagnosed that 25% had normal tissue, and 75% had glomerular disease other than IgAN. The treatment was not performed because the renal prognosis was good in either case.
Figure 4 shows the assumption of the IgAN prognosis of each treatment option depending on the renal function at the biopsy. The rate of TSP therapy was 55% when the participants were in CKD stage 1 or 2 and 45% when they were in CKD stage 3 or below. A good prognosis of TSP therapy was obtained at the rate of 80% when the participants were in CKD stage 1 or 2 and 75% when they were in CKD stage 3 or below, whereas a good prognosis of CKD therapy was obtained at the rate of 65% when the participants were in CKD stage 1 or 2 and 55% when they were in CKD stage 3 or below. When the diagnosis of IgAN was not obtained incorrectly or renal biopsy was not performed, a good prognosis was obtained at the rate of 55%, and the remaining 45% developed ESKD 10 years later and underwent dialysis for 30 years.
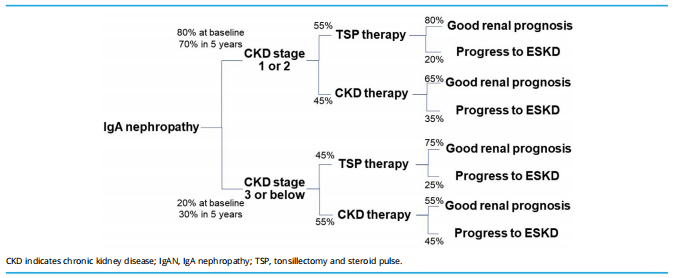
Figure 4. Treatment and prognosis in IgAN. The proportion of patients with CKD stage 1 or 2 at diagnosis was 80% at baseline and 70% in 5 years. The rate of TSP therapy was 55% in CKD stage 1 or 2 and 45% in CKD stage 3 or below, that is, CKD therapy was performed at the rate of 45% in CKD stage 1 or 2 and 55% in CKD stage 3 or below. A good prognosis of TSP therapy was obtained at the rate of 80% in CKD stage 1 or 2 and 75% in CKD stage 3 or below, whereas a good prognosis of CKD therapy was obtained at the rate of 65% in CKD stage 1 or 2 and 55% in CKD stage 3 or below.
3. Intervention Strategy
A screening for IgAN using novel IgAN-specific biomarkers (galactose-deficient IgA1, galactose-deficient IgA1 antibodies, and galactose-deficient IgA1 immune complexes) was performed in addition to the conventional strategy. This new screening strategy consisted of the level of each biomarker in addition to clinical findings including the degree of hematuria, proteinuria, and renal function. According to the research report, the sensitivity and specificity of this screening method were 91% and 81%, respectively.15,18 The nephrologists used the method before renal biopsy only to determine the indications for renal biopsy. The renal biopsy was assumed to be performed in 75% or 60% of patients with positive or negative results, respectively. The clinical flow after renal biopsy using screening with novel biomarkers was the same as the clinical flow using conventional screening.

Cistanche powder
4. Control Strategy
The standard clinical practice provided by nephrologists (including the degrees of hematuria, proteinuria, and renal function) was performed as the conventional screening. The renal biopsy was assumed to be performed in 60% of patients.
5. Outcome
The primary outcome was the renal disease-related medical expense of each strategy for 40 years. For clinical effectiveness, the incremental prevalence of IgAN diagnosis and the incremental proportion of dialysis introduction thereafter were obtained.
6. Model Inputs
We searched the literature by a systematic review, extracted the relevant documents, and applied the representative figures.15,18–24 Interstudy variability reflected the maximum and minimum values in the deterministic sensitivity analysis. We used figures by referring to unpublished research reports and conference reports when the corresponding previous documents were missing. The cost variables were defined by Japanese medical expenses, which were based on the discussion in the Central Social Insurance Medical Council. Each cost variable was calculated according to the clinical scenarios. Hospitalization costs were defined by referring to the Diagnosis Procedure Combination data at Juntendo University Hospital. Dialysis costs were cited from previous literature.23 The variables were reviewed and corrected by 4 specialists (expert panel) and then consensus was obtained from the whole research meeting (March, September, and December 2019).
7. Analysis Method
The model assumes a societal perspective. We considered only direct medical expenses related to renal disease. The expenses were calculated for 40 years in each strategy. Discounting was not conducted. A deterministic one-way sensitivity analysis was performed and expressed as a tornado diagram.
We conducted a sensitivity analysis based on each of the following case scenarios: (1) the rate of the TSP therapy was 80% when the participants were in CKD stage 1 or 2, (2) the proportion of the good prognoses was 99% when the participants with CKD stage 1 or 2 received TSP therapy, and (3) both scenario 1 and 2. The analysis was performed by the software TreeAge Pro 2019, R1 (Tree Age Software Inc., Williamstown, MA).
8. Ethical Consideration
This study did not need the approval of the Ethics Committee because all data used in the analysis were based on existing materials, including published articles and research reports. The authors have no conflict of interest to declare. This evaluation followed all criteria of the Consolidated Health Economic Evaluation Reporting Standards reporting guidelines.

Cistanche extract
Discussion
In this study, we demonstrated that screening for IgAN with novel biomarkers reduced the expected medical expense per person for 40 years by U2.2 million (~$21000, equivalent to 6.65% of that of the conventional screening) among participants with first hematuria with suspected IgAN at 40 years old. Our results suggested that screening for IgAN with novel biomarkers can help diagnose IgAN in the early stages and thus provide timely and appropriate therapy to participants and help prevent induction of dialysis because of IgAN. To our knowledge, this is the first study to conduct a cost analysis on the IgAN screening method.
To assess a long diverse clinical history of chronic diseases or a future medical technology, expert opinion as well as randomized controlled study could be useful to make an analytic model with higher reliability and validity. We developed an analytic decision model based on the 40-year clinical course of IgAN, referenced by an expert panel’s opinions. The formal method to solve these situations is termed “structured expert disciplines.” 25 Unfortunately, we could not use the structured consensus method when making the analytical decision model, nevertheless, we tried to increase the validity of this model by the expert panel’s consideration and consensus of the research group.
To assess a long diverse clinical history of chronic diseases or a future medical technology, expert opinion as well as randomized controlled study could be useful to make an analytic model with higher reliability and validity. We developed an analytic decision model based on the 40-year clinical course of IgAN, referenced by an expert panel’s opinions. The formal method to solve these situations is termed “structured expert disciplines.” 25 Unfortunately, we could not use the structured consensus method when making the analytical decision model, nevertheless, we tried to increase the validity of this model by the expert panel’s consideration and consensus of the research group.
Recently, trends in treatment for IgAN have changed. The results of a national questionnaire survey in 2008 suggested that TSP therapy was performed in more cases of IgAN.27 In this study, the variables for treatment selection and the prognosis for IgAN were based on the results of a multicentered large study conducted from 2002 to 2004.24 We conducted the sensitivity analysis similar to the current clinical situation, in which TSP therapy was performed more highly with a better response rate. According to this analysis, the expected medical expense was reduced by U3.3 million ($30 000), equivalent to 12% of the expected lifelong medical expenses among Japanese people. This result suggested that screening for IgAN with novel biomarkers would be more useful in the current clinical situation.

Cistanche capsules
Conclusion
A screening method with novel IgAN-specific biomarkers would reduce the expected renal disease–related medical expense among individuals with suspected IgAN.
REFERENCES
1. Liyanage T, Ninomiya T, Jha V, et al. Worldwide access to treatment for end-stage kidney disease: a systematic review. Lancet. 2015;385(9981):1975– 1982.
2. USRDS Reference Tables. The United States Renal Data System. https://adr. usrds.org/2020/reference-tables. Accessed March 5, 2021.
3. Therapy-current status of chronic dialysis therapy in Japan in 2018. The Japanese Society for Dialysis Therapy. https://docs.jsdt.or.jp/overview/ Published2020. Accessed March 5, 2021.
4. Magistroni R, D’Agati VD, Appel GB, Kiryluk K. New developments in the genetics, pathogenesis, and therapy of IgA nephropathy. Kidney Int. 2015;88(5):974–989.
5. Koyama A, Igarashi M, Kobayashi M. Natural history and risk factors for immunoglobulin A nephropathy in Japan. Research Group on Progressive Renal Diseases. Am J Kidney Dis. 1997;29(4):526–532.
6. Manno C, Strippoli GF, D’Altri C, Torres D, Rossini M, Schena FP. A novel simpler histological classification for renal survival in IgA nephropathy: a retrospective study. Am J Kidney Dis. 2007;49(6):763–775.
7. Schena FP. Immunoglobulin A nephropathy with mild renal lesions: a call in the forest for physicians and nephrologists. Am J Med. 2001;110(6): 499–500.
8. Ieiri N, Hotta O, Sato T, Taguma Y. Significance of the duration of nephropathy for achieving clinical remission in patients with IgA nephropathy treated by tonsillectomy and steroid pulse therapy. Clin Exp Nephrol. 2012;16(1):122–129.
9. Japanese Clinical Practice Guidelines for hematuria diagnosis 2013. Japanese Society of Nephrology. www.jsn.or.jp/guideline/pdf/hugl2013.pdf. Accessed March 5, 2021.
10. Suzuki Y, Matsuzaki K, Suzuki H, et al. Serum levels of galactose-deficient immunoglobulin (Ig) A1 and related immune complex are associated with the disease activity of IgA nephropathy. Clin Exp Nephrol. 2014;18(5): 770–777.
11. Camilla R, Suzuki H, Dapra V, et al. Oxidative stress and galactose-deficient IgA1 as markers of progression in IgA nephropathy. Clin J Am Soc Nephrol. 2011;6(8):1903–1911.
12. Zhao N, Hou P, Lv J, et al. The level of galactose-deficient IgA1 in the sera of patients with IgA nephropathy is associated with disease progression. Kidney Int. 2012;82(7):790–796.
13. Berthoux F, Suzuki H, Thibaudin L, et al. Autoantibodies targeting galactose deficient IgA1 are associated with the progression of IgA nephropathy. J Am Soc Nephrol. 2012;23(9):1579–1587.
14. Chen P, Yu G, Zhang X, et al. Plasma galactose-deficient IgA1 and C3 and CKD progression in IgA nephropathy. Clin J Am Soc Nephrol. 2019;14(10): 1458–1465.
15. Yanagawa H, Suzuki H, Suzuki Y, et al. A panel of serum biomarkers differentiates IgA nephropathy from other renal diseases. PLoS One. 2014;9(5), e98081.
16. Fukuda T. A pilot program of implementing health technology assessment to decision making in Japan. Jpn J Pharmacoepidemiol. 2018;23(1):3–10.
17. Wang J, Zhang L, Tang SC, et al. Disease burden and challenges of chronic kidney disease in North and East Asia. Kidney Int. 2018;94(1):22–25.
18. Suzuki H. [Aberrantly glycosylated IgA1 as a useful biomarker in IgA nephropathy]. J Clin Exp Med (Japanese). 2015;255:1095–1100.
19. Hoshino Y, Kaga T, Abe Y, et al. Renal biopsy findings and clinical indicators of patients with hematuria without overt proteinuria. Clin Exp Nephrol. 2015;19(5):918–924.
20. Lee HM, Hyun JI, Min JW, et al. The natural course of biopsy-proven isolated microscopic hematuria: a single center experience of 350 patients. J Korean Med Sci. 2016;31(6):909–914.
21. Kim BS, Kim YK, Shin YS, et al. Natural history and renal pathology in patients with isolated microscopic hematuria. Korean J Intern Med. 2009;24(4):356–361.
22. Ubara Y, Kawaguchi T, Nagasawa T, et al. Kidney biopsy guidebook 2020 in Japan. Clin Exp Nephrol. 2021;25(4):325–364.
23. Fukuhara S, Yamazaki C, Hayashino Y, et al. The organization and financing of end-stage renal disease treatment in Japan. Int J Health Care Finance Econ. 2007;7(2-3):217–231.
24. Hirano K, Matsuzaki K, Yasuda T, et al. Association between tonsillectomy and outcomes in patients with immunoglobulin A nephropathy. JAMA Netw Open. 2019;2(5):e194772.
25. Soares MO, Sharples L, Morton A, Claxton K, Bojke L. Experiences of structured elicitation for model-based cost-effectiveness analyses. Value Health. 2018;21(6):715–723.
26. Lifelong medical expenses. https://www.mhlw.go.jp/content/shougai_h28.pdf. Ministry of Health, Labor, and Welfare. Accessed March 5, 2021.
27. Matsuzaki K, Suzuki Y, Nakata J, et al. A nationwide survey on current treatments for IgA nephropathy in Japan. Clin Exp Nephrol. 2013;17(6):827–833.
28. Rodrigues JC, Haas M, Reich HN. IgA nephropathy. Clin J Am Soc Nephrol. 2017;12(4):677–686.
29. Harabuchi Y, Takahara M. Recent advances in the immunological understanding of the association between tonsil and immunoglobulin A nephropathy as a tonsil-induced autoimmune/inflammatory syndrome. Immun Inflamm Dis. 2019;7(2):86–93.
30. Yang X, Zhu A, Meng H. Tonsillar immunology in IgA nephropathy. Pathol Res Pract. 2020;216(7):153007.
31. Yang D, He L, Peng X, et al. The efficacy of tonsillectomy on clinical remission and relapse in patients with IgA nephropathy: a randomized controlled trial. Ren Fail. 2016;38(2):242–248.
Mami Ishida, MD, MPH, Keiichi Matsuzaki, MD, PhD, Hiroshi Ikai, MD, PhD, Hitoshi Suzuki, MD, PhD, Takashi Kawamura, MD, PhD, Yusuke Suzuki, MD, PhD






