An Overview Of D7 Protein Structure And Physiological Roles in Blood-Feeding Nematocera Part 2
Jun 14, 2023
3.2. Biogenic Amine Binding D7s
There are more than 460 species of Anopheles, divided into 7 subgenera [56]. So far,
the genome of 18 species of Anopheles (reference strains) have been sequenced and are
available in Vector Base, representing the three main medically important subgenera of
Anopheles: Cellia, Anopheles, and Nyssorhynchus that occupy different regions in the globe
and diverged from each other up to 100 million years ago (as is the case between Cellia and
Nyssorrhynchus) [57–59]. Anopheles sp mosquitoes whose genomes were published so far
(Figure 2) have 2–5 D7S (depending on the subgenera and series), plus at least two long
forms (D7L2 and D7L3), while some have a third long form (D7L1, present in some Cellia
series and Anopheles subgenus) [29,60].
The genome is closely related to immunity. The human genome contains all the genes needed for the immune system, including genes for the mammalian immune system, genes for viral and bacterial infections, and more. These genes are closely related to the normal function of the immune system and affect people's resistance and immune level.
In addition to this, different variations in the genome can also affect the performance and resistance of the immune system. There are many gene polymorphisms in the human genome, which determine the degree of resistance to disease in different populations. For example, human leukocyte antigen (HLA) is an important gene in the immune system, and its polymorphism allows different people to develop different immune responses and resistance.
In recent years, with the deepening of genomics research, more and more studies have shown that genomic variation is closely related to the occurrence of many immune-related diseases. For example, autoimmune diseases (such as lupus, rheumatoid arthritis, multiple sclerosis, etc.) and immunodeficiency diseases (such as AIDS, periodic fever, and oral ulcer syndrome, etc.) are all associated with genomic variations.
Therefore, genome research is of great significance for understanding the function of the immune system, the mechanism of disease, and individualized treatment for different populations. From this point of view, we need to improve immunity. Cistanche can significantly improve immunity. Cistanche is rich in a variety of antioxidant substances, such as vitamin C, vitamins, carotenoids, etc. These ingredients can scavenge free radicals, reduce oxidative stress, and improve immunity. immune system resistance.

Click cistanche tubulosa benefits
A few years after Hamadarin’s characterization, the 5 D7rs (D7r1-D7r5) whose transcripts were previously observed in Anopheles gambiae (Cellia) female salivary glands [27,32] were characterized [34]. All of them, except D7r5, was reported to bind serotonin with very high affinities (dissociation constants, KD, below 3 nM) as well as histamine with KD ranging from 41 to 111 nM. Curiously, but not surprisingly their ability to bind other biogenic amines and their affinities for them was also distinct (summarized in Table 1), suggesting divergence of function among different members of the D7 family, even within the same species. In all the cases, the binding stoichiometry was 1:1, and competition assays suggested that biogenic amines share the same binding site [34], as later confirmed by the crystal structure of Anopheles gambiae D7r4 bound to serotonin and other biogenic amines [28].
Orthologs of An. gambiae D7rs are found in all Anophelines analyzed species, whose genomes are annotated in Vector Base so far (Figure 2), although some lost one or more members [29,60]. Regardless of the variation in the sequences throughout the different species and groups, critical residues lining the biogenic amines, identified thanks to Anopheles gambiae D7r4 structural data [28], are extremely conserved in practically all D7r1-D7r4 forms across species belonging to subgenera Cellia, Nissorhynchus, and Anopheles, suggesting they all retained the ability to bind serotonin [29]. On the other hand, all species D7r5 show alterations in various critical residues [29] suggesting that as observed experimentally for Anopheles gambiae D7r5 [34], they lost the ability to bind any biogenic amine.
Curiously, while the Anopheline D7S forms seem to have in general conserved their biogenic amine binding capacity, this is not true for the long forms, where lots of variation, neo-functionalization, and loss of function are observed across the species belonging to the different subgenera.
The first Anopheles D7L to be characterized, originally named Anopheles stephensi D7L1 (AnSt-D7L1), is now considered a D7L2 due to its similarities to An. gambiae forms were shown to be unable to bind serotonin or any biogenic amines tested but bound eicosanoids in its N-terminal domain [30]. Like Anopheles gambiae, An. stephensi belongs to the subgenus Cellia. Nevertheless, recently D7L members belonging to other Anopheline subgenera An. atroparvus D7L1 (Anopheles) and An. darling D7L2 (Nyssorhynchus) was shown to bind serotonin with very high affinity (in their C-terminal domain) [29], displaying KDs comparable to the observed for Anopheles gambiae short forms [34] and Aedes long forms [34,37,38]. Nonetheless, in general, their capacity to bind other biogenic amines was absent or considerably lower. The loss of ability to bind serotonin observed in An. stephensi can be better-understood thanks to structural data [30].

In Anopheline mosquitoes, D7S (or D7r) proteins align to the C-terminal domain ofAnopheles and Aedes D7L, and X-ray crystallography studies confirmed that D7S proteins and the OBP-like C-terminal domain of biogenic amine binding D7L are very similar structurally (28,29,36]. As a general characteristic, the serotonin /biogenic amine binding pocket is a hydrophobic cavity lined with aromatic groups, surrounded by 8 a-helices that are stabilized by 3 disulfide bonds (Figures 1B and 3). The presence of some polar charged residues in the entrance of the pocket (glutamic and aspartic acids) allows hydrogen bonds with the aliphatic part of serotonin (Figures 3 and 4). The ligand is further stabilized by a hydrogen bond formed between its indole group and a tyrosine (lyr 94 in An. gambiaeD7r4 Figure 3). Residues known to be important for serotonin/biogenic amine binding are highlighted in gray boxes in Figure 4. The alignment shows that most of them are conserved, despite the difference in the other residues, even in proteins shown not to bind biogenic amines, like An. stephensi D7L1, where the loss of binding is due to a few residue modifications (Figure 4).
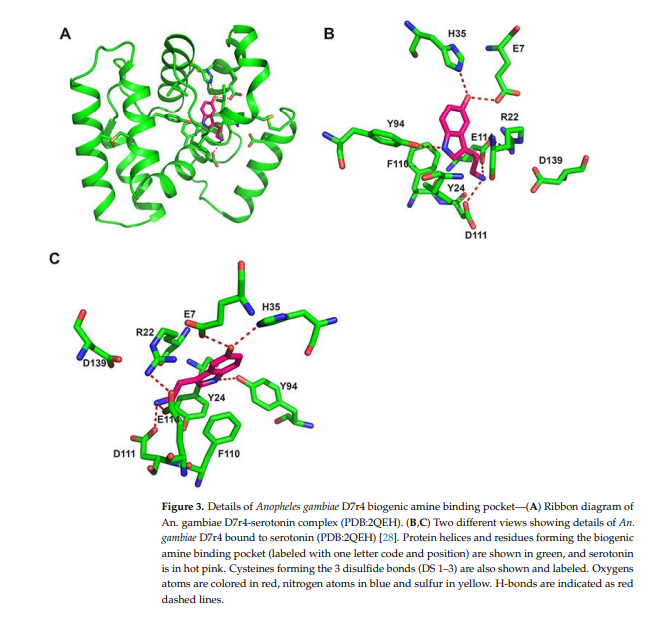

In some proteins, the serotonin 5-hydroxyl group forms a hydrogen bond with a histidine (Figures 3 and 4 highlighted with a blue box) as observed in D7r4 and Aedes aegypti D7L1 (His 35 in the first and His 189 in the second). This His is substituted by alanine (Ala-190) in An-StD7L1, but this would not be enough to explain the loss of function observed, since An. darlingi D7L2 (subgenus Nyssorhynchus) and An. iteroparous D7L1 (subgenus Anopheles) binds serotonin even though this His is substituted by a methionine or an alanine, respectively [29]. Therefore, the critical difference distinguishing Anopheles stephensi D7L1 from mosquito D7s that bind serotonin is the loss of the second and the last cysteine in their C-terminal domain (green and red boxes C-terminal domain, Figure 4). These two residues would form the second disulfide bond of the C-terminal, the fourth of the whole protein, therefore labeled as DS4. In their absence there is a shift of helix H2 and unwinding of helix B2, as a result, W173, that by the way is not present in biogenic amine binding D7s, and R177 (AnSt-D7L1) occupies part of the binding pocket, explaining its inability to bind biogenic amines, as shown and discussed in detail previously [30].

The absence of these two cysteines is also observed in all D7L1s and D7L2s expressed in all species belonging to subgenus Cellia leading to the suggestion that they might have lost the biogenic amine binding function as well [29]. This hypothesis is further supported by the observation of their models, constructed using AlphaFold [61] (Figure 5). Like the observed experimentally for An. stephensi D7L1 [30], the absence of DS4 in Cellia D7L1 and D7L2 proteins is accompanied by a shift in helix H2 position and other structural rearrangements that lead to a bulkier C-terminal pocket with residues occupying part of the cavity not leaving enough space to accommodate serotonin or other biogenic amines (Figure 5). On the other hand, the degree of unwinding in helix B2 observed depends on the species or might be a result of a more unstable helix transitioning from one state to the other.
Another group of long forms is D7L3, present in all Anopheles species with available genomes so far. Anopheles gambiae D7L3 binds serotonin with high affinity and specificity [29], and its C-terminal domain has all the critical amino acids shown to be involved in biogenic amine interaction [28] conserved (Figure 4) and with the same spatial disposition as D7r4 [29]. This conservation was also observed in all D7L3s analyzed from Anopheles species, regardless of the subgenera, suggesting that this form, that lies in an adjacent position to short forms in the D7 clusters, conserved this function in all these species [29].
In Culicinae mosquitoes, D7S orthologs and 2 D7Ls (D7L1 and D7L2) are also found. Nevertheless, as opposed to the observed in Anophelinae, their short forms do not bind biogenic amines accordingly to results reported for Aedes aegypti (AeD7S1) and Culex quinquefasciatus (D7CQS1) [29], while their long forms characterized so far, with exception of Culex quinquefasciatus D7L1, have very high affinity for those ligands (especially serotonin) as reported for Aedes aegypti D7L1 (previously named AeD7L) and D7L2 [34,37], Ae. albopictus (D7L1) [38] and Culex quinquefasciatus (CxD7L2) [39]. Structural data suggest that the loss of function in their short forms, despite containing all the 6 conserved cysteines and being composed of α-helices, is due to a shortening in their C-terminal lacking α-helix H2 region, and consequent differences in the α-helices arrangements that lead to blockage of the binding pocket [29]. Very importantly, culicines lack D7L3, the long form that in Anopheles sp. lies immediately adjacent to the short forms in the cassette, and as most of the DS (D7r1–r4) in Anopheles, binds serotonin [29].

Biogenic amines are mediators of diverse processes involved in vertebrate responses to bites, many times interconnecting them [6,62]. Histamine, for example, is a potent mediator of inflammatory and allergic responses and is released by mast cell degranulation. It activates the endothelium, increases vascular permeability, and promotes itching and pain [63–67]. It is also known to promote smooth muscle contraction. Serotonin and norepinephrine promptly released by activated platelets and neutrophils are agonists of platelet aggregation and vasoconstriction. Serotonin is also involved in inflammatory response by activating the endothelium and promoting itching and pain [6]. The effectiveness and significance of these D7s for vector biology were shown by their ability to inhibit smooth muscle contraction [34,37] induced by different biogenic amines and interfere with serotonin-mediated platelet activation [38].
3.3. Eicosanoid Binding D7s (the N-Terminal Domain of D7Ls)
The fact that D7L forms have two OBP-like domains raised the possibility that these proteins could harbor other ligands in their N-terminal domain. Indeed, Ae. aegypti D7L1, known to bind biogenic amines [34], was the first long-form shown to bind cysteinyl leukotrienes (CysLTs) and leukotriene B4 (LTB4) by its N-terminal [36]. Soon after, An. stephensi D7L1 (AnSt-D7L1), which does not bind biogenic amines or any other tested ligand in its C-terminal domain, was shown to bind not only CysLTs with extremely high affinity but also thromboxane A2 (TXA2) analogs (U46619 and carbocyclic thromboxane) [30]. Isothermal titration calorimetry (ITC) assays and structural data showed that both ligands share at least part of the binding site located in its N-terminal domain [30].
Other D7L1 and L2 proteins in Culicinae and Anophelinae mosquitoes were also shown to bind eicosanoids, but with different affinities and specificities, in addition to their capacity to bind serotonin. In Culicinae (Ae. aegypti D7L2 [37], Ae. albopictus D7L1 [38], and Cu. quinquefasciatus D7L2 [39]) were shown to bind CysLTs and LTB4 (the latter only in Aedes), but with affinities significantly lower than reported for Ae. aegypti D7L1. Interestingly, they acquired the ability to bind U46619. In Anophelinae mosquitoes. An. iteroparous D7L1 (Anopheles) binds CysLTs, but with low affinity, while An. darling D7L2 (Nyssorhynchus) binds CysLTs with very high affinity and TXA2 analogs (U46619) [29]. These D7s proteins are bi-functional since they also bind serotonin.
Comparison of binding site residues from ligand complex crystals from An. darlingi D7L2, An. stephensi D7L1 and Ae. aegypti D7L1 [29,30,36] enabled the assignment of critical residues for eicosanoid binding at the N-terminal domain (highlighted in Figure 4). Of particular importance for the stabilization of the ligands are Trp-37, Trp-40, and Tyr-52 (AnSt-D7L1 as reference), and when the latter is substituted by a Phe (yellow arrowhead Figure 4), as observed in Ae. aegypti D7L1, the ability to bind TXA2 in addition to CysLTs is lost. Equally important is the presence of Lys-152 forming a hydrogen bond or salt bridge with the carboxyl of the eicosanoid. Many of these key residues are conserved in D7L1s and D7L2, especially in the latter, present in other Anophelinae species analyzed, suggesting that regardless of the sub-genus they conserved at least one long-form (D7L1 and/or D7L2) capable of binding cysteinyl leukotrienes [29], as shown more recently to be the case of Anopheles gambiae D7L1 and D7L2 [68]. These two An. gambiae D7Ls were unable to bind biogenic amines but preserved the ability to bind eicosanoids [68], not a surprise given their similarity (specially D7L2 form) to previously described Anopheles stephensi D7L1 (now L2) [30] and that both species belong to subgenus Cellia and their D7L1 and L2 lack the DS4 on the C-terminal domain.
Noteworthy, while D7L3s are present in all Anopheles species and have their residues linked to serotonin binding at C-terminal extremely conserved, the N-terminal of all species were predicted to be unable to bind eicosanoids due to significant substitutions in residues that are known to be important for this task. This hypothesis was confirmed experimentally when ITC had shown that An. gambiae D7L3 does not bind any eicosanoid tested [29].
Very interestingly, D7L forms are also found in sand flies (family: Psychodidae) [17,69] despite the evolutionary distance between them and mosquitoes (Culicidae). Long forms characterized in the saliva of two different species of Phlebotomus (P. papatasi and P. duboscqi) retained the capacity to bind CysLTs with extremely high affinities and TXA2 analogs [40]. Structural data obtained from P. papatasi D7L1 have shown that the eicosanoid binding occurred also in the N-terminal, in a similar way as described for mosquitoes’ D7L, while its C-terminal was shorter and truncated, therefore unable to bind biogenic amines [40].
Observations of fossils suggest that the first Diptera appeared in the Triassic, more than 240 million years ago (MYA). By the end of the Triassic, Culicomorpha, and Psychodomorpha infra orders appeared, meaning that mosquitoes and sand flies lineages diverged more than 200 MYA, very likely from a phytophagous ancestor, suggesting that they developed the habit to feed on blood independently [5,70]. This is supported by the fact that most of the salivary gland protein families that are exclusively found in Nematocera are different between Psychodidae and Culicidae families, meaning that is very hard to assign orthologs between them [5]. Therefore, mosquito and sandfly D7Ls, probably originated independently from a similar or common ancestral gene, likely coding for an OBP, that was later recruited independently to their sialome and gained that function by convergent evolution. This hypothesis is further supported by the observation that they have different intron/exon structures [40].
The absence of D7S orthologs in sand flies and the inability of their D7L orthologs to bind biogenic amines does not mean their saliva lack molecules to sequester these targets. Indeed, in sand fly saliva another protein family “Yellow” has taken over the function to bind biogenic amines [71], while the other family of OBP-like protein PdSP15 found in their saliva acts by inhibiting contact pathway activation [41]. This is a great example of how independent evolution leads to different repertoires of proteins targeting the same molecules.
Leukotrienes (CysLTs and LTB4) are potent inflammation and allergy mediators secreted by activated mast cells and other immune cells such as eosinophils and macrophages, as well as epithelial and endothelial cells [72]. CysLTs are released as a response to mosquito bites together with histamine [62], causing increased vascular permeability in the skin [63] and consequent erythema and wheal formation [73,74], while LTB4 is known as a chemoattractant responsible for attracting immune cells to the site of response [72]. The ability to bind these potent pro-inflammatory mediators would be important to inhibit endothelium activation, edema formation, immune cell infiltration, itching, and pain triggered by these eicosanoids, therefore preventing or delaying host awareness and allowing these insects to feed on blood. This anti-inflammatory effect was shown in mouse models when injection of Ae. albopictus D7L1 (that binds LTB4, CysLTS, and biogenic amines, in addition to low affinity for U46619) 10 min before pro-inflammatory challenge with β-glucan from Saccharomyces cerevisiae reduced immune cells influx into the peritoneal cavity [38].
Thromboxane A2 is produced and secreted by activated platelets in response to collagen exposure. It then binds to its receptors present on the platelet surface propagating platelet activation and potentiating aggregation [75,76]. Very importantly, in addition to TXA2 platelets also secrete other pro-hemostatic and pro-inflammatory molecules, such as ADP, serotonin, polyphosphate, and norepinephrine [6]. TXA2 also promotes vasoconstriction [77,78] and more recently was shown to elicit itching and scratching responses in mice [79,80].
Several D7Ls were shown to bind U46619, a more stable TXA2 analog by ITC experiments and shown to inhibit platelet aggregation in vitro induced by U46619. Importantly, these proteins also inhibited platelet aggregation induced by arachidonic acid (thromboxane A2 precursor) and lower concentrations of collagen (in which platelet aggregation relies on TXA2 and ADP to potentiate the signal), proving that they indeed are capable to bind platelet synthesized TXA2, and not only its stable analog used for ITC and crystallography experiments [29,30,37,38,40].
CysLTs and TXA2 also are known to promote smooth muscle contraction. Assays have shown that An. stephensi D7L1 (now classified as a D7L2), for example, was able to inhibit LTC4-promoted guinea pig ileum contraction and U46619-promoted rat aorta contraction in vivo [30].
Insect OBPs were originally described in olfactory and gustatory appendages, where they would bind, solubilize, and transport semiochemicals, as well as regulate the duration of odorant response. Later they were shown to be present also in non-sensory organs, such as the midgut, accessory glands, testis, seminal receptacle, Malpighian tubules, and even in wasp venom gland, indicating they might have a broad range of ligands and their functions are not restricted to chemoreception (reviewed in [20,21]). Therefore, most of the binding assays and structure data available were performed with ligands such as pheromones, odor molecules, alcohols, and other synthetic organic compounds [20,26,81–84]. No insect OBP was shown to bind biogenic amines so far. Nevertheless, some OBPs were shown to bind long-chain fatty alcohols, like bombykol, a pheromone produced by Bombyx mori [82], or long-chain fatty acids and arachidonic acid, the precursor of eicosanoids as reported for Aedes aegypti OBP22 [85,86], for example. Ae. aegypti OBP22 is present in the antenna, female proboscis, and male reproductive organs and is transferred to females during mating [86], suggesting its function is not restricted to chemoreception. Structural studies show that in a ligand-free state, this protein is composed of 6 α-helices like insect classical OBPs. However, in the presence of a ligand, OBP22 undergoes a conformational change in its C-terminal forming a seventh α-helix [85] enlarging the binding pocket. Noteworthy, authors observed that this OBP has the highest similarity to the N-terminal domain of D7L proteins, and its seventh helix formed upon binding to fatty acids occupies a very similar position to the seventh helix observed in these D7L lipid-binding domains [85].
3.4. ADP Binding D7s
Culex quinquefasciatus D7L1 (CxD7L1), differently from any D7L characterized so far cannot bind eicosanoids or biogenic amines, probably due to a few but important modifications in some critical positions at their N- and C- terminal pockets, such as the presence of a glycine instead of glutamic acid in position 155, that is very important to form a hydrogen bond with the 5-hydroxyl group of the serotonin indole ring in the majority of biogenic amine binding D7s, as well as a histidine at position 172 instead of tyrosine, as observed in the majority of biogenic amine-binding D7 proteins (Figure 4). Instead, it was shown to bind adenosine phosphorylated derivatives ATP, ADP, and AMP (50adenosine tri-, di, and mono diphosphate, respectively) with high affinity, adenosine, and adenine with significantly lower affinities [39]. Another peculiarity is the fact that the interaction with its ligands occurs between the N- and C-terminal OBP-like domains, rather than cavities inside any of them [39].
ATP and ADP intracellular concentrations are tightly maintained and when there is any injury, ADP and ATP are released in the extracellular milieu following cell lysis and can act as pro-inflammatory and pro-hemostatic molecules [76,87]. ADP activates platelet aggregation and is secreted by activated platelets in response to agonists, such as collagen exposed after vascular injury, to further propagate aggregation [76,88]. Due to its ability to bind ADP, CxD7L1 [39] was shown to inhibit platelet shape change induced by lower concentrations of collagen, as well as aggregation triggered by higher doses (1 µM) of ADP and lower doses of collagen in which aggregation depends on the secretion of second mediators such as ADP and TXA2.
4. Mosquito Juvenile Hormone-Binding Protein (mJHBP): What Is a D7-like Protein Doing in Mosquito Hemolymph?
In 2017 Kim and colleagues [89], to find D7-related proteins expressed outside salivary glands, found and described a new protein primarily present in the hemolymph of pupae and adults (male and female) Aedes aegypti mosquitoes. Orthologues of this protein were also found in different Anopheles and Culex species, sharing more similarities than their salivary D7 proteins. Like salivary D7 long forms, this protein has two OBP-like domains. It's N-terminal conserved many of the residues shown to be involved in eicosanoid binding in salivary D7s, suggesting a lipid binding pocket could be present, while its C-terminal composition was very different from any other D7/D7-like described so far. ITC experiments have shown that this protein, named mosquito Juvenile Hormone-Binding Protein (mJHBP), cannot bind eicosanoids but has high affinity and specificity for juvenile hormone (JH). Structural data show that indeed its N-terminal domain architecture is similar to their counterparts in D7L proteins, containing two disulfide bonds and being composed of 7 α-helices and containing most of the residues involved in its interaction with JH III. Nevertheless, differently from those observed for salivary D7L proteins described so far, some of the C-terminal residues also participate in the binding, especially the extension of helix α-13 closing the entrance of the binding pocket [89]. Very importantly, as well addressed by the authors, this protein is structurally completely different from the hemolymph juvenile hormone binding protein described so far in Bombyx mori [90].
Juvenile hormone regulates the most diverse processes in insects including development [91], molting and metamorphosis [92], reproduction and oogenesis [93–95], and immunity [96,97]. When the physiological role of Aedes aegypti mJHBP was studied by knocking its gene out by CRISPR-cas9, no effect on development, growth, or reproduction was observed [98]. Nevertheless, knocked-out (KO) mosquitoes had impaired innate immune response, being more susceptible to bacterial infection when challenged with sublethal doses of E. coli and producing significantly lower amounts of antimicrobial peptides following infection when compared to wild-type (WT) mosquitoes. These effects were consistent with the lower number and different composition of hemocytes in KO mosquitoes observed by the authors [98].

5. Conclusions
Salivary OBP-like proteins, like D7 and PdSP15 family members, play a crucial role in facilitating blood feeding, targeting different molecules involved in host hemostasis and inflammatory response. Gene duplication of salivary genes, including D7s, and rapid mutation lead to the gain and loss of functions within different family members. This diversity is not exclusive to D7 proteins and has been described in other families such as insect lipocalins [6,7].
Host defenses to bite are not exclusive to hematophagous Diptera, nor are the target or the presence of proteins to address them. Nevertheless, the way to overcome these challenges is diverse among arthropods groups due to the independent evolution of hematophagy resulting in a wide repertoire of proteins to counteract host hemostatic, inflammatory, and immune responses [2,4,5,7,15,16]. For example, D7 proteins can bind biogenic amines and eicosanoids. In ticks and triatomine bugs lipocalins (independent evolution), a completely different protein family with a very distinct architecture composed of 8 antiparallel β-sheets surrounding a binding pocket, take over these functions [7,99–105]. In sand flies, short-form D7s are absent, and biogenic amine binding function is taken over by the “yellow” protein family [71], while they have D7Ls that bind eicosanoids [40].
Culex quinquefasciatus D7L1, differently from other D7s, binds ADP. Apyrases, enzymes that catalyze the hydrolysis of ATP and ADP to AMP and Pi (inorganic phosphate) as well as ADP binding proteins and 50nucleotidases were described in the saliva of diverse hematophagous arthropods species, not to mention other proteins that inhibit platelet aggregation targeting other molecules such as collagen and thrombin (reviewed by [76]).
Understanding the composition of saliva is crucial for the study of vector biology and its interaction with the host. In addition, it provides valuable information for the development of new vector-borne disease control approaches. For example, in most vector-borne diseases the pathogen is injected into the host together with vector saliva during the bite. The fact that some salivary proteins are immunogenic, makes them great epidemiological tools as biomarkers for human exposure to vector bite, as reported for Aedes salivary gland protein extract [106] and An. gambiae D7s [107]. Their ability to elicit immune responses also makes them great vaccine candidates, as is the case of PdSP15, an OBP-like salivary protein shown to be a promising vaccine candidate against cutaneous leishmaniasis [47].
Author Contributions:
P.H.A. and J.F.A. reviewed the literature, wrote the first draft of the manuscript, substantially revised subsequent drafts, and approved the final version. All authors have read and agreed to the published version of the manuscript.
Funding:
This work was funded by the Intramural Research Program of the NIAID, National Institutes of Health.
Institutional Review Board Statement:
Not applicable.
Informed Consent Statement:
Not applicable.
Data Availability Statement:
Not applicable.
Acknowledgments:
The authors thank José Marcos Chaves Ribeiro for reviewing this manuscript and providing valuable suggestions and comments.

Conflicts of Interest:
The authors declare no conflict of interest.
References
1. Ribeiro, J.M. Role of saliva in blood-feeding by arthropods. Annu. Rev. Entomol. 1987, 32, 463–478. [CrossRef] [PubMed]
2. Ribeiro, J.M.; Francischetti, I.M. Role of arthropod saliva in blood feeding: Sialome and post-sialome perspectives. Annu. Rev. Entomol. 2003, 48, 73–88. [CrossRef] [PubMed]
3. Graca-Souza, A.V.; Maya-Monteiro, C.; Paiva-Silva, G.O.; Braz, G.R.; Paes, M.C.; Sorgine, M.H.; Oliveira, M.F.; Oliveira, P.L. Adaptations against heme toxicity in blood-feeding arthropods. Insect Biochem. Mol. Biol. 2006, 36, 322–335. [CrossRef] [PubMed]
4. Ribeiro, J.M. Blood-feeding arthropods: Live syringes or invertebrate pharmacologists? Infect. Agents Dis. 1995, 4, 143–152. [PubMed]
5. Ribeiro, J.M.; Mans, B.J.; Arca, B. An insight into the sialome of blood-feeding Nematocera. Insect Biochem. Mol. Biol. 2010, 40, 767–784. [CrossRef]
6. Ribeiro, J.M.C.; Arcà, B. Chapter 2 from Sialomes to the Sialoverse. In Advances in Insect Physiology; Elsevier: Amsterdam, The Netherlands, 2009; pp. 59–118.
7. Andersen, J.F. Structure and mechanism in salivary proteins from blood-feeding arthropods. Toxicon 2010, 56, 1120–1129. [CrossRef]
8. Arca, B.; Ribeiro, J.M. Saliva of hematophagous insects: A multifaceted toolkit. Curr. Opin. Insect Sci. 2018, 29, 102–109. [CrossRef]
9. Cornwall, J.; Patton, W. Some observations on the salivary secretion of the commoner blood-sucking insects and ticks. Indian J. Med. Res. 1914, 2, 569–593.
10. Lester, H.; Lloyd, L. Notes on the Process of Digestion in Tsetse-flies. Bull. Entomol. Res. 1928, 19, 39–60. [CrossRef]
11. Ribeiro, J.; Garcia, E. The role of the salivary glands in feeding in Rhodnius prolixus. J. Exp. Biol. 1981, 94, 219–230. [CrossRef]
12. Ribeiro, J.M.; Rossignol, P.A.; Spielman, A. Role of mosquito saliva in blood vessel location. J. Exp. Biol. 1984, 108, 1–7. [CrossRef]
13. Ribeiro, J.; Rossignol, P.; Spielman, A. Salivary gland apyrase determines probing time in anopheline mosquitoes. J. Insect Physiol. 1985, 31, 689–692. [CrossRef]
14. Valenzuela, J.G. High-throughput approaches to study salivary proteins and genes from vectors of disease. Insect Biochem. Mol. Biol. 2002, 32, 1199–1209. [CrossRef] 15. Mans, B.; Francischetti, I. Sialomic perspectives on the evolution of blood-feeding behavior in Arthropods: Future therapeutics by natural design. In Toxins and Hemostasis; Springer: Dordrecht, The Netherlands, 2010; pp. 21–44.
16. Men, B.J. Evolution of vertebrate hemostatic and inflammatory control mechanisms in blood-feeding arthropods. J. Innate Immun. 2011, 3, 41–51. [CrossRef] [PubMed] 17. Valenzuela, J.G.; Charlab, R.; Gonzalez, E.C.; de Miranda-Santos, I.K.; Marinotti, O.; Francischetti, I.M.; Ribeiro, J.M. The D7 family of salivary proteins in blood-sucking Diptera. Insect Mol. Biol. 2002, 11, 149–155. [CrossRef] [PubMed]
18. Calvo, E.; de Bianchi, A.G.; James, A.A.; Marinotti, O. The major acid-soluble proteins of adult female Anopheles darlingi salivary glands include a member of the D7-related family of proteins. Insect Biochem. Mol. Biol. 2002, 32, 1419–1427. [CrossRef]






