Metallothionein And Cadmium Toxicology—Historical Review And Commentary Ⅱ
Dec 19, 2023
4. Cadmium Toxicity and Its Modulation by Metallothioneins
As mentioned in the introduction, clinical doctors first observed the toxic effects of Cd compounds. In 1858, Sovet reported [1] that respiratory and gastrointestinal symptoms occurred in persons using a Cd-containing polishing agent. Later publications describe toxic effects in the kidneys, liver and skeleton as well as reproductive toxicity and cancer in humans and animals (reviewed in [3]). Cd binds to MTs in all tissues (Section 2) and adverse effects in the lungs, the skeleton, the liver and kidneys as well as cancer [3,19] are likely to be modified by binding to metallothioneins. However, studies of such protective effects in other tissues than the liver and kidneys are few.

4.1. Liver Toxicity of Cadmium-Role of Metallothioneins
Piscator 1964 [5] showed that Cd was bound to MT in the liver of animals repeatedly exposed to small doses of Cd. He suggested a protective role for MT for tissue toxicity. As mentioned in Section 3.3, Cd salts given to experimental animals in a single large dose go primarily to the liver where it causes damage. Wisniewska-Knypl and Jablonska 1970 [53] and Nordberg et al., 1971 [8] showed that Cd was bound to MT in the liver of Cd-exposed animals and suggested that binding would provide protection against toxicity. After a single large dose of Cd there was liver toxicity and MT binding in liver tissue did not occur until several days after dosing [8]. When animals were pretreated with small doses of cadmium and then given a large dose, there was no liver toxicity and Cd was bound to MT [8]. Goering and Klaassen 1984 [54] showed that resistance to hepatoxicity was due to presynthesized MT. These early studies of the role of MT in protecting the liver have later been followed up and expanded with detailed biochemical and morphological investigations, reviewed by Sabolic et al., 2010 [43]. However, while liver toxicity occurs in experimental animals given comparatively large doses of Cd it is not often found in humans, because most exposures are to lower doses for longer periods of time. The following section focuses on kidney toxicity since the kidney effects have long been and still are considered critical effects in long-term Cd exposures [19].

CISTANCHE EXTRACT WITH 25% ECHINACOSIDE AND 9% ACTEOSIDE FOR KIDNEY
4.2. Kidney Toxicity of Cadmium-Role of Metallothioneins and Their Cd/Zn Content
After a single high dose of Cd, toxic effects appear in reproductive organs and the liver. One or a few days after a single dose in animals, an increased proportion of liver Cd is bound to metallothioneins, mainly MT-1 and MT-2 [8,55]. As mentioned in Section 3.3, MT-bound Cd is released from the liver into blood plasma and subsequently filtered through the glomerular membrane in the kidneys. From the glomerular filtrate, Cd-MT is taken up by the proximal renal tubules (Figure 2). In the tubular cells, CdMT enters the lysosomes [56] where Cd ions are released from MT and reach other subcellular organelles Later findings were reviewed by Sabolic 2010 [43]. Released Cd ions cause renal tubular injury if protective MT is not available to bind the released Cd. An injected bolus dose of CdMT in animals with no prior cadmium exposure will cause renal tubular damage at a concentration in the whole kidney of 9 ug/g wet wt. corresponding to a kidney cortex level of 11 ug/g as shown in 1975 [57]. Much higher tissue levels are required to induce renal tubular damage in animals with long-term exposure to cadmium, for example, in the studies by Nordberg et al., 1971 [8] and Nordberg and Piscator 1972 [58], tubular toxicity in the kidney did not appear until the concentration in the whole kidney reached 130–170 µg/g. Nordberg et al., 1975 [57], suggested an explanation for the different tissue levels related to toxicity. After a single injection of CdMT, the fast transport and uptake of CdMT in kidney tubules causes intracellular release of considerable amounts of Cd-ions, because they are released from MT when the protein is degraded. Such release causes renal tubular toxicity at relatively low Cd levels in total tissue. These levels (ca 10 µg/g tissue) would be minimum levels in the kidney cortex for tubular injury from Cd. In long-term exposures; higher levels of total tissue Cd are tolerated because there is time for local MT synthesis to take place, sequestering Cd ions. In long-term exposures, tubular kidney injury occurs at even higher tissue levels when the maximum level of local synthesis of MT is reached. Then, the sequestering action of MT will be insufficient. Nordberg et al., 1994 [59] also showed the protective effect of tissue MT in kidneys, sequestering Cd from sensitive membrane binding sites in the kidney tubules of animals injected with CdMT. Other scientists subsequently confirmed these findings [41,42] in MT null mice. In such mice Cd accumulation in the kidney is limited and because there is no MT-protection of renal tissue, renal damage occurs at low tissue levels (see also [60]).
Studies by Elinder et al. (1987) [61] of Cd-exposed animals (with intact MT synthesis) demonstrated that in MT fractions (MT-1 and MT-2) isolated from the kidneys by gelchromatography, the molar Cd/Zn quotient increases with increasing level of MT in kidney tissue (Figure 3). The MT level is proportional to the Cd tissue level and the cumulative exposure to Cd. The change in Cd/Zn quotient occurs because Cd binds stronger to SHgroups in MT than Zn. Cd thus replaces Zn in the protein. When the Cd/Zn quotient in MT is low, cells are offered protection from Cd toxicity. When the Cd/Zn quotient increases, there are fewer Zn- sites available for Cd to interact with and protection is less efficient. At 0.1 mmol MT, i.e., 0.5 mmol Cd (55 mg Cd/kg) in the kidney cortex, protection is impaired and susceptibility to tubular dysfunction increases. When the Cd/Zn quotient is 6, i.e., 6 of the 7 metal binding sites in MT are occupied by Cd, there was renal dysfunction in all animals (at 0.4–0.5 mmol MT Figure 3). MT with almost all binding sites occupied by Cd, cannot bind more Cd, and the sequestering function of MT is exhausted. The findings show how MT acts in cellular protection against Cd. These findings explain why there is protection up to a specific critical concentration in renal tubular cells. When total cellular Cd concentrations increase above this level toxic effects occur because of Cd interference with Zn-dependent enzymes and membrane functions [62,63]. These findings support the model (Figure 2) that CdMT, after uptake into renal tubular cells and transfer to lysosomes, releases Cd which interferes with cellular function [56,59,64]. Some evidence [65] suggests a role for zinc transporter ZIP8 for the expression of renal toxicity of Cd. Reactive oxygen species are formed when Cd exerts its toxic effects on renal tubular cells [66], but we do not know exactly how different events influence the outcome. However, there is evidence in humans exposed to Cd for long time periods that tubular proteinuria develops when the Cd concentration in the kidney cortex exceeds 80–200 mg/kg [3,19,67]. Although it should be possible to use information about biochemical pathways to refine quantitative risk assessments, such refined models are not yet available. The explanatory model in Figure 2 remains valid even if it does not describe the detailed molecular pathways recently discovered (see above and reviews [11,43]). In addition, Zn status is important because it probably influences the likelihood of Cd replacing Zn in MT and the likelihood of Cd interfering with sensitive molecular intracellular targets. The importance of Zn status was documented in humans [68] residing in a cadmium-contaminated area of China. At comparable Cd exposures, persons with a good zinc status had a considerably lower prevalence of renal tubular dysfunction compared to those with low serum and hair Zn.
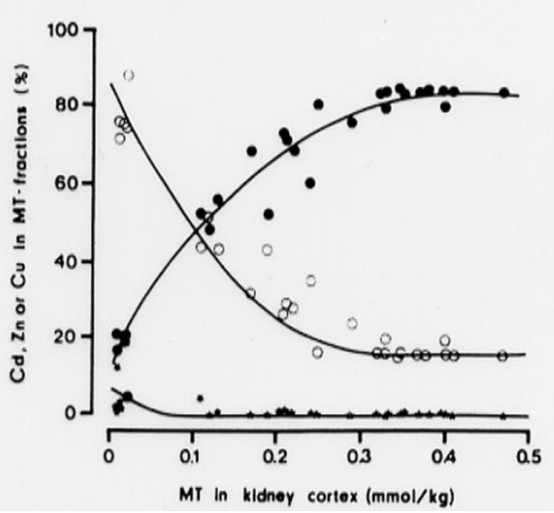
Figure 3. The relative concentrations (percent) of Cd (filled circles), Zn (open circles) and copper (black dots) in MT fractions in relation to the total MT concentration. MT was isolated from the kidneys of rabbits with varying exposure to Cd [61].
4.3. Studies in Humans on Cadmium and Metallothionein
4.3.1. Urinary Metallothionein as a Biomarker of Kidney Dysfunction
As mentioned in Section 3.4 a large proportion of Cd in urine is bound to MT [31,45,69]. Because dysfunction of kidney tubules means deficient reabsorption of all low-molecular-weight proteins (including MT) from primary urine, there will be increased excretion of these proteins in urine. The authors who showed that Cd is bound to MT in urine also showed that Cd-exposed persons with tubular dysfunction excrete more MT in urine than non-Cd-exposed persons. In an epidemiological study, Shaikh et al., 1990 [70] included 3168 men and women in a cadmium-polluted area of Japan and found increased metallothionein excretion among those with Cd-induced renal tubular dysfunction. Increased urinary MT excretion in relation to the occurrence of Cd-related renal dysfunction was also found in cadmium-exposed workers [46] and in persons with type 2 diabetes [47].
4.3.2. Metallothionein Gene Expression in Peripheral Lymphocytes-A Biomarker of Tissue Susceptibility to Cadmium Toxicity
The foregoing text reviewed evidence showing that, MT serves as an efficient intracellular scavenger for cadmium decreasing its toxicity by binding Cd in a number of tissues (reviews [60,71]). Studies in Cd-exposed animals and humans show Cd-induced MT synthesis in liver and kidneys. By in vitro Cd exposure of peripheral blood lymphocytes (PBLs), the inducibility of MT and MTmRNA was measured by RT PCR (i.e., MT gene expression (MT-GE) [72]. Lu et al. [72] performed such measurements and examined their possible use as a biomarker of general tissue detoxification by MT. They recruited Cd-exposed workers in Guangxi province, China. In addition to the measurements of MT-GE, the studies of the works included urinary Cd as a biomarker of exposure and NAG in Urine (UNAG) as effect biomarker. The results showed an increased level of urinary NAG (UNAG) in relation to increased levels of Cd in urine. Workers with high levels of MTmRNA in PBLs had lower NAG levels in urine than those with low MTmRNA levels in PBLs [72], when compared at similar levels of UCd. These results support the hypothesis that the induced level of MTmRNA in PBLs reflects MT expression level both in PBLs and in the kidney cortex. MT-GE in PBLs, thus can be used as a biomarker of tissue susceptibility to cadmium toxicity.
Another study included a group from the general population [73]. Studies of farmers in a Cd-polluted area in China included measurements of MT-GE in PBLs at comparable levels of UCd, those with high MT-GE excreted less urinary NAG than those with low MT-GE (difference statistically significant at UCd > 10µg/g Crea p < 0.001).
The mentioned studies in Cd-exposed workers and farmers show that MT-GE in PBLs can be used as a biomarker of susceptibility to Cd toxicity. However, the in vitro Cd treatment of fresh PBLs is a demanding requirement in field studies. There is a need to develop methods more suitable for field studies and these methods should be standardized in order for results to be comparable among laboratories [74]. Although occupationally exposed groups have been studied [75] large population studies are not yet available.
4.3.3. Autoantibodies against MT in Blood Plasma-A Biomarker of Susceptibility to Cadmium Nephrotoxicity
Jin et al. [76] found a high frequency of elevated levels of antibodies against MT in sera of patients with metal allergy. This finding stimulated our interest in the possibility that such antibodies might interfere with MT protection in the tissues of animals and humans. This was the general background for the following studies: Chen et al., 2006 [46] measured autoantibodies against MT (MTab) in blood plasma by ELISA. In smelter workers and controls in Hunan province, China we measured MTab in blood plasma, UCd, UNAG and UB2M (urinary beta-2-microglobulin). There were increased levels of MTab in relation to increased UNAG or UB2M. At comparable levels of UCd, there was an odds ratio of 4.2 (CI 1.2–14) for tubular dysfunction for persons with increased levels of MTab relative to those with low levels of MTab.
Animal experiments and epidemiological studies provide evidence that diabetes gives rise to an increased sensitivity to the development of Cd-related kidney dysfunction [77,78] reviewed in [3]. Chen et al. [47] performed a study on the role of MTab for the development of renal tubular dysfunction among diabetics in Shanghai, China, with type 2 diabetes. Measurements included MTab in blood plasma, UCd, UNAG, UALB (urinary albumin), UB2M and a number of background variables. UCd was 0.05–4.17, GM 0.38 ug/g crea. There were statistically significant increases of UNAG and UB2M in relation to UCd and a statistically significantly higher odds ratio for tubular dysfunction among those with high MTab versus low MTab.
In summary, the studies demonstrated that among workers and diabetics, elevated levels of MTab were associated with a higher prevalence of tubular kidney dysfunction compared to those with lower MTab levels. MTab in blood plasma thus is a biomarker of susceptibility to the development of Cd-related tubular dysfunction. We do not know the detailed mechanism behind this effect, but it is likely to reflect an interference with tissue protection by MT.

5. Concluding Remarks
Adverse health effects of Cd in humans were first reported 160 years ago and the low molecular weight Cd binding protein metallothionein (MT) was discovered more than 60 years ago. The present review summarized available evidence on the role of MTs in cadmium toxicology as applied to risk assessment. It focused on our own findings from the 1970s and onwards and gave comments on other findings in relation to relevance and applicability in risk assessment. Evidence was presented supporting the scheme (Figure 2) explaining Cd kinetics and interactions with renal targets when eliciting tubular damage in kidneys. Biomarkers like MT gene expression in peripheral blood lymphocytes and MT-antibodies in blood plasma were developed in the last twenty years (Section 4) to be used in the refinement of epidemiological studies and to assist in risk assessments. However, to our knowledge, the use of these biomarkers at present is very limited. We note that our explanatory scheme for cadmium toxicokinetics and toxicodynamics in the kidneys (Figure 2) is still generally accepted and this toxicokinetic and toxicodynamic model is successfully used for quantitative calculations of risks of renal dysfunction in relation to Cd exposure. Such calculations provide a valuable perspective on findings in epidemiological studies (Section 3.5). Opportunities to use more of the available evidence in risk assessments seems to exist, for example, to use the MT-related biomarkers and to take into account the influence of Zn status (Section 4.1). Even without such refinements of risk assessments, it is obvious that very low exposure levels of Cd give rise to adverse effects on the kidneys and other organs in humans. Partly based on the evidence summarized in the present review, it is recognized that present occupational limit values for Cd in high- and middle-income countries are often higher than desired. Actions for lower values are underway for example in the EU. In some low- and lower–middle–income countries with ongoing artisanal small-scale mining, there are excessive exposures to Cd and other metals and there is an urgent need for improved conditions. This is very important at present when there is a growing global demand for metals to combat climate change. We hope that the application of the knowledge summarized in this review will assist in improving risk assessments and conditions for population groups exposed to cadmium in various countries.
References
1. Sovet, U. Poisoning caused by powder used in the cleaning of Silver. Presse. Med. 1858, 9, 69–70. (In French)
2. Landrigan, P.; Bose-O'Reilly, S.; Elbel, J.; Nordberg, G.; Lucchini, R.; Bartram, C.; Grandjean, P.; Mergler, D.; Moyo, D.; Nemery, B.; et al. Reducing disease and death from artisanal and small-scale mining (ASM)-The urgent need for responsible mining in the context of growing global demand for minerals and metals for climate change mitigation. Environ. Health 2022, in press.
3. Nordberg, G.F.; Akesson, A.; Nogawa, K.; Nordberg, M. Chapter 7 Volume II Cadmium. In Handbook on the Toxicology of Metals, 5th ed.; Nordberg, G.F., Costa, M., Eds.; Elsevier: Amsterdam, The Netherlands; Academic Press: London, UK, 2022; pp. 141–196.
4. Margoshes, M.; Vallee, B.L. A cadmium protein from the equine kidney cortex. J. Am. Chem. Soc. 1957, 79, 4813–4814. [CrossRef]
5. Piscator, M. On cadmium in normal human kidneys together with a report on the isolation of metallothionein from livers of cadmium-exposed rabbits. Nord. Hyg. Tidskr. 1964, 45, 7
6. (In Swedish) 6. Nordberg, G.F.; Piscator, M.; Lind, B. Distribution of Cadmium among Protein Fractions of Mouse Liver. Acta Pharmacol. Toxicol. 1971, 29, 456–470. [CrossRef]
7. Nordberg, G.F.; Nordberg, M.; Piscator, M.; Vesterberg, O. Separation of two forms of rabbit metallothionein by isoelectric focusing. Biochem. J. 1972, 126, 491–498. [CrossRef]
8. Nordberg, G.F.; Piscator, M.; Nordberg, M. On the Distribution of Cadmium in Blood. Acta Pharmacol. Toxicol. 1971, 30, 289–295. [CrossRef]
9. Nordberg, M.; Nordberg, G.F. Distribution of metallothionein-bound cadmium and cadmium chloride in mice. Environ. Health Perspect. 1975, 12, 103–108. [CrossRef]
10. Kr˛e ˙zel, A.; Maret, W. The Bioinorganic Chemistry of Mammalian Metallothioneins. Chem. Rev. 2021, 121, 14594–14648. [CrossRef]
11. Thévenod, F.; Wolff, N.A. Iron transport in the kidney: Implications for physiology and cadmium nephrotoxicity. Metallomics 2016, 8, 17–42. [CrossRef]
Supportive Service Of Wecistanche-The largest cistanche exporter in the China:
Email:wallence.suen@wecistanche.com
Whatsapp/Tel:+86 15292862950
Shop For More Specifications Details:
https://www.xjcistanche.com/cistanche-shop