Cistanche Tubulosa Extract (Acteoside ≥ 8%) Factory
Apr 23, 2026
High‑Actives Ethanol Extract | Full‑Chain Supply | Ready Stock (Grade #3+)
Built for B2B brands
We supply high‑actives Cistanche tubulosa extract for nutraceutical, functional food, and wellness brands that need:
consistent marker compounds (Acteoside/Verbascoside; Echinacoside; total PhGs),
reliable, scalable manufacturing, and
documented quality control (HPLC markers + third‑party safety testing).
Flagship grade: Ethanol extract standardized to Acteoside ≥ 8% (Grade #3 and above available as ready stock).
Regulatory positioning: Supplied as a botanical ingredient for further manufacturing. Information is provided for quality and formulation purposes only

Who we are
According to our corporate profile, Chengdu Wecistanche Bio‑Tech Co., Ltd. was established in 2003 and is positioned as a Cistanche‑focused company with an integrated industrial chain, headquartered in Luopu County, Hotan, Xinjiang, China, with a large‑scale cultivation base and a GMP manufacturing facility. The company describes capabilities spanning seed breeding, cultivation, collection & storage, and GMP processing-supporting a full supply chain from farm to extract. (Source: WECISTANCHE "About Us" page text you provided.)
Manufacturing & QA capabilities highlighted in the profile include:
100,000‑class GMP production workshop
advanced ultrafiltration/nanofiltration purification & concentration equipment
10,000‑class microbiology incubation laboratory
certifications mentioned: SC, HACCP, SOCA, NOP and additional product/qualification claims listed on the page text

Why Cistanche tubulosa extract (and why high Acteoside)
Cistanche (Rou Cong Rong; Cistanches Herba) is known for its key bioactives-phenylethanoid glycosides (PhGs)-especially:
Acteoside (Verbascoside)
Echinacoside
stronger marker standardization (COA‑driven purchasing),
cost‑in‑use efficiency (lower dosage to hit label marker targets),
and batch consistency in finished products.
Our commercial focus: Grade #3 and above (Acteoside ≥ 8%+) with ready stock availability.

Full‑chain advantage: cultivation → extraction → sales (ready stock for Grade #3+)
For Grade #3 and above, we position ourselves as one of the few suppliers able to provide:
independent cultivation base (upstream control),
in‑house processing/extraction (process & QC control),
standardized marker specifications (Acteoside ≥ 8% and above),
and ready inventory for faster brand launches.

Evidence‑based QC: published HPLC method and traceable extract sourcing
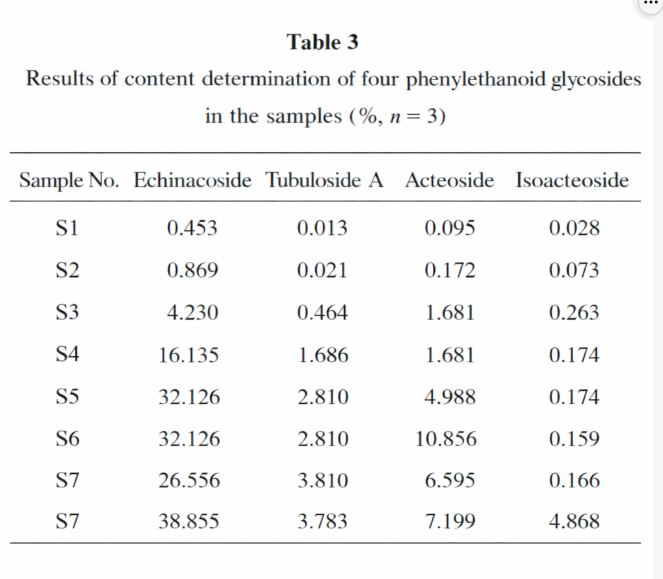
A peer‑reviewed HPLC study in China Pharmaceuticals (2022) established a method to simultaneously quantify four PhGs in Cistanche tubulosa extracts:
Echinacoside
Tubuloside A
Acteoside
Isoacteoside
Most important for procurement & due diligence:
The paper explicitly states that extract samples S5, S6, and S7 were purchased from Hotan Dichen Pharmaceuticals & Biotech Co., Ltd. (Hotan, Xinjiang, China)-providing third‑party traceability for commercial extract supply used in analytical evaluation.
The same paper reports strong method performance (linearity, precision, repeatability, recovery), supporting the reliability of HPLC marker quantification for Cistanche extracts-useful for aligning buyer/supplier QC standards.
Third‑party safety testing snapshot (pesticide residues & heavy metals)
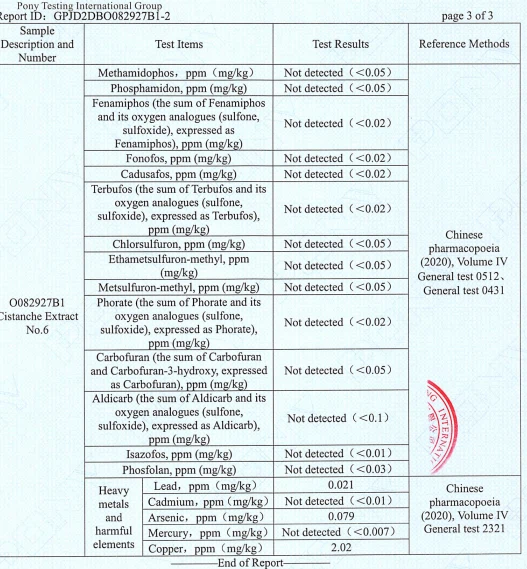
Based on the images provided from a PONY Testing International Group report for "Cistanche Extract No.6" (Applicant: Chengdu Wecistanche Bio‑Tech Co., Ltd.):
Received date: 2021‑08‑13
Test date: 2021‑08‑13 to 2021‑08‑20
Lot/Manufacturing date shown: 20210528
Reference methods shown: Chinese Pharmacopoeia (2020), Volume IV (general tests cited in the report)
Third‑Party Test Results (PONY): Echinacoside & Acteoside Content
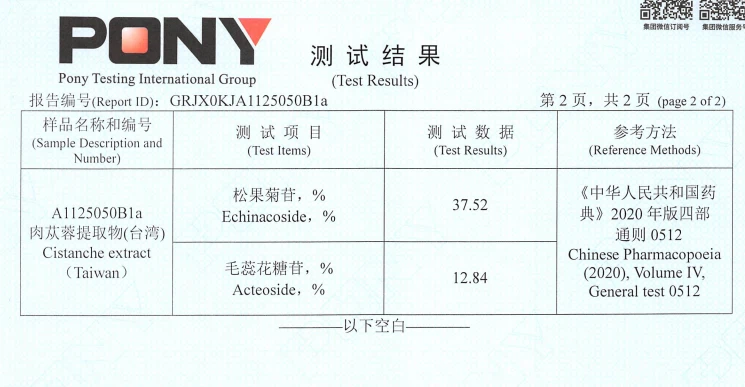
Test results issued by PONY Testing International Group for the sample labeled "Cistanche extract (Taiwan)". Reported contents: Echinacoside 37.52% and Acteoside 12.84%. Reference method: Chinese Pharmacopoeia (2020), Volume IV, General Test 0512.
HPLC Chromatogram of Standard Solution (STD‑1), 330 nm By Wecistanche
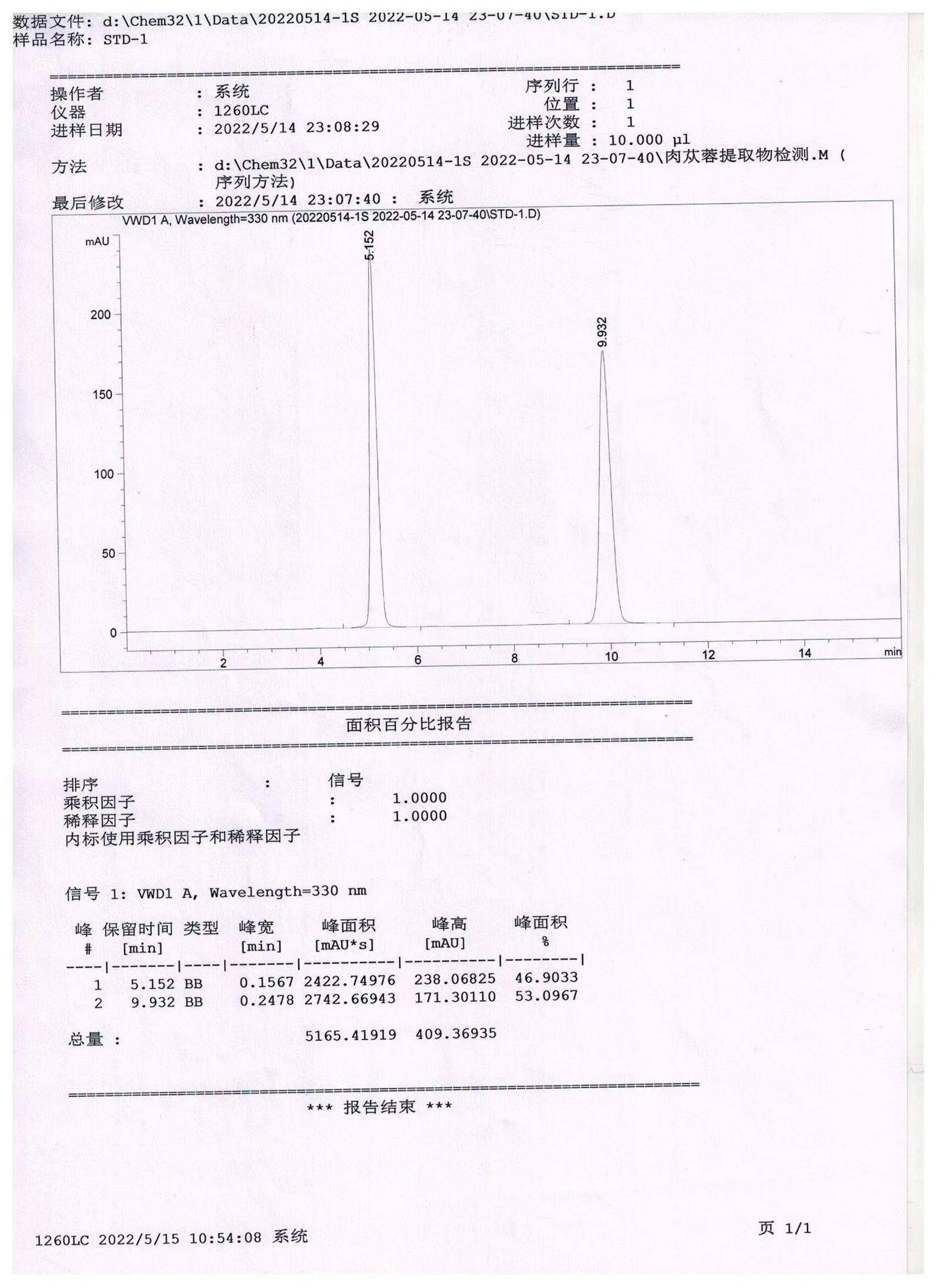
HPLC‑DAD analysis at 330 nm for the standard solution (STD‑1). The chromatogram shows two major peaks with retention times of approximately 5.152 min and 9.932 min; the area‑percentage results are about 46.90% and 53.10% (area normalization), used as a reference for method/system performance and quantitative analysis.
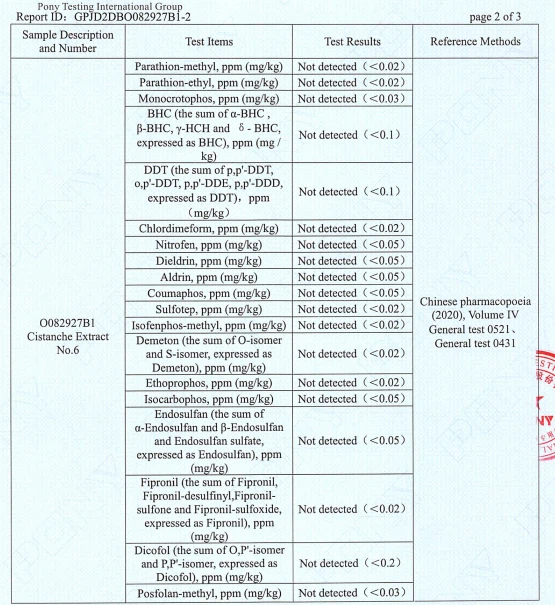
Pesticide residues
Multiple pesticide residue items are reported as Not detected at the stated detection limits (examples visible on the report pages include parathion‑methyl, parathion‑ethyl, monocrotophos, BHC, DDT, chlordimeform, nitrofen, dieldrin, aldrin, coumaphos, and others).
Heavy metals / harmful elements
The following values are visible on the report image:
Lead (Pb): 0.021 mg/kg=0.02ppm
Cadmium (Cd): Not detected (<0.01 mg/kg)
Arsenic (As): 0.079 mg/kg=0.079ppm
Mercury (Hg): Not detected (<0.0007 mg/kg)
Copper (Cu): 2.02 mg/kg=2ppm
If you want this section to look more "Western QA," I can format it as a one‑page "Third‑Party Testing Summary" with: report ID, lab name, method references, detection limits, and a clear "ND" legend-using only what is legible from your files.
Product grades (Grade #3 and above)
We can supply multiple grades of Cistanche tubulosa ethanol extract. For B2B buyers, we typically align grades by:
Acteoside (Verbascoside) minimum (e.g., ≥ 8% and above),
Echinacoside minimum,
Total phenylethanoid glycosides minimum,
and standard safety/quality parameters (ash, microbiology, heavy metals, pesticide residues).
Ready stock: Grade #3+.
Wecisanche Warehouse

Typical application formats (B2B‑friendly, conservative)
capsules / tablets
sachet powders / stick packs
functional beverage mixes
beauty‑from‑within formulas (where permitted)
We support brand teams with:
pre‑shipment COA,
sampling for R&D,
QC marker alignment (HPLC),
packaging and logistics planning.
Compliance disclaimer (recommended for US/EU)
This ingredient is a botanical raw material for further manufacturing. All information is provided for quality assurance and formulation reference. It is not intended to diagnose, treat, cure, or prevent any disease.
Reference
Chinese Pharmacopoeia Commission. (2020). Pharmacopoeia of the People's Republic of China (2020 Edition), Volume IV - General Chapter 0512. Beijing, China: China Medical Science Press.
Chinese Pharmacopoeia (2020), Volume IV, General test 0512".)
2022). [Title of the HPLC method paper]. China Pharmaceuticals (中国药业), [Volume]([Issue]), [Pages].






