Alprostadil Combined With Tripterygium Glycosides Tablet in Treatment Of Diabetic Kidney Disease: A Systematic Review And Meta-analysis
Mar 11, 2022
Contact: joanna.jia@wecistanche.com / WhatsApp: 008618081934791
Hui-Chuan Tian, Jia-Jun Ren, Yi-Chun Shang
1 The First Affiliated Hospital of Tianjin University of Traditional Chinese Medicine, Tianjin 301617, China;
2Tianjin University of Traditional Chinese Medicine, Tianjin 301617, China.
Background: Diabetic kidney disease is now the principal cause of end-stage renal failure worldwide. Alprostadil combined with tripterygium glycosides tablet is a new method for diabetic kidney disease treatment. However, there are currently few systematic reviews on treatment for alprostadil combined with tripterygium glycosides tablet. Therefore, a systematic review and meta-analysis were conducted to analyze the function of alprostadil combined with tripterygium glycosides tablets in the treatment of diabetic kidney disease. Methods: We searched Pubmed, Embase, the Cochrane Library, Chinese databases, and clinical trials for randomized controlled trials of alprostadil combined with tripterygium glycosides tablet in the treatment of diabetic kidney disease, including results from the foundation of the database until August 5. 2020. Two reviewers haye independently performed Literature screening, data extraction, and quality evaluation, This meta-analysis has been carried out by ReyMan5.4 software. Results: Ten randomized controlled trials with 724 patients were involved. Compared with alprostadil alone, the combination of alprostadil and tripterygium glycosides tablet in treatment of diabetic kidney disease could reduce the level of24-hour urine protein(95%CI(-2.05,-0.22),P=0,01),serum creatinine (95%CI(-5.01. -0.20), P= 0.03), level of interleukin-6(95% CI(-4.57,-2.37), P<0.00001), tumor necrosis factor-α(95% CI (-4.57,-2.37), P<0.00001).The combined treatment could also improve the clinical efficacy(95% CI(1.09.1.25)P<0.0001), and reduce the occurrence of serious adverse events(95%CI (0.26,0.94), P=0.03). However, there is no association of two treatments in blood urea nitrogen (95% CI(-4.17.2.11).P=0.52), albumin (95%CI(-1.10. 0 97)P = 0 90)triglyceride (95% CI(-1 441 50)P = 097)Conclusion: Alprostadil combined with tripterygium glycosides tablet contributes to protecting renal function, inhibiting inflammation, and reducing the occurrence of adverse events, which could be considered as a feasible therapy for diabetic kidney disease patients. However, some clinical variables did not accurately conclude due to the low quality of methodology and small sample sizes. More rigorous and more extensive trials are essential to validate our results.Trial registration:Systematic review registration: (CRD42020203725).
Keywords: Alprostadil, Tripterygium glycosides, Diabetic kidney disease, Meta-analysis, Systematic review

cistanche can treat acute renal failure
Background
Diabetic kidney disease(DKD) is one of the most common microvascular complication of diabetes mellitus. which was mainly manifested as microalbuminuria(i.e.,30-300 mg/g creatinine in a spot urine sample)or macroalbuminuria(ie..> 300 mg/g creatinine in a spot urine sample).In recent years some established risk factors like the prevalence of high sugar diet, smoking [1], and population aging [2]were valued by the researchers.
At present, the pathogenesis of DKD remains unclear, mainly including metabolic disorders, renal hemodynamic changes, and genetic susceptibility. With the in-depth study of the inflammatory response, oxidative stress, podocyte injury, and other mechanisms, understanding DKD in the treatment has entered a new level. However, up to now, there is a lack of effective treatment in the medical community. Approximately 50% of DKD patients worldwide eventually reached end-stage renal disease without effective treatment [3].
DKD treatment aims to reduce the progress of kidney damage and control the related complications 4]. According to Mogensen stage, DKD can be divided into 5 stages, and in clinical treatment, most patients were diagnosed as stage 3 or after stage 3 because the symptoms of DKD were inconspicuous when it was in stages 1 and 2[5]. In this way, to treat the DKD, novel treatment options have arisen, which include intensive glycemic control, optimal renin-angiotensin-aldosterone system blockade with angiotensin-converting enzyme inhibitors/angiotensin receptor blockers), precise blood pressure control, and lifestyle modifications such as exercise and dietary restrictions [6,7]. However, although novel treatment options have some factions, it is not always adequate to alleviate proteinuria by using these treatment options. Therefore, it is meaningful to find a treatment to delay the development of DKD.
Multi-glycoside of Tripterygium wilfordii Hook. f. in tripterygium glycosides tablet (TGT) is a stable glycoside extracted from Tripterygium Wilford Hook. f., which is known as traditional Chinese medicine used in the treatment of chronic kidney disease for many years [8, 9]. In recent years, studies showed that TGT could inhibit inflammatory cell-mediated response in kidney tissues and the proliferation of mesangial cells and matrix [10,11]. Alprostadil(PGE1, prostaglandin E1), was used as a cardiovascular drug in the past. In recent years, Luo and Hong[12,13]found that PGE1 may improve renal function and delay progressive renal damage in DKD patients, by decreasing the levels of interleukin-18 and serum inflammatory factors. Hence, based on the current experimental data, TGT has been increasingly used in the treatment of DKD, and TGT combined with PGE1 represents a novel and effective treatment of DK patients with fewer side effects.
However, there is still a lack of sufficient and satisfactory systematic evidence-based medical evidence in the efficacy and safety of TGT in treating DKD patients. Therefore, we have conducted a meta-analysis to evaluate the efficacy and safety of PGE1 combined with TGT to treat DKD.

Cistanche can treat diabetes
Methods
Protocol and registration
This study protocol was registered in the PROSPERO database before starting the review process (CRD42020203725).
Literature search
Electronic data searches. We searched 4 electronic databases in the English version and 3 electronic databases in the Chinese version: PubMed, Embase, the Cochrane Library, Clinical Trial, China Network Knowledge Infrastructure, Chinese Scientific Journal Database. Wan Fang Database. All the databases were searched from the date of inception until August 6, 2020. Applicable languages included English and Chinese. The retrieval scheme contained subject words and free words, keywords, or full text to avoid omission. We searched Chinese words such as"Leigongteng", "Qianliedier", "PGE1", "Tangniaobingshenbing",and English words such as "alprostadil","PGE1","pterygium","tripterygium glycosides","diabetic nephropathy,""diabetic kidney disease,""diabetic renal damage,""DN,"and"DKD"Manual searches. Retrievals were conducted in Tianjin University of Chinese Medicine library collection (2017-2020): Journal of Tiamin University of Traditional Chinese Medicine, Tianjin Journal of Traditional Chinese Medicine, Chinese Journal of Basic Medicine in Traditional Chinese Medicine, and the part of related clinical trials were listed as a supplement.
Inclusion and exclusion criteria
Types of studies. Randomized controlled trials(RCTs), regardless of the blinding methods, reported in either English or Chinese were included; non-RCTs, including animal experiments, experience summary, systematic review, case report, and self-controlled trials were excluded. The definitions of the outcome measures were described in Table 1.

Table 1 Characteristic of PGE1 combined TGT for DKD treatment
Types of participants. Inclusion criteria: patients were diagnosed with DKD based on the World Health Organization diagnostic standard and Mogensen staging criteria, irrespective of other diseases' cause or presence.
Types of interventions. The treatment group was administered PGEl injection(10 ug·gd) and TGT (20 mg:tid) combined with conventional treatment The course of treatment is between 4 weeks to 16 weeks. Types of control groups. The control group was administered PGE1 injection(10 ug-QD)combined with conventional treatment like hypoglycemic drugs and a healthy diet to control blood sugar.
Types of outcome measures. Primary outcome measures include efficacy rate, 24 h urine protein (24 h-Upro), serum creatinine(Scr), blood urea nitrogen (BUN), the albumin (ALB) comparison of interleukin-6(IL-6), tumor necrosis factor-α(TNF-a), and the triglyceride (TG). The secondary outcome measure is the adverse events rate.
Data extraction
Two reviewers (H.T and J.R) independently performed the following information in each study: author’s name,
year of publication, size of the sample (treatment group, control group), the typical course of the disease, interventions (treatment group, control group), outcome and adverse events. Discrepancies were resolved in a consensus meeting or, if the agreement could not be reached, they were resolved by referral to a third reviewer (Y.S). The independent reviewers extracted and tabulated data using a standardized data extraction form, with disagreements finally interpreted by the corresponding author (Y.S).
Quality assessment
Two researchers(H.T and J.R) independently evaluated the method—Cochrane Collaboration Risk of methodological quality of eligible trials with the bias tool (version 5.1.0)to analyze the included RCTs. Each term was divided into 3 grades—high risk, low risk, and unclear risk based on seven aspects: 1)random sequence generation; 2) allocation concealment; 3)blinding of participants and personnel;4)blinding of outcome assessment;5)incomplete outcome data; 6)selective reporting; 7)other bias. The risk of bias graphs was generated by RevMan 5.4 software.
Statistical analysis
Meta-analysis and statistical analysis have been conducted with RevMan 5.4 software provided by the Cochrane Collaboration. The data were expressed as follows: the risk ratio(RR) was used to assess dichotomous data; the mean difference (MD) and standard mean difference(SMD) were used to assess continuous data, and a 95% confidence interval(CI) was used for interval estimation. Heterogeneity was tested using P statistics. If there is homogeneity(P >0.1,I< 50%), we will select the fixed effects model. If not, we will use the random-effects model. Subgroups analysis was performed to avoid heterogeneity.
Results Literature search
The database search identified 1,244 publications and relevant abstracts. After an initial review of abstracts and titles, 844 records were excluded for duplicate (Figure 1). The remaining 12 studies were reviewed in full text, and 10 studies [14-23] were finally included in the analysis.

Figure 1 Literature search process
Characteristics of study
The basic characteristics of the 10 RCTs are shown in Table1. All of the RCTs were conducted and published in Chinese. Seven hundred and twenty-four DKD patients were enrolled, with the size of the sample ranging from 40 to 100. All participants were undergoing basic treatment, such as control of blood pressure and administration of hypoglycemic drugs. All patients were adults over the age of 18. All 10 cases have used PGEl in the control groups [14-23] and the course of treatment ranges from 2 to 12 weeks.
Random sequence generation
Amongthese10cases,4adopted computer-programmed random sequencing, random number table, or random number generator were evaluated as low risk of bias [15,16.19.22]. Other methods or details that do not mention random sequence generation are evaluated as high risk of bias (Figure 2, Figure 3).

Figure 2 Risk of bias graph

Figure 3 Risk of bias summary
Allocation concealment
Of the 10 cases, 4 used sealed-envelops, random list, or random assignment method to determine whether the grouping was given a low risk of bias based on allocation concealment [15,16,19,22]; other 6 cases that the method of allocation concealment has not been described were evaluated as unclear risk of bias.
Blinding of participants and personnel
Due to the nature of the active control, none of the studies has been performed blindly.
Blinding of outcome assessment
For outcome blinding, none of the studies adopted single-blind or double-blind methods to assess the intervention measure.
Incomplete outcome data
Nine cases had no attrition for missing participants, which were considered as having a low risk of bias [15-21,23]; one case missed participants due to the nosocomial infection [14] and one case missed participants due to the adverse events, so we consider it as high risk of bias [22].
Selective outcome reporting
None of the cases registered protocols, but all cases were reported expected outcomes, outcome indicators were complete, and were thus, all cases were considered as low risk of bias.
Other sources of bias
All cases were at low risk of bias due to a lack of clear evidence to display other obvious biases.

Results
Efficacy rate
The effectiveness rate was evaluated in in 6 trials. According to the course of treatment in TGT combined with PGE1,6 trials[15-17,21,22]were divided into 2 subgroup analyses (Figure 4). Comprehensive meta-analysis showed that TGT combined with PGE1 (RR=1.17,95%CI(1.09,1.25),P< 0.0001.fixed model,P= 0%,6 trials), subgroup meta-analysis showed that the course of treatment ≥ 12 weeks in TGT combined with PGE1(RR = 1.14,95% CI (1.05-1.24), P= 0.001,fixed model,P= 0%,5 trials), and the course of treatment <12 weeks in TGT combined with PGE1(RR=1.27,95%CI:(1.04,1.54), P= 0.02,fixed model, 1 trial ).
24 h Upro
Eight trails [15-22] showed 24 h Upro results of 628 DKD patients. With the result of homogeneity after the test (P<0.00001, I= 100%), the random effect model was used. The results suggested that the treatment group (TGT combined with PGE1)was more effective than the control group (PGE1)in decreasing 24 h Upro (MD=-1.14.95%CI(-2.05,-0.22), Z= 2.43, P=0.01)(Figure 5).
Ser
Five trials [17-19,21,22] showed Scr levels of 378 DKD patients. After the heterogeneity test (P<0.00001, I =89%), a random effect model was used to pool the data. Compared with other groups, the patients in Cui's results were quite different, so we selected the SMD effect measure. The results indicated that there is a significant difference existing between the 2 groups in decreasing Scr levels(SMD=-2.60,95% CI(-5.01, -0.20),Z=2.12,P=0.03)(Figure6).

Figure 4 Effect of tripterygium glycosides tablet combined with alprostadil on effectiveness rate in diabetic kidney disease patients
BUN
There trials [16, 19, 22] showed BUN levels of 264 DKD patients. After heterogeneity test (P < 0.00001, I2 = 98%), a random effect model was used to pool the data. The results indicated that there is no significant difference existing between the 2 groups in decreasing BUN levels (MD = –1.03, 95% CI (–4.17, 2.11), Z = 0.64, P = 0.52) (Figure 7).
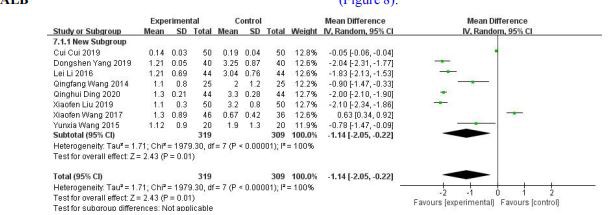
Figure 5 Effect of tripterygium glycosides tablet combined with alprostadil on 24 h Upro of diabetic kidney disease patients

Figure 6 Effect of tripterygium glycosides tablet combined with alprostadil on serum creatinine of diabetic kidney disease patients

Figure 7 Effect of tripterygium glycosides tablet combined with alprostadil on blood urea nitrogen of diabetic kidney disease patients

Figure 8 Effect of tripterygium glycosides tablet combined with alprostadil on the albumin in diabetic kidney disease patients
Other results
The results of TG indicated that there is no difference existing between the 2 groups in decreasing TG while the comparison of IL-6 and TNF-α showed a significant difference between the 2 groups (Table 2).
Adverse events
Seven trials [15, 16, 18–20, 22, 23] showed adverse events in 544 DKD patients. Xiaofen Wang [122] reported 2 patients gave up from the treatment and control groups because of the severe adverse events. Another 45 patients with adverse events did not withdraw from the study (Table 3).
After the heterogeneity test (P = 0.11, I2 = 44%), a fixed-effect model was used to pool the data. The results indicated that the treatment group (TGT combined with PGE1) was more effective than the control group (PGE1) in decreasing the rate of adverse events (OR = 0.50, 95% CI (0.26, 0.94), Z = 2.15, P = 0.03) (Figure 9).

Table 2 Results of other outcomes

Figure 9 Effect of tripterygium glycosides tablet combined with alprostadil on adverse events in diabetic kidney disease patients

Table 3 Details of the adverse events in the different treatments among diabetic kidney disease patients
Discussion
For the first time, this systematic review focused on evaluating the efficacy and safety of TGT combined with PGE1 in the treatment of DKD. The current study results showed the effectiveness of TGT combined with PGEl in treating DKD patients when contrasted with PGEl groups. More importantly, the current analysis support that TGT combined with PGE1 is more effective than PGE1 alone in the treatment of DKD, which can significantly reduce the 24 h-Upro and adverse events of DKD patients. Therefore, we speculated that TGT combined with PGE1 could play an essential role in the treatment of DKD, for its value in effectiveness and safety, which provides a potentially useful and adverse event for the traditional PGE1 therapy in the treatment of DKD.
The systemic treatment therapy of DKD is still complicated until now. PGE1, a drug that can effectively expand renal blood vessels and directly act on glomerular arteries, was found to have protective effects on renal function in the treatment of DKD by inhibiting insulin resistance in renal tubule epithelial cells via autophagy and reducing proteinuria in recent years [24,25]. And in clinical, Wang et al.[23]reported that TGT combined with PGE1 can protect the renal function of DKD patients, reduce hypersensitive C-reactive protein to promote renal vascular smooth muscle hyperplasia. TGT combined with PGE1 in the treatment of DKD has also been reported indicating that this treatment method can reduce the level of TNF-α to debase the level of sodium retention and mesangial cell proliferation.
TGT is a natural anti-inflammatory phytomedicine that has been used for inflammatory and various autoimmune diseases for years [26]. Researchers found that TGT at the dose of 60 mg/day has clinical effectiveness in reducing proteinuria and hematuria by suppressing renal microinflammation, GS, and podocyte injury in DKD [27. Moreover, PGE1 was used to cure vasodilation, inhibit platelet aggregation, and reduce peripheral blood vessels resistance. New research [28] showed that PGE1 could stabilize the cell membrane, inhibit the release of IL-1, TNF-α, and other inflammatory mediators, and reduce the formation of antigen-antibody reactions.
Early studies have shown that microalbuminuria (>300 mg/24 h proteinuria) has a high correlation with the further development of end-stage renal failure [29, 30]. Therefore, the treatment of reducing 24 h-Upro can preserve the kidney function of DKD patients. According to the course of treatment, TGT combined with PGE-1 shows clinically effective in reducing the 24 h-Upro. Besides, our results showed that the levels of I-6 and TNF-αa in TGT combined with PGE1 were significantly decreased. However, due to the small number of trials, although the heterogeneity is less than 50%. there is still a risk of deviation. Therefore, a larger size of sample test is necessary.
In addition, for BUN, ALB, and TG, the meta-analysis results indicated that the therapeutic effect of TGT combined with PGE1 was not significantly different from that of PGE1 alone. For this result, we need to carefully consider whether the causes of heterogeneity are related to the assignable objective factors like the type of diabetes mellitus; the course of the disease; the stage of DKD; whether a similar treatment has been received in the past; different drug types of basic treatment given to patients by the hospital.
Adverse reactions have always been a focus of concern. Surprisingly, this review implied that the TGT combined with PGE1 therapy-induced lower adverse events than controls. In most studies, the dose of TG was 60 mg/d, and the course of treatment was ≤ 3 months; the effect is better and few adverse events. Nevertheless, there is no better efficacy found in longer courses of treatment. So we forecasted conservatively that this treatment plan might be beneficial to some patients in clinical. However, these results are only based on current research, and there is more space to explore in the future.
However, there are also some limitations to this systematic review:1) the overall quality of the included trials was moderate. Out of the 11 trials,5 were RCTs, and another 6 trials mentioned"random". None of the studies provided any detail on single or double-blinding and allocation concealment, which indicated the low quality of methodology and led to a high risk of selection and measurement bias; 2)the number of some outcome events was deficient. Several analyses were based on just 2 or 3 studies, which could affect the analysis of results;3)although we adopted an adequate search strategy to minimize publication bias, there are still some potential trials because of language restriction; 4)the study's inclusion was more concentrated in the same region and country(Asia)and lack of randomized clinical trials of Western and African races.

Conclusion
For the first time, our meta-analysis summarized the application of TGT combined with PGE1 therapy in DKD. We found that TGT combined with PGE1 has some advantages in treating DKD. Meantime, the incidence rate of adverse events is lower in TGT patients combined with PGE1 therapy. Furthermore, this treatment study requires a large sample, multi-center design, and high-quality clinical trials to ascertain its usage in a broader medical field.
References
1. Gall MA. Hougaard P, Borch-Johnsen K, Parving HH. Risk factors for the development of incipient and overt diabetic nephropathy in patients with non-insulin-dependent diabetes mellitus: a prospective, observational study. BMJ. 1997;314(7083):783-788.
2. Ritz E, Zeng XX, Rychlik I. Clinical manifestation and natural history of diabetic nephropathy.Contrib Nephrol.2011;170:19-27. 3. Badal SS, Danesh FR. Diabetic nephropathy: emerging biomarkers for risk assessment. Diabetes.2015;64(9):3063-3065.
4. Thomas MC, Brownlee M, Susztak K, et al. Diabetic kidney disease. Nat Rev Dis Primers. 2015;1:15018. Lim AK. Diabetic nephropathy-complications 5 and treatment Int. J Nephrol Renovasc Dis. 2014;7:361-381.
5. Boer IH, Rue TC. Hall YN, Heagerty PJ, Weiss
6( NS, Himmelfarb J. Temporal trends in the prevalence of diabetic kidney disease in the United States.JAMA.2011305(24):2532-2539. Lewis EJ, Hunsicker LG, Clarke WR,et al.
7. Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med. 2001;345(12):851-860.
8. Liu ZH, Li SH, Wu Y, et al. Treatment of membranous nephropathy with Tripterygium Wilfordi and steroid: a prospective randomized control trial. J Nephrol Dial Transplant. 2009;18:303-309.
9. Li LS.Clinical study of Tripterygium Wilfordi Hook in treating glomerulonephritis. Chin J Intern Med. 1981;20:216-220.
10. Cao Y, Huang X, Fan Y, Chen X. Protective effect of triptolide against glomerular mesangial cell proliferation and glomerular fibrosis in rats involves the TGF-β 1/Smad signaling pathway. Evid Based Complement Alternat Med. 2015;2015:814089.
11. Liang X, Chen B, Wang P. et al.Triptolide potentiates the cytoskeleton-stabilizing activity of cyclosporine A in glomerular podocytes via a GSK3β dependent mechanism. Am J Transl Res. 2020;12(3):800-812.
12. Hong Y. Peng J. Cai X.Zhang X, Liao Y, Lan L. Clinical efficacy of alprostadil combined with α-lipoic acid in the treatment of elderly patients with diabetic nephropathy. Open Med (Wars). 2017;12:323-327.
13. Luo C, Li T, Zhang C, et al.Therapeutic effect of alprostadil in diabetic nephropathy: possible roles of angiopoietin-2 and IL-18. Cell Physiol Biochem.2014;34(3):916-928.
14. Tu WP. Clinical observation of tripterygium wilfordii glycosides combined with alprostadil in treatment of stage I and IV diabetic nephropathy. Chinh RuralMed Pharm.2013;20(22):8-10.
15. Yang DS. Clinical effect of tripterygium glycosides tablets combined with alprostadil in treatment of diabetic nephropathy. Chin / Clin Rat Drug Use.2019;12(01):83-84.
16. Li L, Zeng Y.Study on the effect of tripterygium glycosides tablets combined with alprostadil in treatment of diabetic nephropathy.China Foreign Med Treat. 2016;35(03):11-13.
17. Wang YX. Clinical study on alprostadil and tripterygium glycosides in treatment of diabetic Guide. nephropathy.Chin Med 2015;17(09):920-921.
18. Wang OF, Jin H, Li F. Clinical research on alprostadil combined with tripteryginm glycosides tablet in treating diabetic nephropathy Med. Chin Pract proteinuria. N 2014;41(10):52-54.
19. Cui C. Therapeutic effect of alprostadil combined with tripterygium glycosides on diabetic nephropathy and its effect on inflammatory factors. Chin J Prim Med Pharm. 2019;26(15):1866-1870.
20. Ding QH. Clinical effect of tripterygium glycosides tablets combined with alprostadil in treatment of diabetic nephropathy. China Acad J Electr Publ House.2020;27(06):76.
21. Liu XF, Li YY. Effect of tripterygium glycosides tablets combined with alprostadil in treatment of diabetic nephropathy. J North Pharm. 2017;14(10):63.
22. Wang XF, Li XF, Guo HL, Li Min, Guo YF, Gong L, Tripterygium glycosides tablets combined with alprostadil injection in the treatment of diabetic nephropathy. Med. Hubei Univ 2017;36(04):341-344.
23. Wang XN,Yuan XY,Zhang MZ.Effect of tripterygium glycosides combined with alprostadil on cytokine concentration in patients with diabetic nephropathy.Chin J Chin Pharm. 2017;33(17):1625-1627.
24. Wei W, An XR, Jin SJ, Li XX, Xu M.Inhibition of insulin resistance by PGE1 via autophagy-dependent FGF21 pathway in diabetic nephropathy. Sci Rep.2018;8(1):9.
25. Luo C, LiT, Zhang C, et al. Therapeutic effect of alprostadil in diabetic nephropathy: possible roles of angiopoietin-2 and IL-18. Cell Physiol Biochem.2014;34(3):916-928. Wu W, Yang JJ, Yang HM, et al.Multi-glycoside
26. of Tripterygium wilfordii Hook. f. attenuates glomerulosclerosis in a rat model of diabetic nephropathy by exerting anti-micro inflammatory effects without affecting hyperglycemia. Int./ Mol Med. 2017;40(3):721-730.
27. Zhu B, Wang Y, Jardine M, et al. Tripterygium preparations for the treatment of CKD: a systematic review and meta-analysis. Am J Kidney Dis.2013:62(3):515-530.
28. Wei W, An XR, Jin SJ, LiXX, Xu M.Inhibition of insulin resistance by PGE1 via autophagy-dependent FGF21 pathway in diabetic nephropathy. Sci Rep.2018;8:9.
29.Wang XN,Yuan XY,and Zhang MZ.Effect of alprostadil injection combined with tripterygium glycosides tablets on cytokine concentration in patients with diabetic nephropathy. Chin J Clin Pharm.2017;33(17):1625-1627
30.de Zeeuw D, Remuzzi G, Parving HH, et al. Proteinuria, a target for renoprotection in patients with type 2 diabetic nephropathy: lessons from RENTAL. Kidney Int.2004;65(6):2309-2320.






