Gut Microorganisms And Neurological Disease Perspectives Part 3
Jun 12, 2024
Another study by Parracho et al. using fluorescent in situ hybridization showed elevated levels of Clostridium hystolyticum in children with ASD compared with healthy children [85].
Clostridium histolyticum is a common bacterium that grows in the human body and is usually found in the mouth, intestines, etc. Studies have shown that there is a certain correlation between the level of Clostridium histolyticum and memory.
In the brain, there are a large number of microorganisms that form the so-called "gut-brain axis" and affect human health and cognitive function. Studies have shown that Clostridium histolyticum is closely related to brain activity. It can affect the biochemical reactions and neuroregulation of the brain through various pathways, and these effects in turn affect human cognitive function.
Among them, Clostridium histolyticum can promote the production of serotonin, a neurotransmitter that can affect emotions and cognition. In addition, Clostridium histolyticum can also affect the function of gamma-aminobutyric acid (GABA) receptors in the brain. GABA is one of the most important neurotransmitters in the brain. It can inhibit the activity of neurons and regulate the excitability and inhibition of the brain. This means that changes in the level of Clostridium histolyticum may directly affect the function of the brain, thereby affecting our cognitive ability.
A study published in 2017 showed that compared with subjects with normal levels of Clostridium histolyticum, subjects with lower levels of Clostridium histolyticum performed worse in cognitive tasks, especially in attention and working memory. This conclusion suggests that the level of Clostridium histolyticum may be a key factor that can affect human cognitive function and memory ability.
Accordingly, by changing diet, using probiotics, reducing stress, etc., people may be able to improve memory and cognitive function by adjusting the level of Clostridium histolyticum. At the same time, we also need to pay attention to the fact that any serious infection, medication, and external factors may affect the balance of intestinal microflora, so maintaining good living habits, especially a healthy diet and mental health, is essential to effectively maintain the balance of intestinal microecology.
In short, although the level of Clostridium histolyticum is related to memory, we should actively seek solutions, improve our living habits, keep ourselves away from negative influences, give full play to our potential, maintain good memory and cognitive ability, and better cope with various challenges. It can be seen that we need to improve memory, and Cistanche can significantly improve memory because it can also regulate the balance of neurotransmitters, such as increasing the levels of acetylcholine and growth factors, which are very important for memory and learning. In addition, Cistanche can also improve blood flow and promote oxygen delivery, which can ensure that the brain obtains sufficient nutrition and energy, thereby improving brain vitality and endurance.

Click know supplements to improve memory
Clostridia, which are known propionate producers, have been thought to be inducers of autism since they contribute to neurological changes in rat models [86,87].
It has also been seen that pregnant mice with intestinal bacteria-induced activation of the maternal immune system (also known as maternal immune activation) produce offspring with ASD symptoms of impaired sociability and repetitive marble-burying behaviors [85,88].
Another study observing the prevalence of four types of beneficial bacteria – Bifidobacterium, Lactobacillus spp., E coli, and Enterococcus – revealed that ASD children had much lower levels of Bifidobacterium, slightly lower levels of Enterococcus and much higher levels of Lactobacillus, with more Bacillus spp. and less Klebsiella oxytoca [89].
The same study revealed that lower levels of SCFAs were found in ASD children, and a possible reason could be the lower saccharolytic fermentation by beneficial bacteria. This further adds to the link between the gut microbiome and autism [90].
Following the fact that the abundance of bacteria-produced metabolites has the potential to directly affect neural processes, the increased urinary excretion of an abnormal phenylalanine metabolite of Clostridia species known as HPHPA was found in ASD patients [74].
Through the depletion of catecholamines, Clostridia species influence the symptoms of autism, including stereotyped behaviors, hyperactivity, and hyper-reactivity, in experimental animals.
Furthermore, a study by Kang et al. using 16S rRNA gene pyrosequencing analysis of fecal DNA samples revealed lower levels of Prevotella, Coprococcus, and unclassified Veillonellaceae in children with ASD [91].
In addition to a leaky gut and incorporation of certain foods into the diet of a person, metabolites produced by microorganisms in the gut can influence possible ASD-like symptoms in individuals.
Moreover, an abundance of or decrease in certain microorganisms in the gut is known to be a potential contributor to neurological disorders such as ASD, a link that is further strengthened with the help of various hypotheses.
Multiple sclerosis
MS is a chronic inflammatory and demyelinating neurodegenerative disorder that affects the CNS and results in characteristic brain and spinal cord lesions [92–95]. This disorder may lead to fatigue, numbness, loss of coordination, vertigo, vision loss, dizziness, pain, bladder and bowel dysfunction, and even depression [96]. Approximately 2.1 million people are affected by MS worldwide.
MS also leads to an increase in the number of autoreactive immune cells that target the CNS [97]. Since the gut microbiota contributes to educating the immune system and plays a key role in several autoimmune and metabolic disorders, it is reasonable to link gut commensal flora to MS susceptibility.
In a study by Vartanian et al., human gut colonization by Clostridium perfringens type B in a patient with her first relapse of MS was demonstrated, and this revealed that the epsilon toxin released by this pathogen led to microangiopathy, similar studies showed this can further cause BBB disruption, leading to neuronal and oligodendrocyte damage [98–102].
When MS patients were compared with controls, they were seen to have an increased prevalence of antibodies against epsilon toxin in their sera. Similarly, Jhangi et al. demonstrated an increase in the concentration of the archaea Methanobrevibacter in MS patients [103].
There was also a notable reduction in the concentration of Butyricimonas, Lachnospiraceae, and Faecalibacterium in MS patients compared with controls, and the reduced abundance of Faecalibacterium was noted in the microbiome of inflammatory bowel disease patients [104,105].
Faecalibacterium prausnitzii bacteria are butyrate-producing organisms that lead to an increased Treg cell population; this may link the alterations in the gut microbiota to MS development.
Another study revealed differing levels of Firmicutes, Bacteroidetes, and Proteobacteria in MS patients, and a vitamin D treatment led to increases in Enterobacteriaceae in both healthy controls and MS patients.
Furthermore, a study by Tremlett et al. showed increased levels of Shigella, Escherichia, and Clostridium, which are associated with infection and inflammation, and decreased levels of Eubacterium rectale and Corynebacterium when 20 pediatric MS patients were examined [106].
Baranzini et al.examined 71 MS patients and 71 healthy controls using 16S rRNA gene sequencing, which revealed that patients with MS had higher levels of Akkermansia muciniphila and Acinetobacter calcoaceticus and lower levels of Parabacteroides distasonis in the gut compared with healthy controls [107,108].
The bacterial extracts of species from the gut microbiota of patients with MS strengthened the proinflammatory Tcell response in an in vitro model [109]. Following this, when the researcher transplanted gut bacteria from an autoimmune encephalomyelitis model (an artificially induced mouse model of MS), it was observed that the MS microbiota made encephalomyelitis worse.
Several studies have shown that compared with healthy controls, a decrease or increase in certain gut microorganisms may be related to the development of MS. Moreover, an elevated level of certain antibodies produced as a result of toxins released by microorganisms has also been observed in MS patients.
Alzheimer's disease
AD is a progressive neurodegenerative disorder that includes pathophysiological signs such as impaired cognition and the accumulation of atypical amyloid beta (Aβ) protein within the interstitial space of brain tissues, leading to memory loss [110,111].
About 44 million people are diagnosed with AD worldwide, and it is more likely to affect individuals over 65 years of age. The symptoms of AD are known to worsen over time and lead to difficulties with language, disorientation, mood swings, loss of motivation, and failure to manage self-care.
The gut microbiota contributes greatly to maintaining the host's normal physiology and functions; therefore, changes in the gut microbiota can lead to changes in brain function followed by changes in host behavior [112].
AD remains the most common cause of dementia, with 36 million cases reported in 2010, a number that is predicted to increase to 66 million by 2030 and 115 million by 2050 [113].
The etiology of AD is elusive and is explained mostly as an interaction between genetic and environmental factors [114]. A study by Bray et al. stated that the human gut commensal microbiota modulates brain function and behavior via the microbiota–gut–brain axis and therefore could have a role in AD mechanisms [115].
Following this, another study involving germ-free animals and animals exposed to pathogenic microbial infections, antibiotics, probiotics, and fecal microbiota transplantation revealed a role for the gut microbiota in host cognition and AD-related pathogenesis [116].
The increased permeability of the gut and BBB due to microbiota dysbiosis as well as imbalances in the gut microbiota influences AD pathogenesis and other neurodegenerative disorders.
Moreover, bacteria present in the gut microbiota can secrete large amounts of amyloids and LPSs, which might contribute to the modulation of signaling pathways and the production of proinflammatory cytokines, which may lead to AD [116].
Furthermore, several recently conducted rodent studies suggest a link between gut microbiome alterations and amyloid deposition; however, the microorganisms associated with AD have not yet been characterized in humans [117,118]. DNA was isolated from fecal samples for comparison of the composition of the gut microbiome in participants with and without a diagnosis of dementia due to AD.
This study conducted bacterial 16S rRNA gene sequencing on the DNA isolated and it was revealed that decreased microbial diversity and a distinct composition in control age- and sex-matched individuals of AD patients' microbiome [119].
Aging is also a major risk factor for AD and can trigger the overstimulation of the innate and adaptive immune system, leading to inflammation, which increases gut permeability and bacterial translocation [120–123].
The composition of the gut microbiota changes in the elderly, with a decrease in certain bacteria that are considered beneficial, including Bacteroidetes, Lactobacillus, and Bifidobacterium [124–126]. Several bacterial groups, including Bifidobacterium, Lactobacillus, and Faecalibacterium, may modulate inflammation at the level of the gut epithelium [127–130].

Since the BBB is compromised as a result of age, it influences not only the clearance of Aβ from the brain but also the secretome and receptor-mediated signaling involved in the neuroinflammation that is observed in AD.
Therefore, it can be stated that age can alter the gut microbiota and may facilitate inflammatory processes that contribute to neuroinflammatory effects in AD [131–135]. Bacteria that inhabit the gut may excrete huge quantities of LPSs and amyloids that may contribute to AD pathogenesis as a result of increased permeability of the GI tract epithelium and BBB with age.
LPSs are an important component of the outer membrane of gram-negative bacteria and have been seen to induce the inflammatory and pathological characteristics observed in AD when infused into the fourth ventricle of experimental rat models [136]. Moreover, a study carried out in vitro suggested that bacterial LPSs potentiate the fibrillogenesis of Aβ peptides [137].
In another study, mice that were treated with multiple intraperitoneal injections of LPSs were seen to have higher levels of Aβ in the hippocampus as well as cognitive deficits [138]. Jaeger et al. stated that intraperitoneal injections of LPSs increased the blood-to-brain influx and decreased the brain-to-blood efflux in mice, leading to Aβ accumulation [139].
Similarly, in a study involving Bacteroides fragilis, exposure of LPSs to human primary brain cells revealed that they were an extremely potent inducer of a proinflammatory transcription factor that involved a trigger that facilitated inflammatory neurodegeneration in the AD brain [140].
Furthermore, a major quantity of functional amyloid can also be generated along with LPSs by many bacterial strains, including E coli, Bacillus subtilis, Salmonella typhimurium, Salmonella enterica, Mycobacterium tuberculosis, and Staphylococcus aureus, and may contribute to AD pathology through the accumulation of misfolded proteinaceous Aβ oligomers and fibrils [141,142].
It has also been hypothesized that bacterial amyloids might leak from the GI tract and contribute to the systemic and CNS amyloid burden. These amyloids may induce proinflammatory cytokines, which pass through the GI tract and BBB to the brain, triggering immunoreactivity and signaling of several components that contribute to neurodegeneration [143–145].
LPSs and amyloids produced by microorganisms in the gut trigger inflammatory effects that may contribute to AD pathogenesis, aiding the accumulation of Aβ.
Schizophrenia
Schizophrenia is a complex and debilitating brain disorder that results in a combination of behavioral abnormalities, including hallucinations, delusions, apathy, relapsing episodes of psychosis, and extremely disordered thinking [146,147]. Schizophrenia is known to affect around 21 million people worldwide and leads to social and physical morbidity [148,149].
The causes of schizophrenia include both environmental and genetic factors. The symptoms of schizophrenia usually start between the ages of 16 and 30, and males are more likely to be affected by the disease.
A study by Nemani et al. represented a link between gut microbiome disruption and schizophrenia [150]. Schwarz et al. observed that when patients with first-episode psychosis were compared with nonpsychiatric comparison subjects, they were found to have an increased abundance of the families Lactobacillaceae, Halothiobacillaceae, Brucellaceae, and Micrococcineae and a decreased abundance of the family Veillonellaceae [151].
The intestinal microbiota also plays an important role in regulating neurotransmission, immune homeostasis, and brain development, and an imbalance in the microbiome may lead to immune activation and dysfunction in the gut-brain axis, further contributing to schizophrenia.
Changes in the gut microbiota may reduce protectants and increase neurotoxins and inflammatory mediators, causing neuronal and synaptic damage, which induces schizophrenia. Clostridium sporogenes produces the metabolite indole propionic acid by tryptophan degradation, which is essential for protecting the intestinal barrier and maintaining homeostasis of monocytes/macrophages and T cells [152].
The bacterial endotoxin LPS can damage the intestinal mucosal barrier and activate the immune system, causing mild endotoxemia [153]. Moreover, SCFAs are a major component in maintaining intestinal immunity and microglia-mediated central immunity [154,155], which help to maintain immune homeostasis; however, dysfunction in one can affect the other, with both influencing the development of schizophrenia [156].
In addition, schizophrenia is linked to elevated levels of IL-6, IL-8, and TNF-α as well as reductions in antiinflammatory IL-10 [157]. Increased production of antibodies in response to Saccharomyces cerevisiae, which is known to be a marker of intestinal inflammation and elevation in the bacterial translocation marker sCD14 have been identified in patients with schizophrenia [158,159].
The gut microbiome also plays a role in the regulation of BBB permeability; therefore, dysbiosis may lead to CNS infection and inflammation, facilitating the disease [57160]. Furthermore, an absence of GI microbes leads to a reduction in central BDNF levels, inhibiting the maintenance of N-methyl-D-aspartate receptor (NMDAR) production.
A reduction of NMDAR input in GABA inhibitory interneurons further aids glutamatergic output and leads to aberrant synaptic behavior and cognitive deficits. Since NMDAR is thought to be associated with the development of schizophrenia and other neurological diseases, the alteration in microbiome composition may help explain the development of the disease [161].
In another study noting a link between gut microbes and schizophrenia, researchers transferred gut microbe samples of schizophrenic subjects into the gut biomes of a group of healthy control mice [162].
Transplanting the gut bacteria of schizophrenic subjects into healthy control mice induced some symptoms that were characteristic of murine schizophrenia, including glutamatergic hypofunction.
The mice models also showed lower glutamate and higher glutamine and GABA levels in the hippocampus of the brain. This provided further proof of an association between changes in gut microbiota composition and schizophrenia that was specific to the disease and also correlated with symptom severity. An imbalance in the microbiome leads to immune activation and neuronal dysfunction, both of which are known to be probable causes of schizophrenia development.
In addition, studies have shown that a decrease in protectants and an increase in neurotoxins could lead to the neuronal and synaptic damage observed in schizophrenic patients.
Parkinson's disease
Parkinson's disease (PD) is a long-term degenerative disorder of the CNS that mainly affects dopamine-producing neurons present in a specific region of the brain known as the substantia nigra. PD affects approximately 7–10 million individuals worldwide and is known to be 1.5 times more prevalent in men than women. The incidence of PD increases with age, affecting about 1% of individuals 60 years of age and older [163].
The disease is characterized by several symptoms, which include akinesia, muscular rigidity, tremors, bradykinesia, and difficulty with walking and gait [164]. In addition to these motor traits, other symptoms include dementia, depression, and sensory and autonomic dysfunction. Apart from dopaminergic loss, PD is also characterized by synucleinopathy; that is, deposition of insoluble polymers of α-synuclein in the neuronal body, forming round, lamellated, eosinophilic, cytoplasmic inclusions called Lewy bodies [165].
These Lewy bodies are responsible for neurodegeneration and neuronal death [166,167]. Several studies have demonstrated changes in the gut microbiota of patients with PD, which can serve as both biomarkers of PD and its possible trigger-inducing misfolding of α-synuclein that launches neurodegeneration in individuals with PD [168].

PD patients are also known to frequently exhibit signs and nonmotor symptoms of gastrointestinal dysmotility, which may include delayed gastric emptying and constipation [169–173]. In one study, the gut microbiota of patients with PD and healthy volunteers was analyzed using high-throughput 16S rRNA sequencing of bacterial genomes [174].
Patients with PD showed reduced amounts of Bacteroides massiliensis, Stoquefichus massiliensis, Bacteroides copro cola, Blautia glucanase, Dorea long catena, Bacteroides dorei, Prevotella copri, Coprococcus eutectic and Ruminococcus callidus, Christensenella minute, Lactobacillus mucosae, Ruminococcus bromine, and Papillibacter cinnamivorans.
This pattern of gut microflora can trigger local inflammation followed by aggregation of α-synuclein and generation of Lewy bodies. Another study on PD patients with intestinal inflammation showed that they exhibited an increase in expression of the bacterial endotoxin-specific ligand TLR4 as well as CD3+ T cells and cytokine expression in colonic biopsies and a decrease in SCFA-producing colonic bacteria compared with controls [175].
The association between PD and Helicobacter pylori is the most extensively studied, and H pylori infection has been seen to be high among PD patients, causing motor impairments by hindering the absorption of the drug levodopa, which is used for PD management [176,177]. Similarly, small intestinal bacterial overgrowth, a disorder of excessive bacterial growth in the small intestine, has been observed to be associated with PD as well.
In another study, a significant reduction in Prevotellaceae in the stools of PD patients compared with controls demonstrated further evidence that gut microbiome dysbiosis is related to PD [178]. Additionally, a direct correlation was found between the abundance of Enterobacteriaceae and the severity of postural instability and gait difficulty.
PD patients have also been observed to suffer from increased mucosal permeability and systemic exposure to endotoxins from coliform bacteria [179]. Furthermore, bacteria belonging to the genera Blautia, Coprococcus, Faecalibacterium, and Roseburia were significantly reduced in the feces of PD patients compared with controls, and those belonging to the genus Ralstonia were significantly increased in the mucosa of PD patients. In another study of 34 PD patients and 34 age-matched controls, fecal samples were analyzed using gas chromatography to check for concentrations of SCFAs, which are one of the main metabolic products of gut bacteria and microbiota composition [180].
The SCFA concentrations were found to be significantly reduced in the PD patients compared with the controls. This reduction may induce alterations in the ENS and contribute to gastrointestinal dysmotility in PD patients. Studies show that alterations can be induced through changes in levels of microorganisms or the metabolites produced by them. These alterations lead to the gut dysbiosis observed in PD patients as well as the aggregation of α-synuclein, which further generates Lewy bodies.
Anxiety
Anxiety is a disorder in which an individual experiences apprehension and fear characterized by physical symptoms such as palpitations and sweating. Anxiety is often accompanied by muscular tension, restlessness, fatigue, and problems with concentration. Symptoms of anxiety can be chronic (or generalized) or acute and may lead to panic attacks.
Symptoms can also differ in number, intensity, and frequency, depending on the person [181]. The presence of harmful microbes is known to influence anxiety and depression by triggering chronic inflammation, which occurs when these malicious microbes outcompete beneficial microbes in the gut. When these harmful gut inhabitants take over and cause inflammation, they can activate the vagus nerve, leading to neuropsychological symptoms [182].
These bacteria can also produce peptides that are known to send stress signals, which affect gene expression as well as the CNS. It has been observed that exposure of rodents to microbial pathogens during their developmental phases results in anxiety-like behaviors and impaired cognitive functions [183–185]. A study involving murine models showed an elevation in anxiety-like behavior in elevated plus maze tests 2 days after exposure to a subpathogenic infection of Campylobacter jejuni; this was noted to be significant because of the absence of an immune response in the periphery [186].
Moreover, a study by Lyte et al. involving Citrobacter rodentium and C jejuni revealed an increase in anxiety-like behavior 8 h after infection, with no difference shown in plasma cytokine levels or intestinal inflammation compared with control mice [187]. These studies suggest that the presence of harmful bacteria in the gut following the absence of a systemic immune response triggers anxiety-like behavior.
Such behaviors were also noted in experiments that resulted in increased gastrointestinal inflammation [188,189]. Mice with Trichuris muris showed gastrointestinal inflammation and increased anxiety-like behavior when they were tested using both light/dark and step-down tests. Treatment involving the probiotic B longum helped normalize the anxiety-like behavior in the infected mice.
BDNF is known to influence processes such as the survival and differentiation of neurons, formation of functional synapses, and neuroplasticity during development and adulthood [190–192]. In infection models predicted to lead to changes in microbiota profile, reduced expression of hippocampal BDNF mRNA or protein was found to be associated with increased anxiety-like behaviors [193].
Moreover, dysfunction in the signaling of GABA, which is a major inhibitory neurotransmitter in the CNS, is linked to anxiety and depression [194]. Another experiment using mouse models demonstrated the prevention of anxiety-like behavior with a high-fat diet and the administration of Lactobacillus helveticus for 21 days [195].
When the same experiment was done using IL-10-deficient mice, no changes in anxiety were observed; these results proved the role of the immune system in the gut-brain axis. In another experiment demonstrating that the microbiota can directly affect behavior, researchers performed fecal transplantation from adult germ-free BALB/c mice (a high-anxiety mouse strain) to adult germ-free NIH Swiss mice (a low-anxiety mouse strain), and the BALB/c mice received the microbiota of the NIH Swiss mice [193].
The behavioral profile of the donor was visible in the recipient mice. Although several gut bacteria are known to alleviate stress, depression, and anxiety, several studies also suggest that the gut microbiota is a contributing factor in the development of anxiety-like behaviors. Experiments involving exposure to pathogens as well as a reduction in BDNF mRNA or protein and dysfunctions of GABA signaling have revealed an increase in anxiety-like behavior. Numerous studies have also revealed possible pathways or hypotheses that suggest likely associations of the gut microbiota with the aforementioned neurological disorders (Table 2).
Current Prospects & Future Perspective
With the help of a better understanding of microbiome research as well as strategic support from various countries over the years, several organizations and institutions contributing to gut microbiome research have been established. The Lawson Health Research Institute, Bioaster Technology Research Institute, Broad Institute, and European Society of Neurogastroenterology and Motility are a few of the organizations contributing to the research. The inherent complexity and heterogeneity of the human microbiome require that an increased number of experiments be carried out to counteract the limitations of empirical methods in examining the causation or correlation links between dysbiosis and human diseases [197].
Until recent decades, the properties of the human microbiome as well as host–microbiota interactions remained unknown because of limitations in technology. The increased number of opportunities for research in this particular field was provided by the rapid development of biological research technologies, which include germ-free animal models, next-generation sequencing techniques, and multiomics approaches [198]. The development and application of these technologies aid in the analysis of gut microbiome structure and composition as well as function and its association with health and disease from different interdisciplinary perspectives.

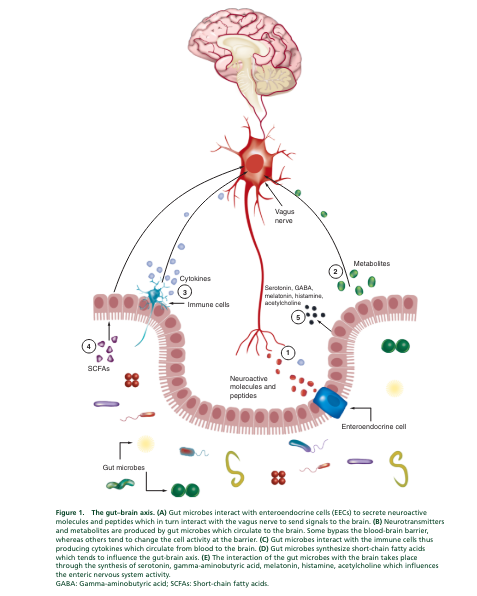
The Human Microbiome Project was a US NIH research initiative [199,200]. It was launched in 2007 to help improve the understanding of the microbial flora involved in both human health and disease.
The Metagenomics of the Human Intestinal Tract consortium, funded by the European Commission, focuses on research on the human GI tract and the relationship of the human GI tract to obesity and inflammatory bowel disease [201].
Industrialization and commercialization of relevant microbiome applications have also progressed, and this has led to the establishment of several biotechnology companies with considerable global investments. With the latest understanding of the gut microbiome as well as next-generation sequencing, identification of specific biomarkers associated with the causation of disorders can be aided.
This identification can enable us to utilize targeted therapeutic approaches/drugs and provide possibilities for early detection with great prognostic value. Drugs can aid in treatments for cancer as well as metabolic, neurodegenerative, and psychiatric disorders. The lack of effective cures and the limited availability of medications offering improvement further emphasize the need to prevent disorders with early diagnostic measures that make use of certain biomarkers.
However, the existence of multiple confounding variables in previously conducted experiments demonstrates the need for larger sample sizes in studies involving metagenomic biomarker screening.
Although animal models are used in preclinical experiments for validating the functions of certain microbial species, they also exhibit several limitations, including the fact that the human genome may share more than 85% of its genomic sequences with the mouse genome, but expression patterns, protein functions, and other factors are not the same.
Probiotics exert a beneficial effect on the health of both humans and animals by maintaining a proper balance of beneficial and harmful microbes between the GI tract and the immune system. Probiotics are also involved in the synthesis and release of antibacterial peptides and gel-forming mucin, which facilitate lubrication of epithelial surfaces and provide protection from bacteria and antigens.
These features exhibited by probiotics indicate their potential use as therapeutics in neurological disorders influenced by the gut microbiome. Moreover, the exploration and evaluation of biomarkers can further demonstrate correlating pathways that may aid in the understanding of the mechanisms underlying disorders. Gut microbiome studies are shifting their focus from correlation to causation to address the mechanisms by which the microbiome influences host health.
A better understanding of these mechanisms utilizing next-generation sequencing technologies among other advancing technologies can further aid in the development of novel diagnostic and therapeutic interventions; the production of broader and more beneficial ranges of probiotics in the future may also lead to possibilities of improved health conditions.
Conclusion
A mutualistic relationship exists between the gut microbiota and the host that aids in digestion, drug metabolism, xenobiotic metabolism, and the fight against pathogens and improves brain function. Although the gut microbiota is known to be beneficial, an imbalance in harmful and nonharmful microbes may contribute to several health problems.
An imbalance in gut microbes, which is influenced by internal and external factors, leads to dysbiosis of the microbiota, eventually leading to the development of several neurological disorders as well as weight gain, cardiovascular disease, and gastrointestinal disorders.
Delivery patterns such as cesarean and normal delivery can affect the composition of gut microbes. Several studies have demonstrated only possible hypotheses or links in terms of an imbalance in microbial composition and lack any direct association with the development of neurological disorders; thus, further research is required in each respective field.
Acknowledgments
The authors thank the members of Swift Integrity Computational Lab (Dhaka, Bangladesh), a virtual platform for young researchers, for their support during the preparation of this manuscript.

Financial & competing Interests disclosure
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in this manuscript. No writing assistance was utilized in the production of this manuscript.
Open access
This work is licensed under the Attribution-NonCommercial-NoDerivatives 4.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/

References
Papers of special note have been highlighted as • of interest; •• of considerable interest
1. Thursby E, Juge N. Introduction to the human gut microbiota. Biochem. J. 474(11), 1823–1836 (2017).
2. Donaldson G, Lee S, Mazmanian S. Gut biogeography of the bacterial microbiota. Nat. Rev. Microbiol. 14(1), 20–32 (2015).
3. Zhu S, Jiang Y, Xu K et al. The progress of gut microbiome research related to brain disorders. J. Neuroinflammation 17(1), 25 (2020). • Discusses possible hypotheses, explaining how gut microorganisms may influence the development of several neurological disorders.
4. Ghaisas S, Maher J, Kanthasamy A. Gut microbiome in health and disease: linking the microbiome-gut-brain axis and environmental factors in the pathogenesis of systemic and neurodegenerative diseases. Pharmacol. Ther. 158, 52–62 (2016). • Discusses the coordination of the microbiome-gut-brain axis and correlates it with the pathogenesis of systemic and neurodegenerative diseases.
5. Mai V, Draganov PV. Recent advances and remaining gaps in our knowledge of associations between gut microbiota and human health. World J. Gastroenterol. 15(1), 81 (2009).
6. Lozupone CA, Stombaugh JI, Gordon JI, Jansson JK, Knight R. Diversity, stability and resilience of the human gut microbiota. Nature 489(7415), 220–230 (2012).
7. Laterza L, Rizzatti G, Gaetani E, Chiusolo P, Gasbarrini A. The gut microbiota and immune system relationship in human graft-versus-host disease. Mediterr. J. Hematol. Infect. Dis. 8(1), e2016025 (2016).
For more information:1950477648nn@gmail.com






