Interaction Of Factors Determining Critical Power Part 1
Oct 10, 2023
Abstract
The physiological determinants of high-intensity exercise tolerance are important for both elite human performance and morbidity, mortality, and disease in clinical settings. The asymptote of the hyperbolic relation between external power and time to task failure, critical power, represents the threshold intensity above which systemic and intramuscular metabolic homeostasis can no longer be maintained. After 60 years of research into the phenomenon of critical power, a clear understanding of its physiological determinants has emerged. The purpose of the present review is to critically examine this contemporary evidence to explain the physiological underpinnings of critical power. Evidence demonstrating that alterations in convective and diffusive oxygen delivery can impact critical power is first addressed. Subsequently, evidence is considered that shows that rates of muscle oxygen utilization, inferred via the kinetics of pulmonary oxygen consumption, can influence critical power. The data reveal a clear picture that alterations in the rates of fux along every step of the oxygen transport and utilization pathways influence critical power. It is also clear that critical power is influenced by motor unit recruitment patterns. On this basis, it is proposed that convective and diffusive oxygen delivery act in concert with muscle oxygen utilization rates to determine the intracellular metabolic milieu and state of fatigue within the myocytes. This interacts with exercising muscle mass and motor unit recruitment patterns to ultimately determine critical power.
Cistanche can act as an anti-fatigue and stamina enhancer, and experimental studies have shown that the decoction of Cistanche tubulosa could effectively protect the liver hepatocytes and endothelial cells damaged in weight-bearing swimming mice, upregulate the expression of NOS3, and promote hepatic glycogen synthesis, thus exerting anti-fatigue efficacy. Phenylethanoid glycoside-rich Cistanche tubulosa extract could significantly reduce the serum creatine kinase, lactate dehydrogenase, and lactate levels, and increase the hemoglobin (HB) and glucose levels in ICR mice, and this could play an anti-fatigue role by decreasing the muscle damage and delaying the lactic acid enrichment for energy storage in mice. Compound Cistanche Tubulosa Tablets significantly prolonged the weight-bearing swimming time, increased the hepatic glycogen reserve, and decreased the serum urea level after exercise in mice, showing its anti-fatigue effect. The decoction of Cistanchis can improve endurance and accelerate the elimination of fatigue in exercising mice, and can also reduce the elevation of serum creatine kinase after load exercise and keep the ultrastructure of skeletal muscle of mice normal after exercise, which indicates that it has the effects of enhancing physical strength and anti-fatigue. Cistanchis also significantly prolonged the survival time of nitrite-poisoned mice and enhanced the tolerance against hypoxia and fatigue.

Click on feeling tired
【For more info:george.deng@wecistanche.com / WhatsApp:8613632399501】
Key Points
Critical power represents the threshold intensity above which steady-state metabolism is no longer attainable, and within the last 15 years, experimental data have emerged that illuminate its underpinning physiological determinants.
Here, we summarise these experimental data to demonstrate that critical power is a parameter of aerobic function that is affected by alterations in the capacities of each step in the oxygen transport and utilization pathways.
Convective/diffusive oxygen delivery and intracellular oxygen utilization rates interact with muscle fiber composition and motor unit recruitment profiles to determine the upper limit for steady-state exercise.
1 Introduction
The determinants of exercise tolerance are of clear interest because of the strong relationships between exercise capacity and athletic performance [1, 2], health in the general population, and clinical outcomes in disease populations [3, 4]. Exercise intensity is, of course, a key factor that determines the tolerability of a given task. Moreover, for individuals or groups of individuals, partitioning the exercise intensity spectrum into domains where the physiological responses to a given task share common qualitative characteristics is an effective approach that can yield insight into the physiological determinants of exercise tolerance. Accordingly, the mechanisms of fatigue and determinants of exercise intolerance are not ubiquitous across the spectrum of exercise intensities [5]. However, above a particular individual-specific power output, the consistent feature of exercise intolerance (and hence, impending task failure) is the inability of pulmonary oxygen uptake (VO2) and [lactate] (L−) to attain a steady state [6–9]. Thus, for each individual, there exists a range of intensities for which a steady state in pulmonary ̇ VO2 is attainable and a range for which it is not [6, 9–12], with the duration of sustainable exercise in the latter being significantly limited compared with the former. The threshold intensity that separates these two ranges of system behavior, and its position relative to other landmarks of aerobic function (i.e. maximal ̇VO2 [VO2max] and the lactate threshold), is, therefore, a fundamental determinant of the ability to sustain exercise [6, 13–15].

This threshold intensity can be determined by undertaking three to five high-intensity, constant-power output cycle ergometer tests to the point of task failure on separate days. The tests should be selected to last no less than 2 and no more than 15 minutes in duration [16–19], with the precise time to task failure and power output at which each test is conducted recorded. These durations are recommended for a valid determination of this intensity, as VO2 max must be attained at the end of the trial to meet the requirement for all prediction trials to be performed within the severe-intensity domain. When the time to task failure is plotted against power output, the relationship is curvilinear, with the ability to sustain exercise falling away more rapidly at higher power outputs (Fig. 1). This power-time relationship is well described by a hyperbolic function [20], with an asymptote known as critical power (CP) and the curvature constant termed W' (i.e. W prime). This relationship is described by the following equation:
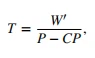

where T is the tolerable duration and P is the power output of a given exercise task [6, 20, 21]. When intensity is measured in units of speed, the asymptote is termed critical speed (CS), and the curvature constant D’ (i.e. with units of distance). This power-time relationship appears to be a universal feature of high-intensity exercise tolerance, being apparent in every species [22–26] and mode of exercise (with appropriate units of force, torque, or velocity [15, 27–30]) in which it has been studied. This relationship can also be converted to its linear equivalents, either with work plotted against time:

where W is work, CP is the slope and W′ is the intercept of the equation, or with power plotted against the inverse of time:

where CP is the intercept and W' is the slope of the equation.
Since the seminal work by Prof. David Poole and colleagues in the late 1980s, it has been repeatedly demonstrated that CP reflects the upper limit at which a metabolic steady state can be sustained. The basis for this has been the ubiquity of steady-state behavior of metabolic variables associated with the aerobic function below, but not above, CP. For example, VO2 rises to VO2max during exercise above, but not at or below, CP [6], accompanied by similarly inexorable trajectories of blood [lactate], [HCO3 −] and pH [6, 31]. Such findings were subsequently confirmed in different populations, including the elderly [32], patients with chronic heart failure [33] patients with chronic obstructive pulmonary disease (COPD) [34, 35], and healthy children [36]. More recently, non-invasive (31P-magnetic resonance spectroscopy, near-infrared spectroscopy) and invasive (i.e. muscle biopsy) studies have demonstrated the achievement of a steady state in the exercising muscle below, but not above, CP in muscle ̇ VO2, [phosphocreatine] ([PCr]), [inorganic phosphate] [Pi], pH and muscle [lactate] [15, 31, 37]; for a review, see [8, 11, 12]. Critical speed (an analog of CP) has also been demonstrated to be a critical threshold for motor unit recruitment patterns, with Copp et al. demonstrating that exercise above CS was accompanied by disproportionate increases in blood flow to type IIb/d/x fibers in the rat hind limb muscle [25].
Despite CP, and its analogs of external expression (i.e. critical speed, torque, force) being widely recognized as reflecting the threshold intensity above which a metabolic steady state cannot be sustained, its physiological antecedents have previously been obscure. Tables 1 and 2 detail interventional and observational approaches to understanding CP. Before the year 2010, intervention studies on CP were scant, and primarily confined to the effect of exercise training alongside additional measures of ̇VO2max and the gas exchange threshold/lactate threshold only, although one of the earliest studies on CP did show an independent effect of O2 availability on CP (albeit in just two participants [21]). Nevertheless, such findings supported the notion of CP as a parameter of aerobic function [20]. In contrast, since 2010, multiple experimental approaches have revealed those factors that, directly or indirectly, determine CP. The purpose of the present review is therefore to examine the physiological and biochemical underpinnings of this fundamental parameter of exercise tolerance. Particular attention will be paid to evidence generated over the last 10–12 years demonstrating that CP is a key parameter of aerobic function that can be affected by any step in the O2 transport and utilization pathway.

2 Interaction of Factors Determining CP
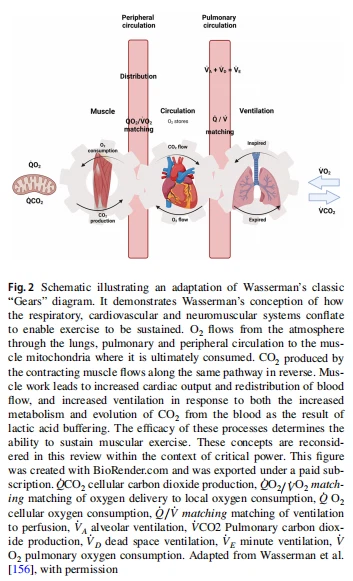
That CP represents the threshold intensity above which exercise cannot be sustained in a steady state indicates that it is a parameter of aerobic function. Consequently, it follows that CP may be affected by any step in the O2 transport and utilization cascade, from atmospheric air down to the muscle mitochondria themselves. Specifically, these steps include: (1) transport of atmospheric O2 into the blood via pulmonary diffusion; (2) bulk transport of O2 to the muscle via convection (i.e. convective O2 delivery); (3) diffusion of O2 from capillary to muscle mitochondria (i.e. diffusive O2 delivery); and (4) the utilization of O2 by the muscle mitochondria (Fig. 2). Whilst the respiratory system may constrain CP in chronic respiratory disease conditions such as COPD [34, 35, 38–42], in most young healthy individuals, the respiratory system appears to be well adapted to ensure a highly efficient and appropriate homeostatic response to high-intensity exercise [43]. Hence, the remainder of this review will focus on the impact of convective and diffusive O2 delivery and mitochondrial O2 utilization on CP, downstream of the respiratory system.
2.1 Convective Oxygen Delivery
Convective O2 delivery refers to that achieved via bulk movement of O2 within the circulation to the exercising muscles. Convective O2 delivery (QO2, L min−1) can thus be defined mathematically as the product of cardiac output (CO, L min−1) and arterial O2 content (CaO2, mL O2 100 mL−1):

where 1.34 is Hüfner’s constant describing the maximum O2 carrying capacity per gram of hemoglobin (mL O2 g−1 Hb), [Hb] is hemoglobin concentration (g dL−1), SaO2 is the arterial saturation of Hb, 0.03 is the solubility coefficient of O2 at body temperature (mL O2 100 mL−1 plasma kPa−1) and PaO2 is the arterial partial pressure of O2 (mmHg). This provides a measure of whole-body convective O2 delivery. However, the O2 fux to each portion of the exercising muscles is not uniform but varies according to regional metabolic demands, vascular control, and fiber type [44–46].
A convenient means by which to experimentally alter CaO2, and hence, convective O2 delivery, is by varying the fraction of inspired O2 (FiO2). Although hypoxia-induced vasodilatation [47] and hyperoxia-induced vasoconstriction [48] often influence blood flow thereby helping to normalize muscle O2 delivery during exercise, many studies that have quantified skeletal muscle O2 delivery under these conditions have demonstrated that hyperoxia can enhance and hypoxia can impair skeletal muscle O2 delivery during exercise, respectively [49–54], thereby impacting upon intra-myocyte PO2 (PO2im) [55]. Indeed, the early work of Moritani et al. [21] showed that, in a limited sample of two participants, the inspiration of a hypoxic gas mixture (FiO2 0.09) resulted in a reduced CP compared with normoxia (i.e. FiO2 0.21; hypoxia: 106±6 W, vs normoxia: 214±4 W). Under conditions of more moderate hypoxia (FiO2 0.15) and in a larger sample of 11 subjects, Dekerle et al. [56] found that CP was reduced by 30 W in hypoxia compared with normoxia, consequent to a reduction in arterial O2 saturation of 12%. Notably, in this latter study, the percentage decrement in CP between hypoxia and normoxia was correlated with VO2max in normoxia, suggesting that those with the greatest ̇ VO2max values were better able to offset the reductions in convective O2 delivery brought about via hypoxia. It is not known if such a protective effect remains in highly trained athletes where pulmonary limitations to high-intensity exercise are more likely [57, 58], causing reductions in arterial saturation and ̇ V O2max even at modest simulated altitudes [59]. Similarly, however, Simpson et al. [60] reported a reduction in CP of 43 W using a FiO2 of 0.13, a finding that was consistent when CP was determined either via the conventional constant-load prediction trial method or via a 3-min all-out test. Moreover, Valli et al. [61] demonstrated that at an altitude of 5050 m (equivalent FiO2~0.11), CP was reduced by 42 W. In all of these studies, SaO2 was reduced either at rest or during exercise in hypoxia, providing indirect evidence that hypoxia impaired convective O2 delivery that contributed to the reduced CP in each study. These findings were subsequently extended to arm cycle ergometry by La Monica et al. [62], who demonstrated that arm CP was reduced by 5 W in moderate (FiO2 0.14) normobaric hypoxia (~ 6% of normoxic CP). Whilst the magnitude of the effect of hypoxia on CP in these studies varied with the fitness of the participants (see, for example, Dekerle et al. [56]), Townsend et al. [63] demonstrated a progressive reduction in CP with decreasing FiO2. Hence, the extant literature is unanimously consistent with the notion that reductions in FiO2 (and by extension, convective O2 delivery) reduce CP.



The consistency of the effects of hyperoxia on CP are similar to those of hypoxia. This was first demonstrated by Vanhatalo et al. [37], who assessed the impact of a FiO2 of 0.7 on CP utilizing a single-leg knee-extension exercise model. These authors showed that CP was increased in hyperoxia compared with normoxia, with a concomitant increase in muscle oxygenation (as determined via near-infrared spectroscopy [NIRS]). The increase in CP was accompanied by a slower rate of change in muscle [PCr], [ADP], [Pi], and pH. Subsequently, these findings for small-muscle mass exercise were confirmed for large-muscle mass exercise by Goulding et al. [64, 65]. Specifically, a hyperoxic inspiration (FiO2 of 0.5) resulted in increases in end-tidal PO2 (and, therefore, alveolar PO2) and muscle oxygenation determined via NIRS both at rest and during exercise [64, 65]. As a result, CP was enhanced during cycle exercise in hyperoxia versus normoxia in both the supine [64] and upright [65] body positions, with the magnitude of improvement being 10% in both studies. Hence, studies have consistently shown that CP is sensitive to both increased [37, 64, 65] and decreased [21, 56, 60–62] FiO2.
Another experimental intervention that has yielded insights into the dependency of CP on convective O2 delivery is via manipulations in the muscle contraction duty cycle. Muscle contraction, particularly during small-muscle mass exercise where compressive forces can be high, increases intramuscular pressure, compresses blood vessels, increases impedance to flow, and may cause temporary blood flow occlusion [66–69]. Hence, the muscular contraction cycle yields rhythmic alterations in intramuscular pressure, and hence blood flow, with the majority of flow occurring during the relaxation phase of contraction, [69–72]. Utilizing a small-muscle mass handgrip exercise, Broxterman et al. [73] directly tested the hypothesis that alterations in the duty cycle would cause concomitant alterations in convective O2 delivery, and hence CP, by measuring brachial artery blood flow via Doppler ultrasound during exercise with a 20% and 50% duty cycle (i.e. muscle contraction comprised 20 and 50%, respectively, of the total contraction-relaxation cycle). Brachial artery blood flow, and thus, convective O2 delivery, was greater in the 20% duty cycle when compared with the 50% duty cycle, with a concomitant increase in CP [73].
In extending the principle of altering convective O2 delivery to observe its effect on CP, Broxterman et al. [74, 75] showed that during blood flow occlusion (which constrains O2 delivery to zero), CP was reduced to a negative value. Whilst a negative CP appears implausible, this funding demonstrates a reliance on CP on convective O2 delivery as there is no sustainable rate of oxidative metabolism without blood flow. Resting (i.e. 0 W) occlusion results in progressive depletion of [PCr] and muscle/capillary O2 stores [76, 77], a feature consistent with non-steady-state conditions [15]. Accordingly, the magnitude of the negative CP during blood flow occlusion would be expected to be proportional to the resting metabolic rate, and as such is entirely plausible.

These findings were recently extended by Hammer et al. [78] where the critical force (CF) was estimated during the final minute of repeated handgrip maximum voluntary contraction (MVC) efforts over a 5-min duration. Under free-flowing conditions without occlusion, force progressively declined with time during the test until a plateau was reached in the final minute of the test, termed CF [78]. With muscle occlusion, however, force continuously declined with time, i.e. there was no plateau in force at the end exercise [78]. Following subsequent reperfusion, the force was able to recover to a level not significantly different from CF determined under free-flowing conditions [78]. These authors also demonstrated that up to and including CF, end-exercise limb blood flow values were linearly related to the constant force requirements of each task [79, 80]. However, during exercise slightly above CF, end-exercise brachial artery blood flow demonstrated a plateau, being no different from the blood flow values obtained during exercise at CF [79]. These findings were subsequently extended to large muscle mass, and whole-body exercises by the same authors [80]. Specifically, leg blood flow and limb vascular conductance were determined using Doppler ultrasound and calibrated finger plethysmography during exercise above and below CP [80]. Post-exercise increases in limb vascular conductance and leg blood flow post-exercise were observed following supra-CP but not sub-CP exercise [80]. The data of Hammer et al. [79, 80] are in contrast to observations in the running rat [25] and from upright, incremental, large muscle mass exercise in humans [52, 81] showing increases in limb blood flow up to ̇VO2max. Nevertheless, these findings raise the intriguing possibility that in certain contexts, CF/CP represents a threshold in relative muscular force that limits skeletal muscle perfusion during exercise. Moreover, the extant literature appears to be unanimously consistent with CP being determined, at least in part, by mechanisms related to convective O2 delivery.
【For more info:george.deng@wecistanche.com / WhatsApp:8613632399501】






