Part Two Helicobacter Pylori Eradication in Renal Transplant Candidates
Jun 20, 2023
Results
In total, 83 patients with a median age of 47 years (IQR: 38 - 56) were analyzed. The main underlying diseases were systemic arterial hypertension and diabetes mellitus, while the main dialysis type was hemodialysis. The median duration of dialysis before the upper gastrointestinal endoscopy was 14 months (IQR: 6 - 48). Table 1 presents the demographic and clinical characteristics of pre-transplantation patients.

The study showed a 61.4% prevalence of H. pylori. Histology demonstrated 98% positivity as a diagnostic method for H. pylori and the rapid urease test, 60.8%. The rapid urease test detected H. pylori separately in only one patient. Of the 51 patients who tested positive for H. pylori, 18 were lost to follow-up. Thus, 33 patients were part of the treatment protocol. The infection eradication rate was 48.5% (Figure 1).
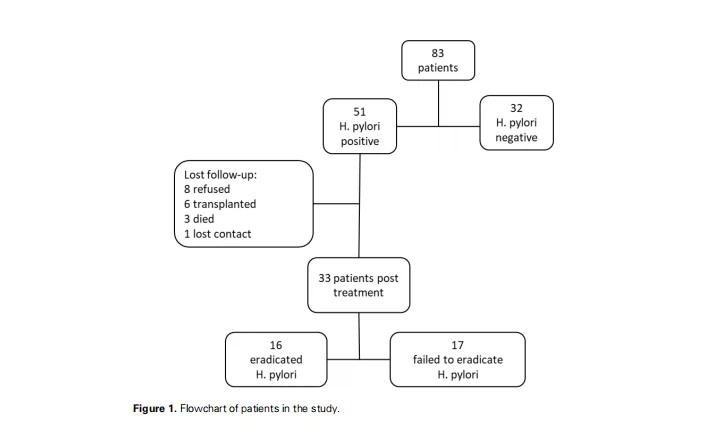
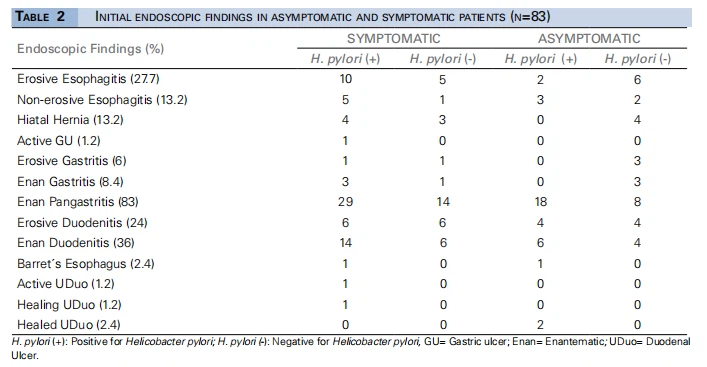
Findings were positive in 96.4% of endoscopies. The most commonly found lesion was exanthematous pangastritis. Few patients had ulcers. Only one patient presented a precancerous lesion, which was Barrett’s esophagus. No malignant lesions were detected. There were no associations between the endoscopic findings, symptomatology, and the presence of H. pylori based on Fisher’s exact test (Table 2).
Gastrointestinal symptoms were reported by 61.4% of patients. Epigastric pain did not occur in patients with endoscopic findings of ulcer. Nausea and pyrosis were frequent symptoms. There was a tendency for vomiting to be more associated with exanthematous pangastritis (Table 3).

After 7 days of follow-up by telephone contact, all patients presented good treatment compliance. Complete compliance was confirmed when patients returned to a nephrologist after 14 days of treatment; treatment did not include supervised pill-taking.

Click here to get the effect of Cistanche
Discussion
A high prevalence of H. pylori was found in renal transplant candidates. The association of diagnostic methods was disadvantageous to the detection of H. pylori by upper gastrointestinal endoscopy. The eradication rate after using the triple scheme for 14 days was low in the included patients.
The eradication rate of H. pylori in the general population after treatment with the first-line triple scheme (proton pump inhibitor, amoxicillin, and clarithromycin) has decreased in recent years, especially after the use of the shorter scheme (7 days)15. It was 93.5% in 2003 and by 2012, it had reduced to 78.8%15. Some meta-analyses show higher eradication rates with a longer treatment of 14 days16,17,18,19. Recent literature reviews demonstrated that the increase in triple therapy duration increased the H. pylori eradication rate (72.9 vs. 81.9%), independently of the type and dosage of antimicrobials17.
The eradication rate of H. pylori in chronic kidney disease patients varies according to the antimicrobial schemes used, whether triple, quadruple, or sequential. In the Seyyedand and collaborators study, the triple scheme with 30 mg lansoprazole twice a day, 250 mg clarithromycin twice a day, and 500 mg amoxicillin twice a day for 14 days, presented a 76.7% eradication rate for H. pylori20. Another double-blind prospective clinical study compared two groups of chronic kidney disease patients. One group of 35 patients received the full dosage of the scheme with 20 mg omeprazole, 500 mg clarithromycin, and 1000 mg amoxicillin twice a day for 14 days, while the other group of 31 patients received the same drugs throughout the same period, but once a day. The eradication rate of both groups was 73% with no statistical difference between the two regimes (p= 0.973)21.
Although the extended time frame of the triple treatment was preferred by our patients, we found a lower eradication rate. This could have been due to poor compliance with the prolonged treatment or an unknown resistance to one of the medications used especially clarithromycin. Resistance to clarithromycin is still the most common cause of triple therapy failure, and the period of treatment does not affect the high rates of clarithromycin resistance in the general population13. In a previous study that evaluated the 5 macro-regions of Brazil, bacterial resistance to clarithromycin varied from 15 to 20%22. The current study could not assess resistance to clarithromycin, since neither culture nor an antibiogram was conducted13.
The influence of pre-therapeutic histological parameters on the H. pylori eradication rate is still controversial. Georgopoulos and collaborators suggested that the coexistence of high scores of antral gastritis degree and activity with any degree of corpus gastritis may favorably affect the outcome of treatment, supporting the idea of facilitated diffusion of antibiotics in the inflamed mucosa23. However, in the current work, we found a lower eradication rate, although the most prevalent endoscopic finding was pangastritis and enema.
H. pylori is the most important etiological factor for gastric cancer. H. pylori causes chronic gastric inflammation that can progress to precancerous alterations in atrophic gastritis and in intestinal metaplasia. The risk of gastric cancer increases according to the extension and severity of these precancerous alterations. Eradication of H. pylori can induce resolution of gastric inflammation, stop the progression of gastric mucosa damage, prevent additional damage to the DNA induced by H. pylori, improve gastric acid secretion, and restore normality of the internal environment8. Thus, we believe that treatment with an antimicrobial regime should be considered in patients with H. pylori and chronic kidney disease.

Cistanche pills
Patients with chronic kidney disease present a greater risk of gastroduodenal disorders. It is recommended that all hemodialysis and peritoneal dialysis patients be submitted to endoscopic evaluation to reduce the chances of developing peptic ulcers, especially in patients with a history of gastroduodenal bleeding or use of anticoagulants and/or non-steroidal anti-inflammatory drugs24. We used two diagnostic methods associated with upper gastrointestinal endoscopy to increase the probability of detection of H. pylori. The literature demonstrates high sensitivity and specificity of the rapid urease test, varying between 80 and 100% and 97 and 99%, respectively16,25. However, in the current study, H. pylori detection by the rapid urease test was low. A disadvantage of the test that could explain this situation is false-negative results due to the reduction in the activity of urease caused by the recent use of antimicrobials, bismuth compounds, or PBIs, or because of achlorhydria. In addition, the presence of gastric bleeding from uremia reduces the sensitivity and specificity of the method. Furthermore, the amount of bacteria present affects the sensitivity of the test; quantities above 10,000 in the sample indicate positive results, while quantities below this can generate false negatives. The rapid urease test should be performed with a biopsy of one gastric region (antrum), differently from the histology from a biopsy of two gastric regions (body and antrum). For now, we suggest that the association of both methods in our patients increased the evaluation costs and did not lead to an increase in the detection rate.
Patients with chronic kidney disease have a high incidence of gastrointestinal diseases, even though the occurrence and type of symptoms can vary considerably between patients11. Many gastrointestinal symptoms such as anorexia, nausea, vomiting, and dyspepsia are common in patients with chronic kidney disease waiting for a renal transplant. These symptoms could be indicative of uremia or the result of medications and electrolyte imbalance, which makes it difficult to confidently predict the presence of a meaningful lesion in the superior gastrointestinal tract. However, many chronic kidney disease patients with peptic ulcers are asymptomatic26,27 and can present significant complications before and after renal transplant, especially during the period of high immunosuppression28. This finding is very relevant, as an active peptic ulcer is a contraindication for renal transplant29 and strongly supports the recommendation for upper gastrointestinal endoscopies before transplantation. There is no solid evidence on the role of routine triage with upper gastrointestinal endoscopy for H. pylori in asymptomatic candidates during the evaluation before renal transplantation and no consensus between transplant centers30. In a recent study performed in the same renal transplant center, Homse and collaborators showed that lesions in the gastrointestinal tract were found in all analyzed patients, even though patients did not present symptoms6. It is believed that the findings of the study justify the recommendation for upper gastrointestinal endoscopy in the preparation routine for renal transplantation in chronic kidney disease patients.

Cistanche tubulosa and Standardized Cistanche
This is one of the first studies to evaluate findings from upper gastrointestinal endoscopies for pre-transplant preparation and the efficiency of H. pylori treatment in renal transplant candidates. The verification of drug compliance by phone contact 7 days after the beginning of treatment was also considered a strength of the study. Complete compliance was confirmed when patients were seen by the nephrologist after the end of treatment, but supervised pill-taking was not performed. An association was found between H. pylori infection and upper gastrointestinal endoscopy findings in this population. Some limitations of the study should be considered, such as the low number of patients analyzed and the losses to follow-up, which were partly due to the difficulties faced by patients to access transplant centers to repeat the upper gastrointestinal endoscopy. Therefore, our results should be interpreted with caution. Another limitation of the study was that no upper digestive endoscopy was performed after kidney transplantation. This was not included in the original study, as the coverage of its performance in asymptomatic patients is limited in the National Health System. Another limitation was that two of the drugs used against H. pylori are filtered during hemodialysis (amoxicillin and clarithromycin). To minimize this effect, patients were instructed to take these medications after hemodialysis.
There is no clinical protocol for a detailed endoscopic evaluation and eradication therapy for H. pylori in patients on dialysis. Therefore, future studies must be developed to confirm and expand the findings of the current study24,31.
Conclusion
A high prevalence of H. pylori was found in renal transplant candidates, and triple antimicrobial therapy applied over a long period had a low eradication rate. The performance of routine upper gastrointestinal endoscopy in pre-transplant evaluation detected gastrointestinal lesions in most patients and the endoscopic findings did not relate to symptoms. The association of the rapid urease test with gastric mucosa histology did not increase the detection rate of H. pylori.
Cistanche capsules
References
14. Malfertheiner P, Megraud F, O’Morain C, Bazzoli F, El-Omar E, Graham D, et al. Current concepts in the management of Helicobacter pylori infection: the Maastricht III Consensus Report. Gut. 2007 Jun;56(6):772-8.
15. Kim SE, Park MI, Park SJ, Moon W, Choi YJ, Cheon JH, et al. Trends in H. pylori eradication rates by first-line triple therapy and related factors in eradication therapy. Korean J Intern Med. 2015 Nov;30(6):801-7.
16. Calvet X, Sánchez-Delgado J, Montserrat A, Lario S, RamírezLázaro MJ, Quesada M, et al. Accuracy of diagnostic tests for Helicobacter pylori: a reappraisal. Clin Infect Dis. 2009 May;48(10):1385-91.
17. Yuan Y, Ford AC, Khan KJ, Gisbert JP, Forman D, Leontiadis GI, et al. Optimum duration of regimens for H. pylori eradication. Cochrane Database Syst Rev. 2013 Dec;(12): CD008337.
18. Karatapanis S, Georgopoulos SD, Papastergiou V, Skorda L, Papantoniou N, Lisgos P, et al. “7, 10 and 14-days rabeprazole based standard triple therapies for H. pylori eradication: are they still effective? A randomized trial”. Acta Gastroenterol Belg. 2011;74:407-12.
19. Puig I, Baylina M, Sánchez-Delgado J, López-Gongora S, Suarez D, García-Iglesias P, et al. Systematic review and meta-analysis: triple therapy combining a proton-pump inhibitor, amoxicillin, and metronidazole for H. pylori first-line treatment. J Antimicrob Chemother. 2016 Oct;71(10):2740-53.
20. Majidi MRS, Pirayyatlou PS, Rajabikashani M, Firoozabadi M, Majidi SS, Vafaeimanesh J. Comparison of Helicobacter pylori eradication regimens in patients with end-stage renal disease. Gastroenterol Hepatol Bed Bench. 2018;11(1):15-9.
21. Ardakani MJE, Aghajanian M, Nasiri AA, MohagheghShalmani H, Zojaji H, Maleki I. Comparison of half-dose and full-dose triple therapy regimens for Helicobacter pylori eradication in patients with end-stage renal disease. Gastroenterol Hepatol Bed Bench. 2014;7(3):151-5.
22. Sanches BS, Martins GM, Lima K, Cota B, Moretzsohn LD, Ribeiro LT, et al. Detection of helicobacter pylori resistance to clarithromycin and fluoroquinolones in Brazil: a national survey observational study. World J Gastroenterol. 2016 Sep;22(33):7587-94.
23. Georgopoulos SD, Ladas SD, Karatapanis S, Mentis A, Spiliadi C, Artikis V, et al. Factors that may affect treatment outcome of triple helicobacter pylori eradication therapy with omeprazole, amoxicillin, and clarithromycin. Dig Dis Sci. 2000 Jan;45(1):63-7.
24. Sugimoto M, Hideo Y, Andoh A. Nutrition status and Helicobacter pylori infection in patients receiving hemodialysis. World J Gastroenterol. 2018 Apr;24(15):1591-600.
25. Vaira D, Perna F. How useful is the rapid urease test for evaluating the success of Helicobacter pylori eradication therapy? Nat Clin Pract Gastroenterol Hepatol. 2007 Nov;4(11):600-1.
26. Al-Mueilo SH. Gastroduodenal lesions and Helicobacter pylori infection in hemodialysis patients. Saudi Med J. 2004 Aug;25(8):1010-4.
27. Milito G, Taccone-Gallucci M, Brancaleone C, Nardi F, Filingeri V, Cesca D, et al. Assessment of the upper gastrointestinal tract in hemodialysis patients awaiting renal transplantation. Am J Gastroenterol. 1983 Jun;78(6):328-31.
28. Ardalan MR, Etemadi J, Somi MH, Ghafari A, Ghojazadeh M. Upper gastrointestinal bleeding during the first month after renal transplantation in the mycophenolate mofetil era. Transplant Proc. 2009;41:2845-7.
29. Dianne B, McKay EL, Milford NE, Tolkoff R. Management of the patient with renal failure. 6th ed. Philadelphia: Saunders; 2000.
30. Bunchorntavakul C, Atsawarungruangkit A. Prevalence of asymptomatic gastroduodenal lesions and Helicobacter pylori infection in kidney transplant candidates. J Med Assoc Thai. 2014 Nov;97(Suppl 11):S62-S68.
31. Sugimoto M, Yamaska Y. Review of Helicobacter pylori infection and chronic renal failure. Ther Apher Dial. 2011 Feb;15(1):1-9.
Mariana E. Maioli1 Raquel F. N. Frange2 Cintia M. C. Grion2 Vinicius D. A. Delfino 2
1 Universidade Estadual de Londrina, Departamento de Clínica Médica, Londrina, PR, Brasil.
2 Pontifícia Universidade Católica, Departamento de Clínica Médica, Londrina, PR, Brasil.






