PART Ⅱ:Confounders For Kidney Carcinogenicity And Renal Tubule Tumor
Mar 13, 2022
Contact: Audrey Hu Whatsapp/hp: 0086 13880143964 Email: audrey.hu@wecistanche.com
Gordon C Hard
Inclusion of tumors of spontaneous origin in final tumor counts:
The aim of conducting 2-year bioassays is to determine the long-term safety of a test article, in particular, to eliminate the possibility of human exposure to agents with carcinogenic activity. In doing this it is also important to avoid the inclusion of any neoplasms of spontaneous origin in the final tumor count, which would result in an inaccurate number because tumors unrelated to chemical treatment would be included. Accordingly, attention is drawn to a phenotypically distinctive tumor in rats and a predisposition to cystic tubule formation that the author has encountered in CD-1 mice. Amphophilic-vacuolar tumor of the rat: In rats, there is a spontaneous renal tubule tumor that has been fully recognized only in the last 20 years. This neoplasm has distinctive epithelial morphology enabling its separation from kidney neoplasms that are induced by renal carcinogens. This spontaneous renal tubule tumor, named amphophilic-vacuolar (A-V) tumor, has been encountered in long-term studies conducted in the US, Europe, Great Britain, and Japan. It occurs in rats of various strains including the Fischer 344, Sprague-Dawley, and Wistar strains9,10, and affects both genders. The author first became aware of this distinctive neoplasm in 1994 in 90-day toxicity studies11, although it had also been reported by other groups12. The Thurman et al12 report was most significant because it identified that these neoplasms could occur in littermates, suggesting that this proliferation might have a genetic basis.

Cistanche is good for renal tubule tumor
The distinctive morphology includes well-developed, often large, epithelial cells with eosinophilic or amphophilic staining character, and cytoplasmic vacuoles. The vacuoles can be intracellular vacuoles or represent mini lumen formation where the perimeter of the vacuole is contributed by several neighboring cells. The nuclei are often quite large and can contain a hypertrophic nucleolus. The tumors can be adenomas or carcinomas. They appear to arise in the cortex, from foci of atypical hyperplasia. With growth, carcinomas develop as well-defined lobules of tumor cells, frequently with a central area of cell degeneration. Carcinomas protrude from the kidney surface, and their widest part is in the cortex. Typically, the tumor extends in a tapering, wedge-shaped fashion down into the outer and inner stripes of the outer medulla. Occasionally, basophilic lobules develop, but examination of all of the tumor area usually discloses the distinctive, staining, and vacuolar character in parts. There is no record that this tumor phenotype is capable of metastasizing.

benefit of cistanche: improve kidney function
The occurrence of this tumor type was studied in detail in the archived renal tissue held in the NTP Archives9. This Archive had 150 long-term studies in which renal tubule tumors had been recorded, representing a pool of some 90,000 rats, predominantly Fischer 344 strain. Of these rats, 1012 had been diagnosed as having a renal tubule tumor. Histological re-examination of each of these tumors showed that half of the studies with this diagnosis (74) had at least one tumor with the A-V phenotype. So the neoplasm is uncommon at approximately 0.1% incidence in 2-year studies but is encountered relatively frequently in approximately 50% of long-term studies diagnosed with renal tubule tumors. None of the lesions in this survey of 74 studies had metastasized. Most studies with A-V tumors had a single neoplasm of this type, but the number of A-V tumors in a study varied from 1 to 5. This distribution again suggested that a single litter in the study may have been carrying a genetic defect. Statistical analysis of the data from this histological review showed that the distinctive tumor type was spontaneous and had no association with chemical exposure 9 Therefore, tumors of this distinctive phenotype should not be included in kidney tumor counts in cancer bioassays, but should be recorded separately.
The single carcinoma in the high-dose males of the NTP quercetin study was unquestionable of the A-V phenotype (Figure 5) and therefore of spontaneous origin. In summarizing the numbers of quercetin-related neoplastic lesions, excluding the CPN proliferative foci, and the A-V lesions, there was a modest increase in foci of preneoplasia and adenomas, but no carcinomas in high-dose male rats. That modest increase in foci of atypical tubule hyperplasia and adenomas was associated with CPN exacerbation.
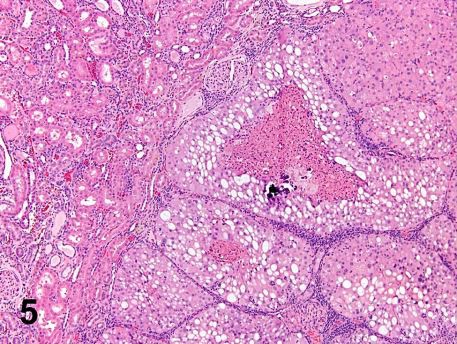
Fig. 5. A typical cross-section of the carcinoma in the high-dose males of the quercetin study. The tumor is characterized by well-defined lobules of amphophilic and vacuolated cells with central degeneration. It is unquestionably an A-V tumor that should be excluded from the test article-induced tumor count. (Courtesy of, and adapted from, Hard et al, 2007, Food Chem Toxicol 45, 600-608).
Cystic tubules in the CD-1 mouse: On several occasions, this author has encountered a predisposition in the CD-1 mouse for developing solitary or sporadic cystic renal tubules in the cortex (Hard GC, unpublished observations). This situation appears to lead to the development of renal tubule adenomas and banal carcinomas. The tumors are faintly basophilic and initially located in the cortex, where the sporadic cystic tubules are located. In one study, there were intermediate stages of hyperplasia and early adenoma formation in cystic tubules, providing a definite link between cystic tubule and renal tubule neoplasia. However, the incidence of cystic tubules did not correlate with exposure to the test article but occurred spontaneously in all groups, including control mice. The tumors were most prevalent in the high-dose male mice, but this was a very high dose that exceeded regulatory authority guidelines for high dose selection. Such a high exposure would very probably result in saturation of metabolic pathways, resulting in very different metabolic profiles from those occurring under realistic conditions of exposure, thus predisposing to biologic stress13,14,15. In addition, the test article did not induce any pathologic indication of renal tubule cell injury at any time point, including exposures at 4 weeks, 52 weeks, and 104 weeks. There was also no increased mitotic response in renal tubules, which would have occurred had there been any chemically-induced compensatory activity to replace chemically-damaged tubule epithelium. It was concluded that the renal tubule neoplasms in this mouse study were due to the spontaneous development of cystic tubules with abnormal cell lining, probably as a result of a genetic aberration. This case represents another example of spontaneous development of renal tubule tumors that should not be attributed to a test article response.
cistanche extract improve kidney function
Karyomegaly: A markedly enlarged nucleus in a renal tubule cell (Figure 6), known as karyomegaly, has long been viewed as an adverse finding that might predict the development of renal tubule tumors and identify the inducing chemicals as potential renal carcinogens16, 17, 18, 19, 20. A literature review of karyomegaly in laboratory animals and humans was conducted in 2018 by the author21. At least 50 chemicals/substances have been reported to induce this nuclear alteration in rats, but it is a much less common occurrence in mice and other laboratory animals21. A number of chemicals that induce this change in rats do not produce the same effect in other species. Of particular potency in the rat is the food component lysinoalanine, and mycotoxin, ochratoxin A. Both of these compounds have been tested in other laboratory animal species, with negative results. Feeding an excessively high dose of lysinoalanine, 10,000 ppm to Swiss mice, was required to elicit a minimal response of karyomegaly in this species compared to 50 ppm in the rat 22. A review of the literature indicates that the rat has a predisposition for developing karyomegaly as a response to chemical toxicity21. This literature review also demonstrated that karyomegaly in the rat kidney is not a reliable predictor of renal tumor development.

Fig. 6. Renal tubule cell karyomegaly is an abnormally enlarged tubule cell nucleus. It is recommended that this diagnosis be reserved for nuclei of octoploid or higher. (Courtesy of, and adapted from, Hard et al, 2000, Toxicol Sci 53: 237-244).
The modest increase in renal tubule nuclear size is not limited to chemical exposure but also occurs in some physiological conditions. It has been known for many decades that, in general, nuclear volume doubles with each increase in ploidy level 23. During the cell cycle, there is a change in ploidy from 2n to 4n in the DNA synthesis (S) phase, persisting into the G2 phase. There is also an increase in nuclear size (up to doubling) in pathological conditions such as temporary renal ischemia 24, and following unilateral nephrectomy25. Therefore, the threshold for diagnosing renal tubule karyomegaly needs to be set at a level of ploidy that discriminates the abnormal from the normal. It is suggested that this threshold be at least 4x normal tubule nucleus size (octoploid) for diagnosing renal tubule karyomegaly.
Karyomegaly is rare in human kidneys and the occasional cases observed in renal biopsy or autopsy tissue are from patients with a genetic condition termed karyomegalic interstitial nephritis (KIN). This condition was shown by Zhou and 43 co-authors26 to be caused by an autosomal recessive mutation of the FAN1 gene, the protein which functions in the repair of DNA interstitial crosslinks within the Fanconi anemia DNA damage response pathway. Karyomegaly in the human kidney has also been observed occasionally in HIV patients that have received the antiviral drug, tenofovir, but this association appears to be inconsistent27. Although the rat is uniquely predisposed to responding to chemically-induced toxic injury with renal tubule karyomegaly, renal tubule karyomegaly in the rat is not consistently associated with the development of renal tubule tumors or their precursors21. Therefore karyomegaly in the rat kidney is considered to be an inaccurate predictor of the renal carcinogenic potential of chemicals.

cistanche flavonoids anti-tumor and anti-cancer
References
10. Thurman JD, Hailey JR, Turturro A, and Gaylor DW. Spontaneous renal tubule carcinoma in Fischer 344 rat littermates. Vet Pathol. 32: 419-422. 1995.
11. Counts JL, and Goodman JI. Principles underlying dose selection, and extrapolation from, the carcinogen bioassay: dose influences mechanism. Regul Toxicol Pharmacol. 21: 418-421. 1995.
12. Barton HA, Pastoor TP, Baetke K, Chambers JE, Diliberto J, Doerrer NG, Driver JH, Hastings CE, Iyengar S, Krieger R, Stahl B, and Timchalk C. The acquisition and application of absorption, distribution, metabolism, and excretion (ADME) data in agricultural chemical safety assessments. Crit rev Toxicol. 36: 9-35. 2006.
13. Bus JS. “The dose makes the poison”: key implications for the mode of action (mechanistic) research in a 21st toxicology paradigm. Current Opin Toxicol. 3: 87-91. 2017.
14. Scheuer PJ. Histochemical changes in rat liver in Senecio and thioacetamide poisoning. J Pathol Bacteriol. 85: 507-516. 1963.
15. Payne BJ and Saunders LZ. Heavy metal nephropathy of rodents. Vet Pathol. Suppl 15: 51-87. 1978.
16. Dees JH and Kramer RA. Sequential morphologic analysis of the nephrotoxicity produced in rats by single doses of chlorozotocin. Toxicol Pathol. 14: 213-230. 1986.
17. Luitjen M, Speksnijder EN, van Alphen N, and 6 others. Phenacetin acts as a weak genotoxic compound preferentially in the kidney of DNA repair-deficient Xpa mice. Mutat Res. 596: 143-150. 2006.
18. NTP. National Toxicology Program Nonneoplastic Lesion Atlas. Research Triangle Park, NC. National Toxicology Program, NIEHS, NIH. 2015.
19. Hard GC. A critical review of renal tubule karyomegaly in non-clinical safety evaluation studies and its significance for human risk assessment. Crit Rev Toxicol. 48: 575-595. 2018.
20. Feron VJ, van Beek l Slump P, and Beems RB. Toxicological aspects of alkali treatment of food proteins, In: Biochemical Aspects of New Protein Food. Symposium 3. 1978.
21. Rather LJ. The significance of nuclear size in physiological and pathological processes. Ergeb Allg Pathol Pathol Anat. 38: 127-198.1958.
22. Cain H, Fazekas S, and Ross W. Studien über die Folgen einer vorübergerhenden experimentellen Nierenischämie III. Karyometrie und Häufigkeitsanalyse des “bunten Kemmusters” während der Regeneration. Virchows Arch Path Anat. 337: 53-64. 1963.
23. Schmeidt E. Zellkerngrösse und sogenannte kompensartorische Hypertrophie der Mäuseniere. Zeits Micr Anat Forsch. 57: 249-275. 1951.
24. Zhou W, Otto EA, Cluckey A, and 41 others. Fan1 mutations cause karyomegalic interstitial nephritis, linking chronic kidney failure to defective DNA damage repair. Nature Genet. 44: 910-915. 2012.
25. Karras A, Lafaurie M, Furco A, Bourgarit A, Droz D, Sereni D, Legendre C, Martinez F, and Molina JM. Tenofovir-related nephrotoxicity in human immunodeficiency virus-infected patients: three cases of renal failure, Fanconi syndrome, and nephrogenic diabetes insipidus. Clin Infect Dis. 36: 1070-1073. 2003.








