Could The Fbromyalgia Syndrome Be Triggered Or Enhanced By COVID‑19? Part 2
Sep 28, 2023
Could fibromyalgia syndrome be exacerbated by COVID‑19?
Based on the clinical manifestations of FM, it has been proposed that people living with FM are an extremely vulnerable population in the setting of COVID-19 (Mohabbat et al. 2020). As shown in Table 1, musculoskeletal pain, fatigue, myalgia, and sleep impairment are some of the common complaints in both diseases (Choy 2015; Abdullahi et al. 2020; Tuzun et al. 2021; Nasserie et al. 2021). In this sense, it has been demonstrated that patients with FM are subject to more significant mental stress and anxiety during COVID-19. Thus, these patients have exacerbated symptoms of pain, fatigue, and sleep quality compared to control patients (without FM) with COVID-19 (Salaf et al. 2021). Therefore, the SARS-CoV-2 infection may be an essential factor in exacerbating the symptoms of FM patients.
Cistanche can act as an anti-fatigue and stamina enhancer, and experimental studies have shown that the decoction of Cistanche tubulosa could effectively protect the liver hepatocytes and endothelial cells damaged in weight-bearing swimming mice, upregulate the expression of NOS3, and promote hepatic glycogen synthesis, thus exerting anti-fatigue efficacy. Phenylethanoid glycoside-rich Cistanche tubulosa extract could significantly reduce the serum creatine kinase, lactate dehydrogenase, and lactate levels, and increase the hemoglobin (HB) and glucose levels in ICR mice, and this could play an anti-fatigue role by decreasing the muscle damage and delaying the lactic acid enrichment for energy storage in mice. Compound Cistanche Tubulosa Tablets significantly prolonged the weight-bearing swimming time, increased the hepatic glycogen reserve, and decreased the serum urea level after exercise in mice, showing its anti-fatigue effect. The decoction of Cistanchis can improve endurance and accelerate the elimination of fatigue in exercising mice, and can also reduce the elevation of serum creatine kinase after load exercise and keep the ultrastructure of skeletal muscle of mice normal after exercise, which indicates that it has the effects of enhancing physical strength and anti-fatigue. Cistanchis also significantly prolonged the survival time of nitrite-poisoned mice and enhanced the tolerance against hypoxia and fatigue.

Click on feeling tired all the time
【For more info:george.deng@wecistanche.com / WhatsApp:8613632399501】
It is worth mentioning that the acute symptoms of COVID-19 can persist in COVID-19 survivors after their recovery (Fig 1) (Kucuk et al. 2020; Nalbandian et al. 2021). Thus, it is unclear if the exacerbation of FM symptoms returns to normal after patients with FM have recovered from COVID-19 (Salaf et al. 2021). Nonetheless, it has been shown that patients suffering from pre-existing musculoskeletal pain conditions before COVID-19 seem to experience increased intensity, extension, or frequency of their symptoms for at least the first seven months after infection (Fernández-de-las-Peñas et al. 2021b, 2022). Thus, we might speculate that the enhanced FM symptoms in post-COVID situations can also last for an extended time. Patients with FM are at increased risk of mood disorders, even in non-pandemic situations (Clauw 2014, 2015; Häuser et al. 2017). During the COVID-19 pandemic, the social and economic environments where people live and work suffered changes (Karos et al. 2020). Thus, the COVID-19 pandemic prompted feelings of isolation, uncertainty, depression, mental stress, generalized anxiety, and fear of the virus, mainly in vulnerable subgroups (Liguori et al. 2020; Clauw et al. 2020; Varga et al. 2021). Indeed, some studies have revealed that fear and anxiety levels during the COVID-19 pandemic in patients with FM were higher than in control patients (without FM) (Cankurtaran et al. 2021). In another study, 67% of 32 patients with FM reported that their general health status (well-being) worsened during the lockdown imposed by the COVID-19 pandemic (Cavalli et al. 2021; Cankurtaran et al. 2021). Consequently, these feelings seem to be associated with the severity of painful symptoms of FM (Aloush et al. 2021; Cankurtaran et al. 2021). The lack of social interactions may also play an essential role in modulating pain and dealing with chronic pain (Mogil 2015). Furthermore, patients with FM appear to be more susceptible to mood disorders than patients with other rheumatic diseases in the settings imposed by the COVID-19 pandemic (Iannuccelli et al. 2021), reinforcing the need to care for patients suffering from FM.
FM symptoms can be substantially managed using non-pharmacological measures (Sarzi-Puttini et al. 2020). Multidisciplinary interventions and physical exercise are strongly recommended to treat pain and improve the well-being of patients with FM (Busch et al. 2002; Sarzi-Puttini et al. 2020). However, one of the consequences caused by the pandemic was the reduction in daily activities related to the health of individuals, as many patients avoided leaving home to prevent SARS-CoV-2 infection (Clauw et al. 2020; Attal et al. 2021). Indeed, a study in Israel showed that all 129 patients with FM who used non-pharmacological treatments interrupted their activities after restrictions imposed by the COVID-19 pandemic, and over half of the patients with FM lost contact with their physician (Aloush et al. 2021). Another study showed that in patients with FM, the most commonly self-reported cause of exacerbating symptoms was the inability to exercise during restrictions (Cavalli et al. 2021).
Although we have been living with it for more than three years, the long-term effects of the pandemic on patients with FM are still unknown. The findings so far indicate that FM symptoms might be exacerbated during COVID-19, at least in a subset of patients (Salaf et al. 2021), and it can persist for months after recovery (Nalbandian et al. 2021). Furthermore, the numerous and persistent stressors imposed by the COVID-19 pandemic seem to aggravate FM disease (Aloush et al. 2021; Salaf et al. 2021; Cavalli et al. 2021). Based on this, both the viral infection and scenario of the COVID-19 pandemic may collectively exacerbate FM symptoms in the short and possibly long term.
Similar mechanisms between fibromyalgia syndrome and COVID‑19
FM and SARS-CoV-2 infection share mechanisms that could help us understand why both conditions culminate in the development of similar complaints. As these conditions are heterogeneous, and not all patients develop the same symptoms, multiple underlying mechanisms are likely to be implicated (Sarzi-Puttini et al. 2020; Scherlinger et al. 2021). Table 2 summarizes the similarities among the mechanisms of post-COVID and FM syndromes.
Accumulating evidence has clarified how the host immune response reacts to SAR-CoV-2 infection and contributes to infectious symptoms. The “cytokine storm” is an excessive or uncontrolled innate immune response to severe COVID-19 associated with untoward pathological consequences (Mahmudpour et al. 2020). However, immunological reactions might also occur in milder cases of COVID-19, aiming to combat viral infection. In this case, some COVID-19 clinical features, mainly painful and fatigue symptoms, are believed to be caused by an inflammatory response due to the invasion of the virus in the muscles and joints (Widyadharma et al. 2020). Indeed, SARS-CoV-2 cell-to-cell inflammatory mechanisms (i.e., cytokine storm) provoke hyperexcitability of PNS and CNS leading to the development of painful symptoms post-COVID or exacerbating pre-existing pain symptoms (Goodman et al. 2021; Fernández-de-lasPeñas et al. 2023). A similar immunological response also occurs in a subset of patients with FM. Although the cause of FM is unknown, and in some patients, it is not caused by a viral infection, dysregulated immune processes have been proposed to mediate FM symptoms (Goebel et al. 2021). In agreement, cytokines such as TNF-α, IL-6, and IL-8 are found in higher levels in the serum of patients with COVID-19 or FM (Littlejohn and Guymer 2018; Mahmudpour et al. 2020).

It is complicated to establish whether the immunological responses in the acute phase of COVID-19 evolve or are correlated with long-term sequelae (Moghimi et al. 2021). However, Huang et al. (2021b) have demonstrated that some pro-and anti-inflammatory cytokines and chemokines gradually decreased from symptoms onset to 12 months, as the symptoms of fatigue, weakness, and sleep problems were attenuated. This statement supports the critical role of the inflammatory process in developing symptoms in patients with COVID-19.
Saleh et al. (2020) have suggested inflammatory responses of SARS-CoV-2 may modulate mitochondria metabolism. Additionally, the opposite may happen. The mitochondrial metabolic manipulation by SARS-CoV-2 triggers an enhanced inflammatory response in patients with COVID-19 (Ajaz et al. 2021). Mitochondrial dysfunction has been implicated in numerous pathologies, including FM (Meeus et al. 2013) and SARS-CoV-2 infection (Saleh et al. 2020; Ajaz et al. 2021). In both diseases, there is impaired mitochondrial oxidative phosphorylation and production of ATP, which culminate in mitochondrial membrane permeabilization and mitophagy (Cordero et al. 2010; Saleh et al. 2020; Ajaz et al. 2021). Once patients with COVID-19 and FM cannot produce their required energy as in physiological conditions, increased glycolysis occurs to compensate for high energy demands (Eisinger et al. 1994; Moghimi et al. 2021). These impairments are associated with a higher inflammatory response and may lead to peripheral and central sensitization, explaining the pain and other systemic complaints in these patients (Meeus et al. 2013).
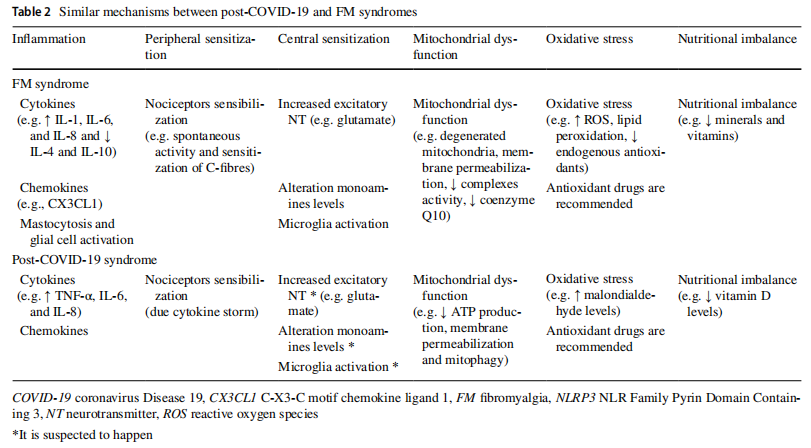
Mitochondria is the primary source of reactive oxygen species that contributes to normal cell function but is also linked to intracellular oxidative stress (Rinnerthaler et al. 2015). Besides, a heightened inflammatory state is associated with deleterious systemic events, including oxidative stress (Saleh et al. 2020). In this way, oxidative stress is indicated as an important player in the pathogenesis and severity of both COVID-19 (Saleh et al. 2020) and FM (Hung et al. 2020). Increased levels of malondialdehyde have been observed in patients with COVID-19 (Forcados et al. 2021) and FM (Meeus et al. 2013). Although literature data did not provide enough information on other oxidized substances in patients with COVID-19, antioxidant therapeutic strategies successfully reverted the severe symptoms of patients with COVID-19 (Cecchini and Cecchini 2020). Besides, the antioxidant therapeutic strategies also revert several symptoms of FM (Cordero et al. 2012a; Schweiger et al. 2020), suggesting that the oxidative status is implicated in the symptoms of both diseases.
Studies have shown significantly lower levels of vitamin D in patients with COVID-19, which can be correlated with disease progression (Forcados et al. 2021). Similarly, some patients with FM may have health problems associated with low levels of vitamin D (Bjørklund et al. 2018). It is important to emphasize that deficiency of vitamin D is related to the development of myopathy and severe muscle weakness, depression, and anxiety (Shipton and Shipton 2015), similar to the complaints of patients with COVID-19 and FM. In this way, therapy with vitamin D could improve the FM patient’s quality of life (Bjørklund et al. 2018), reduce the progression of COVID-19, and enhance the survival rate (Forcados et al. 2021). Thus, patients with COVID-19 could benefit from vitamin D supplementation.
Another mechanism shared by FM and COVID-19 is peripheral and central sensitization. More than one-third of patients with COVID-19 have experienced several neurologic manifestations(Josephson and Kamel 2020). Dorsal root ganglion (DRG) sensory neurons are a potential target for SARS-CoV-2 invasion, and viral infection of nociceptors may be responsible for causing some of the persistent neurological effects seen in COVID-19(Shiers et al. 2020). Besides, neuropilin 1,expressed in nociceptors, was recently reported as a host entry receptor for SARS-CoV-2 (CantutiCastelvetri et al. 2020). Similar to COVID-19, neuronal alterations also occur in some patients with FM, which are associated with abnormalities in peripheral sensory afferents fibers, triggering spontaneous activity and sensitization of C-fibers, and loss of epidermal innervation (Üçeyler et al. 2013; Evdokimov et al. 2019; Fasolino et al. 2020).
More recently, it was demonstrated that dysregulated immune processes produce autoreactive antibodies (e.g., IgG) that bind antigens expressed in the DRG of patients with FM and may cause an increased noxious peripheral input (Goebel et al. 2021). Similarly, immune dysregulation was also observed in patients with COVID-19. The presence of autoantibodies against cytokines and chemokines seems to perturb the immune function and impair virological control (Wang et al. 2021). Thus, the lack of host immunological control might be correlated with the occurrence of peripheral sensitization and the development and maintenance of painful symptoms in both diseases (Goebel et al. 2021; Komarof and Lipkin 2021).
Systemic inflammation leads to decreased monoamine levels and activation of microglia, resulting in increased levels of glutamate and N-methyl-D-aspartate receptor and excitotoxicity, which are suspected to happen in SARSCoV-2 systemic inflammation (Boldrini et al. 2021). It has been recently suggested that alterations of the dopamine and serotonin synthetic pathways might be involved in the pathophysiology of COVID-19, culminating in reduced levels of neurotransmitters (Nataf 2020). In this way, neuropsychiatric disorders that affect patients with COVID-19 could be explained, at least in part, by neurotransmission dysfunction or dysregulation (Boldrini et al. 2021). Quite similar processes also happen in FM, such as activation of glial cells (Littlejohn and Guymer 2018; Albrecht et al. 2019), increased excitatory neurotransmitters (glutamate, substance P, and others), and reduced inhibitory neurotransmitters (biogenic amines and gamma-aminobutyric acid (GABA)) (Sarzi-Puttini et al. 2020). Together, these mechanisms suggest the development of central sensitization in FM syndrome and COVID-19.
Patients with FM have enhanced pain-specific brain processing (named neurologic pain signature) (López-Solà et al. 2017) and brain glial activation (Albrecht et al. 2019), while decreased cortical and subcortical grey-matter density (Choy 2015). SARS-CoV-2 is known to penetrate the olfactory mucosa, causing loss of smell, and may enter the brain. However, dedefinitive evidence for SARS-CoV-2 penetrating the blood–brain barrier(BBB) is lacking. Inflammatoryoryory cytokines induce BBB instability, and once across the BBB, cytokines can activate glial cells (Boldrini et al. 2021). Given the propensity for cytokines (e.g., TNF-α and IL-6) and oxidative substances to contribute to peripheral and central neuronal sensitization, it was already hypothesized by my studies that the increased prevalence of neurological manifestations seen in severe cases of COVID-19 may be sociated with elevations of these markers (McFarland et al. 2021). Besides, stressors, whether physical(e.g., viral infection), mental, emotional, or financial, common stressors served in a pandemic situation), directly and negatively affect the underlying process of central sensitization, exacerbating the symptoms of FM and COVID-19 (Mohabbat al. 2020).
As described in detail, FM and COVID-19 share many similar mechanisms (Table 2). However, we strongly encourage further studies to elucidate better the analogy between the e development and maintenance of both diseases.
Could fbfibromyalgiarugs be used to treat post‑COVID symptoms?
More than three years after the start of the COVID-19 pandemic, it is time to recognize (i) the possibility of an FMFM-related syndrome in SARS-CoV-2-infected patients and (ii) the achievability of SARS-CoV-2 infection or pandemic situation (isolation, uncertainty, depression, mental stress, generalized anxiety, and fear of the virus) to exacerbate FM symptoms. Recognizing these approaches might encourage dressed treatments and strategies to alleviate the impact of COVID-19 on overall health. Nowadays, no standard protocols are available for treating post-COVID-19 symptoms. However, due to the similar symptoms between post-COVIDFMan d FM syndrome, as well as the frequent syndrome diagnosis, it is possible to hypothesize that dications already approved to treat FM syndrome may be positioned and efefficaciousor treating FM-related symptoms in post-COVID-19 patients.
The clinical treatments of FM are based only on symptom management and involve pharmacological or non-pharmacological approaches (Sarzi-Puttini et al. 2020). The first for the treatment of FM approved by the FDA are gabapentinoids (pregabalin) and SNRIs (duloxetine and milnacipran) (Clauw 2015; Sarzi-Puttini et al. 2020). Studies about the benefits of pregabalin in patients with COVID-19 have already been published (Oh et al. 2021; Pektas et al. 2021; Oddy et al. 2021; Jena et al. 2022). Jena al. (2022) demonstrated that pregabalin, associated or not with antidepressants, relieved pain symptoms in hospitalized patients with COVID-19. Besides, a case report scribed the effects of pregabalin use in two patients with COVID-19 and pneumonia who were admitted to the ICU with complaints of pain and cough. The patients received treatment with acetaminophen, tramadol, and pregabalin. Immediately after adding pregabalin, the patients related a significant reduction in cough complaints, chest pain, and myalgia. Furthermore, the patients began to tolerate more non-invasive mechanical ventilation and prone positions due to the absence of such complaints (Pektas et al. 2021). Evidence indicates that pregabalin use is not associated with creased in-hospital mortality among patients with COCOVID-19Oh et al. 2021; Oddy et al. 2021).
Additionally, pregabalin seemed to be an attractive therapeutic alternative to treat pain in patients during and after COVID-19. In addition to its pain-reducing efeffectspregabalin has sedative properties and can reduce anxiety and chronic cough (Vertigan et al. 2016; Bach et al. 2018; Slee al. 2019). Pregabalin is also effective in reducing pain and anxiety symptoms in FM patients (Clauw 2015; Salaf et al. 2021). Pregabalin also presents advantages over other medications, and it causes minimal respiratory depression, has a low drug-drug interaction potential, and has minor metabolism (Ben-Menachem 2004; Pektas et al. 2021), which is relevant to critical patients. Since the treatment with pregabalin had positive effects on cases with severe COVID-19, and this subset of patients is considered more susceptible to veloping post-COVID syndrome, we wonder: could early treatment with pregabalin or another gabapentinoid attenuate the progression of COVID-19 symptoms and, therefore, prevent the development of post-COVID syndrome?

In this sense, Aksan et al. (2020) published a case report of a 40-year-old woman diagnosed with COVID-19 with symptoms quite similar to the complaints reported by patients with FM, including constant pain, mechanical and thermal hypersensitivities, and sleep problems (Häuser et al. 2017). Acetaminophen, NSAIDs, and opioids, which are usually ineffective in relieving the pain of patients with FM, did alleviate this patient’s pain. On the other hand, gabapentin gradually reduced the pain, mechanical and thermal sensitivity, sleep impairment, and respiratory symptoms during hospitalization. One month later, the pain had eased slightly and continued to be relieved by gabapentin. It is worth noting that gabapentin is also a gabapentinoid, with a somewhat similar pharmacokinetic prprofileut re slowly and variably absorbed than pregabalin (Bockbrader et al. 2010). Additionally, gabapentin and pregabalin are safe treatment options with no significant interactions with treatments for COVID-19 (Plasencia-García et al. 2021).
Duloxetine, another drug approved by the FDA as a therapy for FM, decreased the incidence of COVID-19 in patients (Blanch-Rubió et al. 2020), and seems to be likely to interact with treatments recommended for COVID-19 (Plasencia-García et al. 2021). Furthermore, it is among the most commonly prescribed drugs annually in the United States, has an established safety prprofileand is generally well-tolerated (Fanelli et al. 2021). In contrast, milnacipran, another SNRI used as a first-line treatment for FM (Sarzi-Puttini et al. 2020), has not been evaluated in the COVID-19 setting. However, milnacipran may represent a good therapeutic alternative since it lacks interaction with the cytochrome P450 enzymes (frequently involved in the metabolism of antidepressants) (Fanelli et al. 2021). Therefore, milnacipran is less likely to interact with antiretroviral drugs used in patients with COVID-19 (Plasencia-García al. 2021).
Although tricyclic antidepressants, selective serotonin uptake inhibitors, and muscle relaxants are prescribed by clinicians and have strong clinical evidence (level I, A) for e in FM, they are not FDA-approved drugs for this purpose (Clauw 2014, 2015; Sarzi-Puttini et al. 2020). Some of these bugs have also been tested for repositioning for COVID-19. The antidepressants amitriptyline, imipramine, paroxetine, and sertraline have potential anti-viral activities and deserve studies targeting COVID-19, especially for those patients suffering from depression (Kutkat et al. 2022). Additionally, amitriptyline has the potential to benefit patients with postCOVID-19 headaches (Gonzalez-Martinez et al. 2022) and, when associated with pregabalin, relieves pain symptoms in hospitalized patients with COVID-19 (Jena et al. 2022). In the same way, the muscle relaxant tizanidine, used for ting myofascial pain disorders and FM (McLain 2000; Sarzi-Puttini et al. 2020), could be repositioned for treating post-COVID-19 symptoms (Kumar et al. 2021).
Nonetheless, there is a need to carefully evaluate the use of the therapeutic approaches commonly used in FM for treating some complaints of COVID-19 patients. A study demonstrates the contradictory effects of pregabalin, including an increased incidence of COVID-19 in patients who it is (Blanch-Rubió et al. 2020). Besides, serotonin syndrome (i.e., a serotoninergic overactivity at synapses of the CNS and PNS) has to be considered when using drugs metabolized by cytochrome P450 enzymes, such as duloxetine, in patients with COVID-19 on antiretroviral therapy with lopinavir-ritonavir (Plasencia-García et al. 2021). However, serotoninergic syndrome seems to disappear when duloxetine is stopped (Sabe et al. 2021). Therefore, until now, the literature data have not provided enough backing to confirm whether patients with COVID-19 will benefit or be harmed ing gabapentinoids or SNRIs during the infection. The hypotheses must be considered, and double-blinded, controlled, and randomized clinical trials of these drugs in patients with COVID-19 and post-COVID are needed to better understand the benefits of this disease.
Non-pharmacological interventions used for FM treatment, such as psychotherapy, physical exercise, and acupuncture, should be considered for patients with COVID-19 syndrome. Studies have demonstrated that ercise therapy might be a safe and effective relief for post-COVID-19 syndrome (Hernando-Garijo et al. 2021; Dotan al. 2022). Indeed, a telerehabilitation program based on aerobic exercise relieved the painful symptoms and psychological distress in women with FM during the COVID-19 pandemic (Hernando-Garijo et al. 2021). Although it may be laborious to transpose non-pharmacological FM treatments to another st-COVID syndrome, telerehabilitation, neuromodulation, and resistance exercises have improved primary complaints in post-viral syndromes (Perez et al. 2021; Chandan al. 2022; Gentil et al. 2022).
Pharmacotherapies to Inflammation Treatment
Although therapies focusing on inflammation are not recommended for treating FM, we cannot disregard their prominent use in the post-COVID syndrome, given all the solid inflammatory mechanisms triggered by the infection. The immune responses against SARS-CoV-2 are one of the main features of disease pathogenesis (van de Veerdonk et al. 2022), leading to the recruitment and activation of leukocyte subsets and the release of inflammatory cytokines (such as IL-6, IL-2, IL-8, interferon (IFN)-γ, IFN-α, and TNF-α), featuring the “cytokine storm” in the COVID-19 (Mahmudpour et al. 2020). Additionally, the IL-1–IL-6 axis represents one of the most important pathways in the SARSCoV-2-induced hyperinflammatory reaction (Chen et al. 2020a, b; Giamarellos-Bourboulis et al. 2020). All these mechanisms contribute to an ininflammatoryesponse that is sociated with different clinical features and symptoms of COVID-19 (Chang et al. 2021; Huang et al. 2021a), such painful symptoms since increased cytokines can induce hyperalgesia and allodynia and, therefore, sustain widespread chronic pain (Ji et al. 2018; Goodman et al. 2021). Furthermore, post-COVID-19 painful symptoms may be used by an inflammatory response due to the invasion of the virus in the neurons, muscles, and joints (Shiers et al. 2020; Widyadharma et al. 2020). Thus, preventing viral progression or limiting the cytokine storm could help reestablish health, eliminate or alleviate pain, and assuage the impact of COVID on patients.

In this sense, several drugs with immunomodulatory properties have been strongly recommended by the panel for COVID-19 treatments, which vary according to the disease verification (mild, severe, or critical) (Lamontagne et al. 2020). These drugs include systemic corticosteroids (dexamethasone), IL-6 inhibitors (tocilizumab and sarilumab), and jaJanus nase inhibitors (baricitinib, tofacitinib, and ruxolitinib) (Lamontagne et al. 2020).
Corticosteroids are among the drugs recommended to nage patients with COVID-19 and are widely used for eating other diseases closely related to COCOVID-19such as Severe Acute Respiratory Syndrome and Middle Eastern Respiratory Syndrome (Caiazzo et al. 2022). This drug class habits many inflammation-associated molecules, such as cytokines(IL-6, IFN-γ, and TNF-α), chemokines, arachidonic acid metabolites, and adhesion molecules, besides-regulating anti-inflammatory mediators (Barnes 2011). Corticosteroids are recommended for severe or critical COVID-19 to counteract excessive inflammation, not non-severe patients (Lamontagne et al. 2020). Indeed, clinical large-scale randomized trials have proposed that corticosteroids contribute to the recovery of severe or critically ill patients infected with SARS-CoV-2 (Sterne et al. 2020; RECOVERY 2021; Cano et al. 2021). Its long-term use of light helps control post-disease complications, especially in patients with persistent respiratory distress (Kostorz-Nosal al. 2021). However, the long-term corticosteroid treatment must be carefully evaluated as it might contribute to adverse effects involving the gastrointestinal, cardiovascular, endocrine, nervous, ocular, and immune systems (Oray et al. 2016).
The IL-6 released by macrophages is one of the key factors in initiating cytokine storms, and its increase is associated with the severity of COVID-19 (Chen et al. 2020a; McGonagle et al. 2020; Huang et al. 2021b). Thus, it is suggested that blocking the action of IL-6 may alter the course of the disease and be beneficial to ill patients (Zhang et al. 2022). Tocilizumab and sarilumab are monoclonal antibodies that act as immunomodulatory, directly targeting IL-6 via the IL-6 receptor (Xu et al. 2021). Both are approved by the FDA for rheumatoid arthritis treatment and have recently been approved in the United States and United Kingdom for use in patients with severe COVID-19 (Department of Health and NHS 2021; Salama al. 2021). Although the effectiveness of IL-6 inhibitors on the SARS-CoV-2-triggered immune response has en proven (Abani et al. 2021; Godolphin et al. 2022), its age as a sole drug and the optimal timing to introduce it to the patient treatment regimen remains controversial (Huang and Jordan 2020; Abidi et al. 2022).
The IL-6 is one of the primary activators of jaJanus nase (JAK)/signal transducer and activator of the transcription (STAT) pathway (Kamimura et al. 2003). The activation of JAK/STAT pathways orchestrates important biological processes, including cell proliferation, and immunomodulation (Xin et al. 2020). By exerting effects controlling immune regulation and inflammation of the cells, and reducing ininflammatoryytokines, some JAK inhibitors have been approved FDA, such as baricitinib and tofacitinib for treating rheumatoid arthritis (Harrington et al. 2020) and ruxolitinib for treating hahematologicaliseases (Kirito 2022). In is sense, many clinical trials have researched the role of JAK inhibitors in COVID-19 treatment (Cao et al. 2020; Bronte et al. 2020; Kalil et al. 2021; Marconi et al. 2021; Ely et al. 2022). The effectiveness of JAK inhibitors in critically ill patients with COVID-19 has been proven (Cao al. 2020; Bronte et al. 2020). However, inconsistent conclusions about efficiency for the reduction of COVID-19 mortality were reported (Kalil et al. 2021; Marconi et al. 2021; Ely et al. 2022).
In addition to the drugs listed above, several drug classes with anti-inflammatory properties have been tested to manage COVID-19, which include colchicine (Tardif et al. 2021; Horby et al. 2021), inhaled corticosteroids (budesonide and ciclesonide) (Yu et al. 2021; Clemency et al. 2022), IL-1 inhibitor (anakinra) (Cavalli et al. 2020; Huet et al. 2020), Bruton's tyrosine kinase (BTK) inhibitors (acalabrutinib and ibrutinib) (Roschewski et al. 2020; Rada et al. 2021) and NSAIDs (indomethacin and aspirin) (Perico et al. 2022). However, it is essential to point out that all of these drugs are currently under investigation in clinical trials and the treatment regimen remains controversial. Until now, they were not recommended by the panel for COVID-19 treatments or still need to be approved by the FDA.
Finally, it is worth noting that pharmacotherapies recommended by the panel for COVID-19 treatments need to be evaluated, especially for their use on painful parameters in post-COVID syndrome. More medical evidence is still required to demonstrate their rational application, including dosage, course of treatment, the timing of administration, and combination with other drugs. Future clinical studies are also relevant to investigate whether these therapies effectively treat FM-related symptoms in post-COVID patients. Moreover, clinicians should investigate whether these therapies could also reduce COVID-19-caused intense inflammation and, consequently, improve patients' general health.
【For more info:george.deng@wecistanche.com / WhatsApp:8613632399501】






