PART Ⅰ: Kidney-Targeted Epoxyeicosatrienoic Acid Analog, EET-F01, Reduces Inflammation, Oxidative Stress, and Cisplatin-Induced Nephrotoxicity
Mar 24, 2022
Contact: joanna.jia@wecistanche.com / WhatsApp: 008618081934791
John D. Imig, Md Abdul Hye Khan & et al.
1. Introduction
Renal disease in cancer patients and survivors has emerged as a major healthcare issue and continues to escalate sharply [1–3]. Newer cancer therapies have increased cure rates and survival times, but therapy-associated fluid and electrolyte abnormalities and acute kidney injury progressing to chronic kidney disease are common sequelae [3–5]. Anti-cancer drugs, vascular endothelial growth factor receptor tyrosine kinase inhibitors (VEGFRI), and platinum derivatives can also cause severe renal injury that significantly compromises their effective and safe use [3–5]. There is a significant clinical need for novel therapies to effectively treat and stop kidney disease in cancer patients. This healthcare issue has led to the development, evolution, and emergence of a subspecialty, Onco Nephrology [5]. Current treatments only slow the loss of kidney function or have no benefit at all [2,5,6]. New approaches are urgently needed. Our ongoing research is attacking this emerging cancer arena and we are focused on developing epoxyeicosatrienoic acid (EET)analogs to protect the kidney from cancer therapy-associated toxicity.

bioflavonoids tablets and cistanche for kidney function
EET analogs are based on the pharmacophoric moiety of EETs and designed with improved solubility and resistance to auto-oxidation and metabolism by soluble epoxide hydrolase (sEH)[7,8]. Previous studies have demonstrated that synthetic EET analogs are kidney protective in several pathological conditions like hypertension, drug-induced nephrotoxicity, and lupus nephritis [9-13]. EET analogs decrease kidney damage in these disease states through vasodilator, diuretic, anti-inflammatory, anti-oxidative, and anti-apoptotic activities[10-13]. These studies clearly demonstrated that an EET-based approach combats kidney damage in pathological situations of different etiologies. Recently, attempts have been made to develop EET analogs that possess the unique property to target the kidney. A novel form of kidney-targeted EET analog was designed and synthesized by conjugating the EET pharmacophore with folic acid (EET-F01). In the present study, we investigated the ability of EET-F01 to target the kidney when compared to the EET analog, EET-A. EET-FO1 and EET-A were compared for their ability to decrease cisplatin-induced kidney injury. Lastly, this study further delineated the kidney protective anti-inflammatory and anti-oxidative actions of EET-F01.

cistanche benefits for kidney
2. Results
2.1. EET-F01 Targets Kidney Tissue
Chemical structures for EET-A and EET-FO1 are provided in Figure 1. Purity for EET-A and EET-F01 was >95% as determined by NMR. In the acute animal study, equal doses by weight of EET-A and EET-F01 were administered intravenously for 6 h followed by plasma and kidney tissue collection. We demonstrated a 5-fold higher plasma level of EET-F01 compared to EET-A after 6-h continuous administration (Figure 1). Interestingly, we also demonstrated that after 6-h continuous administration the kidney content of EET-A was almost un-detectable compared to EET-F01 content in the kidney (Figure 1). These data demonstrate that EET-F01 targets the kidney.

Figure 1. Comparison of plasma and kidney EET-F01 and EET-A following i.v. administration for 6h. Top panel: Chemical structures for EET-A and EET-F01. Left panel: Plasma concentration of EET-FO1 and EET-A. Right panel: Kidney levels of EET-F01 and EET-A. Data expressed as mean±SEM, n=3/group.
2.2. EET-F01 Protects against Cisplatin Nephrotoxicity
In a separate set of experiments, groups of rats were pre-treated with EET-F01 and EET-A for two days prior to induction of cisplatin nephropathy and then further treated with the test compounds for another 5 days. The kidney protective actions of EET-F01 and EET-A were determined from BUN levels, urinary NAG content, and urinary KIM-1 levels. EET-F01 was as effective as EET-A in decreasing BUN, NAG, KIM-1, and renal histological injury five days following induction of cisplatin nephrotoxicity (Figure 2). Next, histological examination was conducted to further assess cisplatin nephrotoxicity. Cisplatin administration resulted in tubular injury manifested by vacuolation and desquamation of the renal epithelial cells along with severe intra-tubular proteinaceous cast formation in both cortical and medullary regions of the kidney. EET-A and EET-F01 treatment protected the kidney from cisplatin nephrotoxicity in rats(Figure 2).EET-F01 was effective at 20 mg/kg/d and at a 10-fold lower dose of 2 mg/kg/d despite its almost 2×-greater molecular weight due to the folate compared with EET-A. These data clearly demonstrate that EET-F01 targets the kidney and facilitates a lower, yet still efficacious dose. Thus, we have developed a kidney-targeted EET analog, EET-F01, that demonstrates excellent potential as a therapeutic for kidney diseases.

cistanche deserticola benefits
Figure 2. Kidney injury induced by cisplatin is reduced by EET-F01 and EET-A treatments. Left panel: blood urea nitrogen (BUN), Middle panel: urinary N-acetyl-β-(D)-glucosaminidase (NAG), Right panel: kidney injury molecule-1 (KIM-1) in cisplatin administered rats treated with vehicle, EET-FO1, or EET-A. Bottom panel: Representative photomicrographs of Periodic acid-Schiff (PAS)Staining depicting vacuolation and desquamation of the renal epithelial cells along with severe intra-tubular proteinaceous cast tubular cast formation in the renal sections of different experimental groups.*p<0.05 vs. control-vehicle group;t p<0.05 vs. cisplatin-vehicle group.Data expressed as mean ±SEM, n=6/group.
2.3. EET-F01 and EET-A Reduce Renal Inflammation and Oxidation Stress in Cisplatin Nephrotoxicity
Renal IL-6 and TNFo mRNA expression levels revealed a 4 to 5-fold increase in cisplatin-induced nephrotoxicity. EET-F01 and EET-A treatment reduced renal inflammatory genes IL-6 and TNFα by 30-50% in cisplatin nephrotoxicity (Figure 3). Increased renal expression of the oxidative marker genes NADPH oxidase subunits p47phox, p67phox, gp91phox, and NOX4 is observed in cisplatin administered rats. Kidney tissue TBARS levels were increased during cisplatin-induced nephrotoxicity. These cisplatin-induced increases in oxidative stress were attenuated60-100% by EET-F01 and EET-A treatment (Figures 4 and 5). These data demonstrate that kidney-targeted EET-F01 is as effective as EET-A in reducing inflammation and oxidative stress in cisplatin nephrotoxicity.
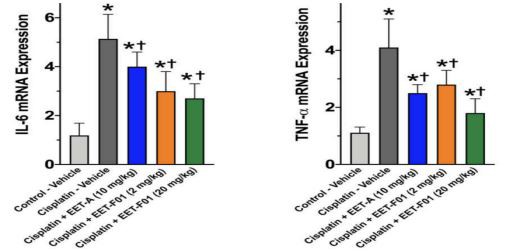
Figure 3. Renal inflammation in cisplatin nephrotoxicity is reduced by EET-F01 and EET-A treatments. RT-PCR analysis for mRNA expressions of IL-6(left panel) and TNFα(right panel).*p<0.05 vs. control-vehicle group; tp<0.05 vs. cisplatin-vehicle group. Data expressed as mean±SEM, n=6/group.
2.4. EET-A Does Not Enhance Tumor Growth
The use of EET analogs might promote cancer metastasis when administered for long periods of time [14,15]. Although the current therapeutic approach would administer EET analogs for a short period of time to prevent cisplatin-induced nephrotoxicity, we evaluated the long-term effects of EET-A on tumor growth of a human breast cancer cell line, MBA-MB-231, in athymic mice. Tumor volume and weight increased at the same rate in vehicle or EET-A treated mice over a three-week period (Figure 6). Histological evaluation of the tumors failed to demonstrate any changes in cellular hyperplasia or proliferation in EET-A-treated mice. These data demonstrate that EET-A does not enhance tumor growth.
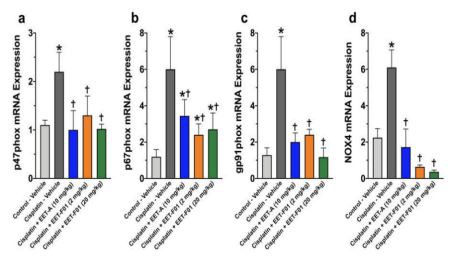 | 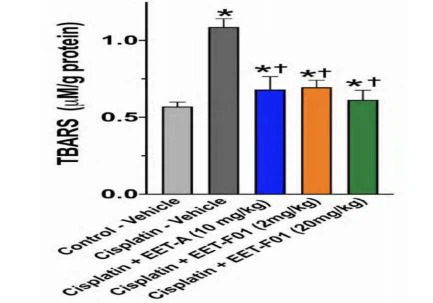 |
| Figure 4. Renal oxidative stress in cisplatin nephrotoxicity is reduced by EET-F01 and EET-A treatments. RT-PCR analysis for mRNA expressions of (a)p47phox, (b)p67phox, (c)gp91phox, (d) NOX4.*p<0.05 vs.control-vehicle group;tp<0.05 vs. cisplatin-vehicle group. Data expressed as mean ± SEM, n = 6/group. | Figure 5. Kidney thiobarbituric acid reactive substances (TBARS)in cisplatin nephrotoxicity are reduced by EET-F01 and EET-A treatments.*p<0.05 vs.control-vehicle group;+p<0.05 vs. cisplatin-vehicle group. Data expressed as mean ± SEM, n=6/group. |

Figure 6. Tumor growth of a human breast cancer cell line, MBA-MB-231, in athymic mice is not altered by EET-A treatment. Left panel: tumor volume, Right panel: tumor weight in MBA-MB-231 tumor-bearing mice treated with vehicle (control)or EET-A. Bottom panel: Representative photomicrographs of Periodic acid-Schiff (PAS) Staining depicting similar tumor hyperplasia and proliferation in human breast tumors. Data expressed as mean± SEM,n=5 mice and n=10 tumors/group.
CLICK HERE TO PART Ⅱ

cistanche in hindi





