Echinacoside Hampers The Malignant Progression Of Breast Cancer MCF-7 Cells By Modulating AKR1B10/ERK Signal Transduction
Nov 09, 2022
Abstract: This study analyzes the impact of echinacoside (ECH) in the proliferation, metastasis and adriamycin (ADR) resistance of breast cancer (BC) MCF-7 cells via the modulation of aldo-keto reductase family 1 member 10 (AKR1B10)/extracellular signal-regulated kinase (ERK) pathway. The chemical structure of ECH was firstly confirmed. MCF-7 cells were treated with different concentration (0, 10, 20, 40 μg·mL-1 ) of ECH for 48 h. Western blotting was used to analyze expression of AKR1B10/ERK pathway-associated proteins and cell counting kit-8 (CCK-8) assay to determine cell viability. MCF-7 cells were collected and classified into control group, ECH group, ECH + Ov-NC group, and ECH + OvAKR1B10 group. Then Western blotting was employed to analyze the expression of AKR1B10/ERK pathway-associated proteins. CCK-8 and 5-ethynyl-2’-deoxyuridine (EdU) assay were used to examine cell proliferation. Cell migration was appraised with scratch assay, transwell assay, and Western blotting. Eventually, MCF-7 cells were treated with ADR for 48 h to induce ADR resistance. Cell viability was tested by CCK-8 assay and cell apoptosis was estimated based on terminaldeoxynucleoitidyl transferase mediated nick end labeling (TUNEL) assay and Western blotting. Based on Protein Data Bank (PDB) and molecular docking, the binding affinity of ECH to AKR1B10 was assessed. Various doses of ECH decreased the expression of AKR1B10/ERK pathway-associated proteins in a dose-dependent manner and declined cell viability compared with the control group. Compared with the control group, 40 μg·mL-1 ECH blocked the AKR1B10/ERK pathway in MCF-7 cells and inhibited the proliferation, metastasis and ADR resistance of the cells. Compared with the ECH + Ov -NC group, ECH + Ov-AKR1B10 group showed the recovery of some biological behaviors of MCF-7 cells. ECH also targeted AKR1B10. ECH can inhibit the proliferation, metastasis, and ADR resistance of BC cells by blocking AKR1B10/ERK pathway.
Keywords: echinacoside; AKR1B10/ERK signaling; breast cancer; adriamycin resistance
Breast cancer (BC) is by far the most common female malignancy, with a median age of diagnosis of around 60 years [1]. The proportion of new cases of BC in China accounts for 12.2% of the global proportion, and the proportion of BC deaths in the world accounts for 9.6% [2]. Genetic and environmental factors such as high-fat diet, alcohol intake, and insufficient physical activity are risk factors for breast cancer [3-4]. Modern treatments still include surgery, radiation therapy, and drug therapy [1]. Common clinical drugs include cyclophosphamide, 5-fluorouracil, pirarubicin, tamoxifen citrate, etc. [5], and the existence of drug resistance in BC cells also brings about the treatment of BC to a certain extent. great challenge [6]. At the same time, as a heterogeneous tumor, BC is more likely to metastasize to organs such as bone, lung, liver, and brain, which makes metastatic breast cancer largely incurable [7]. Cistanche is a traditional medicinal material in my country. It belongs to the plant family and is known as "desert ginseng"[8]. Echinacoside (ECH) is a phenylethanoid glycoside extracted from Cistanche deserticola. Recent studies have emphasized that echinacoside has strong antioxidant, anti-inflammatory and anticancer properties. For example, by inhibiting the mTOR/STAT3 pathway, ECH can alleviate the inflammatory response and oxidative stress in paracetamol-induced liver injury [9]. Multiple evidences have confirmed that ECH mainly plays a tumor suppressor role in BC, and its mechanism may be related to down-regulating the expression of miR-4306 and miR-4508 [10] or blocking Wnt/β-catenin signaling [11].
Aldo-keto reductase 1B10 (AKR1B10), a member of the aldo-keto reductase superfamily, can convert toxic substances into non-toxic substances through redox reactions and produce corresponding alcohols, thus exerting a protective effect on host cells [12]. Studies suggest that AKR1B10 is mainly expressed in normal human tissues such as colon and small intestine [13]. More studies revealed that AKR1B10 mainly acts as an oncogene in breast cancer and may become a potential biomarker of breast cancer [14-16]. As a key member of the MAPK family, extracellular regulated protein kinases (ERKs) control various evolutionarily conserved cellular processes in metazoans, and ERK dysregulation can lead to various diseases [17]. LI J et al [18] revealed that AKR1B10 can promote breast cancer progression by activating ERK signaling. More interestingly, the SwissTargetPrediction website predicted AKR1B10 as a potential target of echinacea. However, whether ECH is involved in breast cancer progression by mediating AKR1B10/ERK signaling is unknown.
This study focused on exploring the specific effects of ECH on the phenotype of BC malignant cells and elucidating the relationship between ECH and the AKR1B10/ERK pathway in BC cells.

1 material
Human normal breast epithelial cell line MCF-10A and human breast cancer cell line MCF-7 were provided by the Cell Resource Center of Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences; MCF-7 ADR-resistant cells were purchased from Shanghai Huzhen Industrial Co., Ltd. DMEM/F1 medium (batch number PM150310), horse serum (batch number 164215), and fetal bovine serum (batch number 164210) were purchased from Wuhan Proceeds Life Technology Co., Ltd.; ECH (batch number 07668) was purchased from Sigma-Aldrich, USA; ADR ( Lot number KGA8181) was purchased from Jiangsu Kaiji Biotechnology Co., Ltd.; RIPA lysis solution (lot number R0020) was purchased from Beijing Soleibao Technology Co., Ltd.; BCA protein quantitative reagent (lot number CX0013S) was purchased from Kangwei Century Biotechnology Co., Ltd.; CCK-8 reagent (batch number C0037) was purchased from Shanghai Biyuntian Biotechnology Co., Ltd.; Lipofectamine 2000 transfection reagent (batch number 11668019) was purchased from Thermo Fisher Scientific Co., Ltd.
Ov-AKR1B10 and Ov-NC plasmids were provided by Chongqing Youbao Biotechnology Co., Ltd.; EdU reagent (lot number C00003) and TUNEL kit (lot number C11026-1) were purchased from Guangzhou Ribo Biotechnology Co., Ltd.; DAPI (lot number 40727ES10) was purchased from Guangzhou Ribo Biotechnology Co., Ltd. from Shanghai Yisheng Biotechnology Co., Ltd.; Matrigel (Lot No. 354230) and transwell chamber (Lot No. 3450) were purchased from Corning Company, USA; Paraformaldehyde (Lot No. 158127), Crystal Violet (Lot No. C0775) and Triton X-100 solution (Lot No. T8787) was provided by Sigma Company in the United States; rabbit AKR1B10 (batch number ab192865), phosphorylated ERK1/2 (p-ERK1/2, batch number ab278538), ERK1/2 (batch number ab184699), E-cadherin (E-cadherin, Lot ab212059), N-cadherin (N-cadherin, lot ab76011), Snail (lot ab216347), B-cell lymphoma-2 (Bcl-2, lot ab182858), Bcl-2 related X protein (Bcl-2-associated X, Bax, batch number ab182733) antibody and HPR-labeled goat anti-rabbit secondary antibody (batch number ab6702) were purchased from Abcam, USA; ECL luminescence kit (batch number SL1350) was purchased from Beijing Cool Lamb Technology Co., Ltd. Image-Pro Plus software was purchased from Media Cybernetics, USA; Leica DM1000 fluorescence microscope and Leica DM ILLED inverted microscope were purchased from Leica Microsystems, Germany; SPSS 22.0 software was purchased from IBM, USA.
2 methods
2.1 Cell Culture and Handling
MCF-10A cells were cultured in DMEM/F12 containing 5% horse serum, 20 ng·mL-1 epidermal growth factor, 0.5 μg·mL-1 cortisol, 10 ng·mL-1 insulin, 100 ng·mL-1 cholera toxin culture medium; MCF-7 and MCF-7/ADR cells were
Cultured in DMEM/F12 medium with 10% FBS. All media were supplemented with 1% double antibody and stored at 37°C in a humidified atmosphere with 5% CO2. Subsequently, MCF-7 cells were intervened with ECH at different concentrations (0, 10, 20, 40 μg·mL-1) for 48 h; and 1.0 mg·L-1 ADR was applied to maintain the drug resistance of MCF-7/ADR cells. sex. 2.2 Western blot method The protein in MCF-7 cells was extracted by RIPA lysate, and the protein concentration was determined by BCA reagent. Proteins separated by 10% SDS-PAGE were transferred using PVDF membrane; after blocking with 5% skimmed milk for 2 h at room temperature, the membrane was /10 000), E-cadherin (1/10 000), N-cadherin (1/5 000), Snail (1/1 000), Bcl-2 (1/1 000), Bax (1/1 000) The primary antibody was incubated overnight at 4 °C, followed by the addition of HPR-labeled goat anti-rabbit secondary antibody (1/2 000) for 1 h at room temperature. The target protein was detected by adding ECL luminescence kit, and the protein abundance was analyzed by Image-Pro Plus software.

Rich in Echinacoside Cistanche Coffee for the treatment of Breast Cancer
2.3 CCK-8 assay for cell viability
MCF-10A and MCF-7 cells were seeded in 96-well plates for culture (5×103 cells/well), and after 24 h, different concentrations (0, 10, 20, 40 μg·mL-1) of ECH were applied After intervention, 48 hours later, they were treated with 10% volume fraction of CCK-8 reagent, and cell viability was detected 2 hours later. In order to further explore the effect of ECH on the viability of MCF-7 cells, transfected or untransfected MCF-7 cells were cultured for 24 h, and different concentrations (0, 10, 20, 40 μg·mL-1) of ECH were used for intervention. , 10% volume fraction of CCK-8 reagent was added at 24, 48, and 72 h, respectively, and the cell viability detection method was the same as the above. To explore the effect of ECH on doxorubicin resistance of MCF-7 cells, MCF-7/ADR-resistant cells were co-cultured with ECH for 48 h, and then CCK-8 working solution was added to detect cell viability.
2.4 Plasmid transfection
MCF-7 cells were seeded and cultured in 6-well plates (5×104/well). When the cells reached 60%~70% confluence, Ov-AKR1B10 and Ov-NC were transiently transfected in strict accordance with the instructions of Lipofectamine 2000 transfection reagent. The cells in each group were transfected, and the transfection was completed after 48 h.
2.5 EdU staining to detect cell proliferation
After MCF-7 cells were treated with ECH and transfected with plasmids, 20 µmol·L-1 EdU was added to each well and incubated at 37 °C for 2 h. Subsequently, cells were fixed and permeabilized with 4% paraformaldehyde and 0.5% Triton X-100, respectively. Finally, the EdU-positive cells stained with DAPI were detected under a fluorescence microscope.
2.6 Wound healing assay to detect cell migration
MCF-7 cells in log phase of growth were added to 6-well plates, and when the cells reached 90% confluence, use a 200 µL sterile pipette tip to make a streak and wash 3 times with PBS. The wound width was measured at 0 and 48 h under an inverted microscope, and the number of transferred cells was determined.
2.7 Transwell assay to detect cell invasion
The ECH-treated and plasmid-transfected MCF-7 cells were seeded in the upper chamber of a transwell containing Matrigel, and 500 µL of DMEM medium containing 10% FBS was added to the lower chamber of the transwell. After 24 h, the surface cells were wiped off with cotton swabs, and the cells were fixed and stained with paraformaldehyde and crystal violet, respectively, and the invasive cells were counted under a microscope.
2.8 TUNEL staining to detect apoptosis
After intervention with 15 µmol L-1 ADR, ECH-treated and plasmid-transfected MCF-7/ADR-resistant cells were seeded in 96-well plates and fixed with 4% paraformaldehyde and 0.5% Triton X-100 After the permeabilization treatment, TUNEL detection solution was added, and the reaction was carried out at 37 °C for 45 min under the guidance of the kit instructions, and DAPI cell nuclei were stained. Five visual fields were randomly selected to observe cell apoptosis under a fluorescence microscope.
2.9 Molecular docking
The three-dimensional structure of AKR1B10 protein (PDB ID: 1ZUA) was obtained from the PDB database (https://www.rcsb.org/). Use PyMOL 2.2.0 software to remove water from active ingredients and remove irrelevant original ligands, then convert the chemical structure of ECH into mol2 format by OpenBabel 2.2.1 software (http://openbabel.org/wiki), and finally use Autodock 4.2 performs molecular docking, and selects the pose with the lowest free energy (-10.8 kcal·mol-1) for visualization.

2.10 Statistical analysis
In this study, SPSS 22.0 software was used to analyze and process the experimental data and expressed as mean ± standard deviation (x ± s). One-way analysis of variance was used for comparison between groups. When P<0.05, the difference was statistically significant.
3 results
3.1 ECH inhibits AKR1B10/ERK signaling in MCF-7 cells First, the results of Western blotting revealed that with the increase of ECH exposure dose, the protein levels of AKR1B10 and pERK1/2/ERK1/2 in MCF-7 cells decreased, and the downward trend was dose-dependent P<0.001) (Fig. 1). It can be speculated that ECH may participate in the malignant progression of BC by regulating AKR1B10/ERK signaling.

Fig.1 Effect of echinacoside on AKR1B10/ERK signal transduction in MCF-7 cells (x̄ s, n = 3)
3.2 Overexpression of AKR1B10 reversed the inhibitory effect of ECH on MCF-7 cell proliferation
The experimental data of CCK-8 showed that, compared with MCF-10A normal cells, exposure to different concentrations of ECH (0, 10, 20, 40 μg·mL-1) resulted in a concentration-dependent decrease in the activity of MCF-7 cancer cells (P< 0.05), and when the mass concentration of ECH was 40 μg·mL-1, the cell activity decreased most significantly, so 40 μg·mL-1 ECH was used for subsequent experiments. To further verify whether ECH regulates BC development by regulating AKR1B10/ERK signaling, MCF-7 cells were first transfected with the Ov-AKR1B10 plasmid. After transfection of Ov-AKR1B10, the expression level of AKR1B10 was significantly increased (P<0.001). Western blotting results showed that transfection of Ov-AKR1B10 plasmid induced ECH exposure-induced reduced AKR1B10 and p-ERK1/2 protein levels again increased (P<0.001). In addition, the experimental data of CCK-8 and EdU demonstrated that the inhibitory effect of ECH treatment on the proliferation of MCF-7 cells could be alleviated by the up-regulation of AKR1B10 (P<0.05) (Fig. 2). Thus, overexpression of AKR1B10 rescued the inhibitory effect of ECH on the proliferative capacity of BC cells.

Fig.2 Effect of echinoside on the proliferation of MCF-7 cells by regulating AKR1B10 expression (fluorescence staining, ×200; x̄ s, n=3)
3.3 Overexpression of AKR1B10 reversed the inhibitory effect of ECH on MCF-7 cell migration Similarly, compared with the control group, ECH administration resulted in a significant decrease in the number of cell migration and invasion (P<0.001); In contrast, increased expression of AKR1B10 resulted in enhanced cell migration and invasion (P<0.05). In addition, compared with the control group, the protein levels of N-cadherin and Snail were significantly decreased after ECH treatment, while the level of E-cadherin was significantly increased (P<0.001); The protein levels of cadherin and Snail increased, while the level of E-cadherin decreased (P<0.001) (Fig. 3). In conclusion, up-regulation of AKR1B10 expression rescued the inhibitory effect of ECH administration on BC cell migration and invasion.
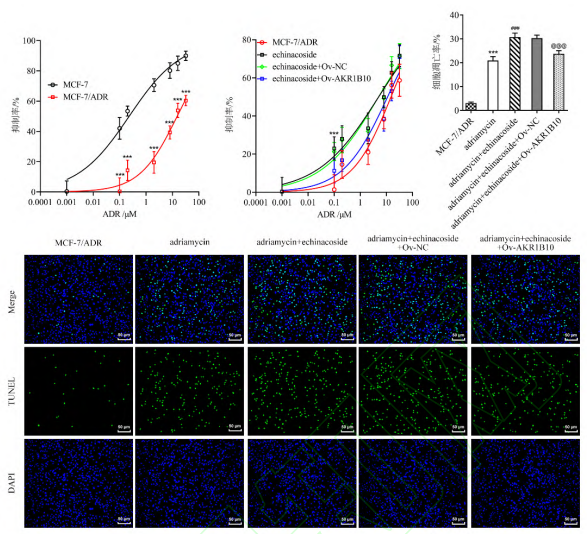
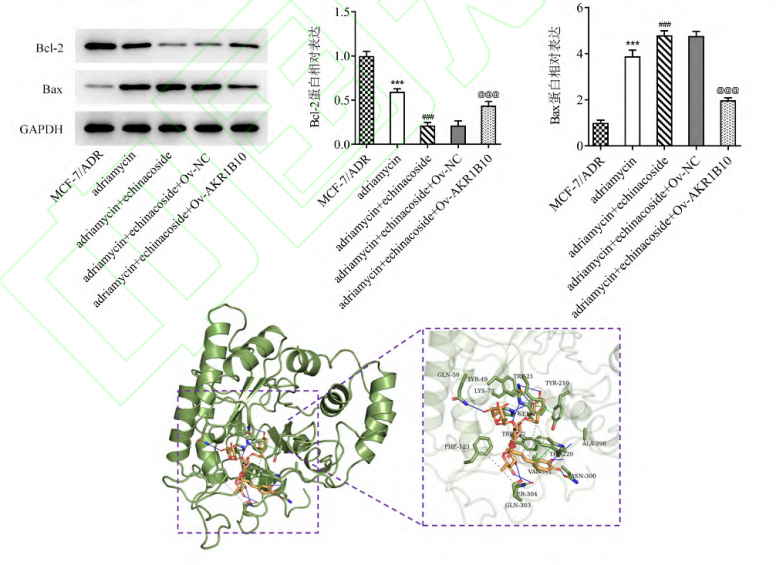
Fig.4 Effect of echinoside on ADR resistance of MCF-7 cells by regulating AKR1B10 expression (fluorescence staining, ×200; x̄ s, n=3)
4 Discussion
Breast cancer refers to the uncontrolled proliferation of mammary epithelial cells under the action of various carcinogenic factors, and it is also the main cause of cancer death in women [1]. Tumor metastasis is one of the most threatening characteristics of tumors, and it is also the root cause of long-term cure of tumors [19]. Chemotherapy is a common method for clinical treatment of breast cancer [20]. With the continuous progress and update of breast cancer treatment methods and concepts, the prognosis of breast cancer has been significantly improved [20]. However, the emergence of drug resistance greatly limits the clinical effectiveness of drugs [21]. Studies have found that about 90% of metastatic breast cancer patients and 50% of advanced breast cancer patients will develop drug resistance [22]. ADR is an anthracycline antibiotic that is widely used in the clinical treatment of metastatic or recurrent breast cancer by acting on the chemical structure of DNA, and can effectively reduce the recurrence rate and mortality of breast cancer patients [23]. Therefore, this study focused on the proliferation, metastasis and ADR resistance of BC cells, so as to deeply uncover the regulatory mechanism of BC progression and search for potential molecular targets for intervention.

Echinacea is a phenylethanoid compound derived from the rhizomes of natural herbs Cistanche deserticola and Echinacea purpurea. It has various pharmacological effects such as antioxidant, antidepressant, anti-inflammatory, antiviral, antitumor and antibacterial [24-25]. Recent studies have reported that ECH can inhibit the proliferation, metastasis and angiogenesis of ovarian cancer cells by regulating the PI3K/Akt pathway [26]; ECH can exert its anticancer activity in liver cancer by activating the TGF-1/Smad pathway [27]. ECH can hinder BC tumor growth by inactivating the Wnt/β-catenin signaling pathway [11]. In this study, AKR1B10 was first predicted to be a potential target of ECH by SwissTargetPrediction. Therefore, the authors speculate that ECH may participate in BC disease progression by regulating AKR1B10-related signaling pathways. Up to now, it has been found that AKR1B10 is increased in expression in various cancers and plays a role in promoting cancer [28]. In addition, AKR1B10 can aggravate the development of bladder cancer, lung adenocarcinoma, and BC by activating ERK signaling [18, 29-30]. Ectopic expression of AKR1B10 in breast cancer cells promotes the phosphorylation of ERK 1/2 to activate the ERK signaling pathway [18]. The ERK signaling pathway promotes the degradation of extracellular matrix by activating MMPs, especially MMP2, thereby promoting tumor cell invasion and tumor metastasis [31-33]. The results of this experiment also showed that the expression of AKR1B10/ERK pathway-related proteins decreased in a dose-dependent manner after treatment with different concentrations of ECH, indicating that ECH may participate in the regulation mechanism of BC by inhibiting the AKR1B10/ERK pathway. In addition, ECH exposure also resulted in varying degrees of reduction in BC cell viability. To further prove this point, this study transfected AKR1B10 overexpression plasmid into MCF-7 BC cells and found that the inhibitory effects of ECH treatment on AKR1B10/ERK pathway and cell proliferation, migration and invasion, and epithelial-mesenchymal transition were overexpressed by AKR1B10 rescued. Furthermore, in MCF-7/ADR-resistant cells, ECH exposure resulted in a decrease in IC50 and an increase in the number of apoptotic cells; whereas overexpression of AKR1B10 in this setting again increased IC50 and reduced apoptotic cell production. At the end of the experiment, the targeting relationship between ECH and AKR1B10 was also fully verified. The above experimental results show that,
AKR1B10 overexpression achieves an antagonistic effect on ECH-suppressed BC development.
In conclusion, ECH administration can attenuate the proliferation, migration, invasion, EMT and ADR resistance of BC cells, and the mechanism may be related to targeting AKR1B10 and inhibiting the AKR1B10/ERK signaling pathway. This study initially clarified the potential mechanism of ECH in BC, and provided valuable experimental basis and theoretical support for the application of ECH in anti-BC drugs. However, there are still some shortcomings in this study. First, this experiment did not involve the analysis of the impact of ECH on the survival rate and prognosis of BC patients; secondly, this study lacked in vivo animal experiments to further verify the role of ECH in the growth of BC tumors. In future studies, the clinical significance of ECH in breast cancer treatment will be further explored.
Support:
wallence.suen@wecistanche.com 0015292862950






