Estimating The Causal Effect Of Treatment With Direct-acting Antivirals On Kidney Function Among Individuals With Hepatitis C Virus Infection
Dec 21, 2023
Abstract
Background Direct-acting antivirals (DAA) are highly effective at treating Hepatitis C virus (HCV) infection, with a cure rate >95%. However, the effect of DAAs on kidney function remains debated.
Methods We analyzed electronic health record data for DAA-naive patients with chronic HCV infection engaged in HCV care at Boston Medical Center between 2014 and 2018. We compared the following hypothetical interventions using causal inference methods: 1) initiation of DAA and 2) no DAA initiation. For patients with normal kidney function at baseline (eGFR>90 ml/ min/1.73m2 ), we estimated and compared the risk for reaching Stage 3 chronic kidney disease (CKD) (eGFR-60 ml/min/1.73m2 ) under each intervention. For patients with baseline CKD Stages 2–4 (15<eGFR-90 ml/min/1.73m2 ), we estimated and compared the mean change in eGFR at 2 years after baseline under each intervention. We used the parametric g-formula to adjust our estimates for baseline and time-varying confounders.
Results First, among 1390 patients with normal kidney function at baseline the estimated 2-year risk difference (95% CI) of reaching Stage 3 CKD for DAA initiation versus no DAA was -1% (-3, 2). Second, among 733 patients with CKD, Stage 2–4 at baseline the estimated 2-year mean difference in change in eGFR for DAA initiation versus no DAA therapy was -3 ml/min/ 1.73m2 (-8, 2).
Conclusions We found no effect of DAA initiation on kidney function, independent of baseline renal status. This suggests that DAAs may not be nephrotoxic; furthermore, in the short term, HCV clearance may not improve CKD.

GET NATURAL ORGANIC CISTANCHE EXTRACT WITH 25% ECHINACOSIDE AND 9% ACTEOSIDE FOR KIDNEY FUNCTION
For more info please contact us at:
Email:wallence.suen@wecistanche.com
Tel/Whatsapp: +86 15292862950
Introduction
Hepatitis C virus (HCV) infection is a major cause of morbidity and mortality throughout the world and has important extrahepatic sequelae [1–3]. In addition to diabetes and cardiovascular disease [4], patients with HCV infection are at increased risk for chronic kidney disease (CKD) and end-stage renal disease (ESRD) compared with the general population, possibly due to systematic inflammation and vascular damage [5–7].
Treatment with direct-acting antivirals (DAA) is highly effective at clearing HCV: approximately 95% of patients treated with DAAs achieve a sustained virological response (SVR) equivalent to cure [8–11]. Clinical trials have shown that an SVR following treatment with DAAs is associated with liver function improvements for patients with and without CKD [12– 14]. Furthermore, treatment with DAAs has been associated with a reduction in both the incidence and severity of extrahepatic manifestations of HCV infection such as diabetes and cardiovascular disease [15]. However, the effect of treatment with DAAs on kidney function remains ambiguous and needs to be further investigated.
Clinical trials that led to DAA approval generally had relatively short follow-up duration and did not estimate the mid-and long-term effect of DAA treatment on kidney function. Recent observational studies using real-world data comparing changes in estimated glomerular filtration rate (eGFR) before and after DAA treatment provide inconsistent results. Some studies observed a stabilization of or an increase in eGFR, while one demonstrated a decline in eGFR, specifically among DAA-treated individuals with normal baseline kidney function [10, 16–20]. However, current HCV guidelines do not recommend routine, frequent renal monitoring after DAA treatment in individuals with normal kidney function [21]. Therefore, studies that compare the pre-and post-DAA rate of eGFR decline could be biased by the frequency of and cause for creatinine measurements. In addition, while clinical trials of DAA therapies did not indicate renal toxicity, there have been concerns about kidney adverse effects, especially in connection with sofosbuvir, which is mainly cleared really [20, 22, 23,].
Target trial emulation is a novel approach to the design of comparative effectiveness studies [24]. With this method, the study design and statistical analysis of an observational study closely emulate the protocol components of a target trial, i.e., the ideal trial a researcher would conduct to answer the research question. We have, therefore, used the target trial approach to estimate the effect of DAA treatment on kidney function by comparing two hypothetical interventions: if all patients had initiated DAA treatment versus if all patients had not initiated DAA treatment.

Methods Study data
We extracted de-identified electronic health record (EHR) data from the Boston Medical Center Clinical Data Warehouse on all patients with a record of chronic HCV infection between 2014 and 2018. Boston Medical Center is the largest safety net hospital in New England; it serves as a leading primary care provider for Boston's vulnerable populations, including minority and low-income patients; and it has a large HCV testing and screening program [25, 26]. We collected information on inpatient, outpatient, and emergency room visits; laboratory measures; prescriptions; active problem lists (i.e., clinician-compiled list of a patient's health diagnoses); procedures; insurance status; vital status; and demographic characteristics. Chronic HCV infection was defined as having an HCV-RNA result above the laboratory level of detection or a positive HCV genotype result. We used the CKD-EPI algorithm to calculate eGFR [27, 28]. The Boston University Medical Campus Institutional Review Board approved this study. The need for participant consent was waived by the committee because the data was de-identified.
Target trial emulation
In our real-world data, individuals with CKD or impaired kidney function tended to have more frequent renal monitoring than those with normal kidney function (S1 and S2 Figs). The differential monitoring and the potential bias due to it were important considerations. Additionally, the interpretation of changes in eGFR differs for individuals with and without CKD. Therefore, we designed two separate target trials. In Trial 1 we included patients with normal baseline kidney function, defined as eGFR>90 ml/min/1.73m2. In Trial 2 we included patients with CKD Stage 2–4 at baseline, defined as eGFR between 15 and 90 ml/min/1.73m2. The target trials were identical in all components except for the baseline eGFR inclusion criteria and the outcome definition. We then designed two cohort studies to emulate the protocols of each target trial. S1 and S2 Tables detail the target trials and trial emulations.
Eligibility criteria
Eligible individuals met the following criteria between January 2014 and December 2018: age �18 years, chronic HCV infection as described above, no history of kidney transplant or ESRD (defined as CKD Stage 5 or on dialysis), and the presence of serum creatinine (to compute the baseline eGFR), transaminase, and the platelet measurements within three months of each other (to calculate a Fibrosis-4 [FIB-4] score). In addition, to capture the patients actively engaged in HCV care, we restricted eligibility to individuals with an outpatient visit for HCV infection within three months of their identified HCV infection in the EHR. Individuals who met these eligibility criteria were split into Cohort 1 and Cohort 2 according to their baseline eGFR and they were analyzed separately. A flow chart of these samples is presented in Fig 1.

Interventions
For both cohorts, we compared the following two DAA initiation strategies: 1) initiation of DAA within 3 months of baseline and 2) no DAA initiation. S3 Table details the DAA therapies utilized.
Outcome The outcome definitions differed by cohort. The outcome for cohort 1 (individuals with normal kidney function) was the development of Stage 3 CKD, defined as the first recorded eGFR�60 ml/min/1.73m2.
The outcome for cohort 2 (individuals with baseline CKD Stage 2–4) was changed from baseline eGFR to the last recorded eGFR between 24- and 30 months post-baseline.
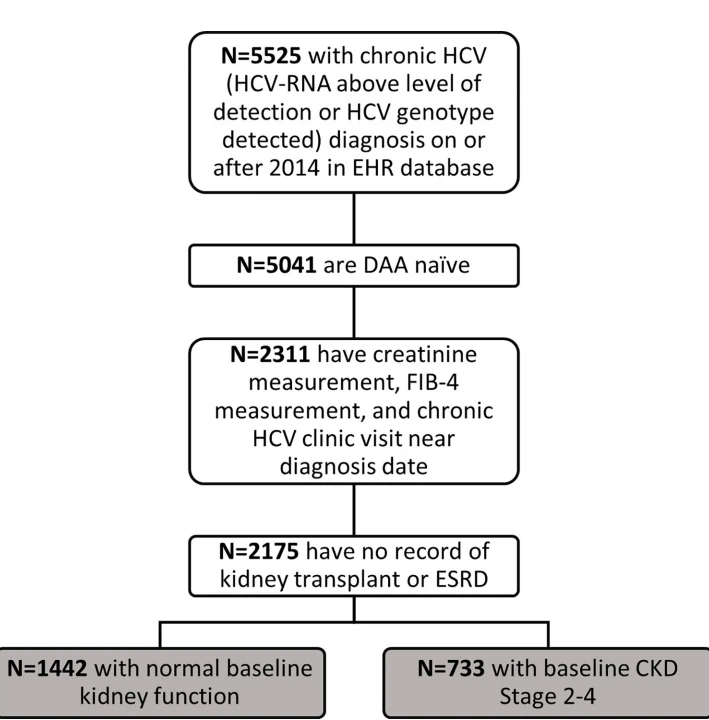
Fig 1. Flow chart of study participants.
Follow-up For both cohorts, follow-up started at baseline, defined as the earliest date all eligibility criteria were met, and ended at the earliest of one of the following events: death, kidney transplant, ESRD (defined as CKD Stage 5 or on dialysis), 2 years post-baseline, administrative end of follow-up (December 2018), or, in Trial 1, date of outcome. A summary of the study design is presented in Fig 2 [29].
Statistical analyses
We summarized the baseline characteristics of each cohort separately using descriptive statistics.
For cohort 1, we used a time-to-event framework to estimate and compare the 2-year cumulative incidence of reaching Stage 3 CKD under the two DAA initiation strategies. For cohort 2, we estimated and compared the mean 2-year change from baseline eGFR under each intervention. Because standard statistical methods cannot appropriately adjust for time-varying confounding, we used the parametric g-formula to generate adjusted estimates [30–32]. Covariates were chosen a priori based on the co-authors' clinical judgment and review of the published literature.

Fig 2. Description of cohort entry timeline. End-stage renal disease (ESRD) includes dialysis and Stage 5 chronic kidney disease (CKD). b Baseline covariates: Fibroscan, genotype, body mass index, insurance type, alanine transaminase, aspartate transaminase, platelets, HIV, drug or alcohol use disorder, mental illness, diabetes, hypertension. c Time-varying covariates: alanine transaminase, aspartate transaminase, platelets, change in eGFR, diagnosis of drug or alcohol use disorder, mental illness, hypertension, clinic visits, treatment with DAAs. d Follow-up window ends at the earliest of one of the following events: death, kidney transplant, ESRD, 104 weeks (2 years) post-baseline, administrative end of follow-up (December 2018), or, in Trial 1, date of outcome.

Like all causal inference methods, the parametric g-formula estimates the causal effect of a hypothetical intervention (here, the DAA therapy initiation) under the identifiability assumptions (positivity, exchangeability, and consistency) and assumes a correct model specification [30]. The parametric g-formula algorithm has been described in detail elsewhere [33–35]. Briefly, it works in two steps: 1) parametric regression models estimate the joint distribution of the outcome, treatment, and time-varying covariates conditional on previous treatment and covariate history and 2) the estimates in step 1 are used to simulate the distribution of the outcome under each intervention using the Monte Carlo method.
In the first step, continuous and binary time-varying covariates were modeled with separate linear and logistic regression models, respectively. All models adjusted for baseline covariates and the most recent lagged value of time-varying covariates. Baseline covariates included age, sex, race/ethnicity (Hispanic or Latino, White, Black, other), body mass index, education level, HCV genotype, insurance type, degree of liver fibrosis (by fibroscan), HIV coinfection, and diabetes. Time-varying covariates were alanine transaminase (ALT), aspartate transaminase (AST), platelet count, change from baseline eGFR, drug or alcohol use disorder [36], mental illness, outpatient visits, and hypertension. We also treated the timing of the laboratory measurements (AST, ALT, platelet count, eGFR) as potential confounders: we fit logistic regression models for these visit processes with a binary outcome of 1 if a measurement was taken at that time and 0 if not. For cohort 1, continuous eGFR was included as an additional baseline covariate, and for cohort 2 CKD stage was included as an additional baseline covariate. Skewed continuous covariates (ALT and AST) were log-transformed for normality. Details on the definition and ascertainment of covariates are presented in S4 Table.
In the second step, we used the parameter estimates from the first step to simulate the post-baseline time-varying covariates and the outcome separately under each of the two interventions. In our real-world data, eGFR is measured sporadically and long periods can elapse between measurements. Therefore, we imposed an eGFR measurement after 12 months in individuals without a new measurement.
We used nonparametric bootstrapping with 500 samples to obtain 95% confidence intervals (CI) around our estimates based on percentiles.
To explore the validity of our parametric assumptions, we compared the observed outcome means and time-varying covariates with those predicted by our models (S3 and S4 Figs).
All analyses were conducted in SAS version 9.4 using the publicly available GFORMULA macro [37].
Subgroup analyses
As secondary analyses, we reran the estimation procedure in the following patient subgroups with elevated risk of CKD [38]: those with 1) type II Diabetes, 2) hypertension, and 3) age>45 years. The cut-off for age was chosen according to the bimodality of the distribution of age in the study sample (S5 Fig).
Sensitivity analyses
We conducted several sensitivity analyses to assess the robustness of our results. First, we reran the analysis for both cohort 1 and cohort 2 excluding HCV and HIV-coinfected patients [39]. Second, we reran the analysis for both cohorts restricting them to outpatient eGFR measurements only. This was done to attempt to exclude acute renal injuries. Third, we reran the analysis for cohort 1 with the follow-up extended to the database close (2018), and we reran the analysis for cohort 2 with the follow-up extended to 3 years. Fourth, we conducted the analyses with time discretized into months, instead of weeks as was done in the main analysis. Fifth, we ran a series of analyses reordering the time-varying covariates, since each parametric model conditions on past covariates. Sixth, we reran the analyses by 1) imposing an eGFR measurement after 6 months, instead of 12 months, and 2) not imposing a new eGFR measurement. In addition, for cohort 1, we ran a sensitivity analysis defining Stage 3 CKD as the first of two eGFR measurements <60 ml/min/1.73m2 to be consistent with the official CKD diagnosis guidelines [23]. We also reran the primary analysis additionally adjusting for concomitant use of nephrotoxic medications (nonsteroidal anti-inflammatory drugs, diuretics, and angiotensin-converting enzyme inhibitors or angiotensin receptor blockers). Finally, we reran the primary analyses to estimate the effect of treatment with Sofosbuvir-containing regimens on renal function. We did this by censoring patients treated with non-Sofosbuvir-containing regimens at their treatment start date.