Gossypitrin, A Naturally Occurring Flavonoid, Attenuates Iron-Induced Neuronal And Mitochondrial Damage Part 1
Mar 16, 2022
Please contact oscar.xiao@wecistanche.com for more information
Abstract: The disruption of iron homeostasis is an important factor in the loss of mitochondrial function in neural cells, leading to neurodegeneration. Here, we assessed the protective action of gossypitrin(Gos), a naturally occurring flavonoid, on iron-induced neuronal cell damage using mouse hippocampal HT-22 cells and mitochondria isolated from rat brains. Gos was able to rescue HT22 cells from the damage induced by 100 uM Fe()-citrate (EC50 8.6 uM). This protection was linked to the prevention of both iron-induced mitochondrial membrane potential dissipation and ATP depletion. In isolated mitochondria, Gos (50uM) elicited almost complete protection against iron-induced mitochondrial swelling, the loss of mitochondrial transmembrane potential, and ATP depletion. Gos also prevented Fe(Ⅱ)-citrate-induced mitochondrial lipid peroxidation with an ICs0 value (12.45 μM) that was about nine times lower than that for the tert-butyl hydroperoxide-induced oxidation. Furthermore, the flavonoid was effective in inhibiting the degradation of both 15 and 1.5 mM 2-deoxyribose. It also decreased Fe(II) concentration with time, while increasing O2 consumption rate, and impairing the reduction of Fe(II) by ascorbate. Gos-Fe(I) complexes were detected by UV-VIS and IR spectroscopies, with an apparent Gos-iron stoichiometry of 2:1. Results suggest that Gos does not generally act as a classical antioxidant, but it directly affects iron, by maintaining it in its ferric form after stimulating Fe(II) oxidation. Metal ions would therefore be unable to participate in a Fenton-type reaction and the lipid peroxidation propagation phase. Hence, Gos could be used to treat neuronal diseases associated with iron-induced oxidative stress and mitochondrial damage.

Please click here to learn more
Keywords: gossypitrin; iron; HT-22 cells; mitochondria; neurodegeneration; neuroprotection
1. Introduction
Iron is an essential cofactor in many crucial biological processes such as DNA synthesis and restoration, oxygen transport, cellular respiration, xenobiotic metabolism, and hormone synthesis [1. However, at high intracellular levels, it can increase cellular reactive oxygen species with a modification in the cellular redox potentials [2,3]. Iron dyshomeostasis is observed in almost every neurological condition such as Parkinson's disease (PD), Alzheimer's disease(AD), Huntington's disease(HD), multiple sclerosis, Friedreich's ataxia, epilepsy, restless legs syndrome, and stroke [4,5]. Particularly, in intracerebral hemorrhage, where there is a rupture of the microvasculatures and the blood released produces a blood-derived iron exposure of neural cells, the main mechanisms of neurodegeneration are iron- and heme-induced ROS production, inflammation, and excitotoxicity [6,7]. In vitro and in vivo experimental models of brain hemorrhage have shown the presence of ferroptosis, an iron-dependent form of regulated necrosis [8]. Furthermore, increasing evidence suggests that the disruption of iron homeostasis is essential in the loss of mitochondrial function in neural cells [9,10], which could, in turn, lead to neurodegeneration [11-14. A relationship was recently reported between the early-onset form of Parkinson's disease with iron metabolism dysregulation, and mitochondrial impairment [15]. The co-existence of relatively high amounts of both iron and ROS in the mitochondrial compartments makes this organelle susceptible to hydroxyl radical-mediated damage. Pharmacological agents targeting brain iron regulation and iron-mediated mitochondrial impairment may therefore be therapeutically useful against neurodegeneration. Several iron-chelating agents have been found to exhibit neuroprotective actions in different experimental models [16-19]. On the other hand, there have been many reports on the neuroprotective potential of naturally occurring polyphenols due to their antioxidant actions based on a combination of iron chelation and free radical scavenging activities [20,21]. Our group has observed that catechol-containing polyphenols like mangiferin, guttiferone-A, and rapanone interact with iron, both in vitro and in vivo, thus annulling its catalytic role in promoting ROS [22-28]. We also recently reported the potent antioxidant effects of gossypitrin(Gos), a naturally occurring flavonoid(Figure 1), indicating its potential use as a neuroprotective agent [29]. Nevertheless, little is known of the mechanisms involved in these antioxidant effects. The presence of a planar six-member cyclic system with electron delocalization, a catechol moiety, and three more aromatic hydroxyl groups(two of them adjacent to a carbonyl group), strongly suggest that this molecule could act as an iron-chelating agent since these are structural features that favor iron chelation by flavonoids 30]. Hence, the purpose of this study is to evaluate the ability of Gos to protect HT-22 neuronal cells and isolated rat brain mitochondria from iron-mediated oxidative damage. Other studies in cell/mitochondria-free systems were carried out to further characterize the nature of the Gos-iron interaction.

2. Results
2.1. Gos Rescues HT-22 Cells from Fe(I)-Induced Cell Death
Fe(II)(100 μM) reduced HT-22 cell survival to 17.4% after 24 h of exposure, as estimated by the MTT assay. Gos prevented hippocampal cell death in a dose-dependent manner. The EC50 was 8.64± 1.58 μM(Figure 2A, inset). Full cell protection was observed with 100 μM Gos was more effective than Trolox(500 uM), the well-known hydrosoluble antioxidant, which prevented cell death by 74%.
HT-22 exposure to iron for 2 h (prior to cell death execution) induced the dissipation of the mitochondrial membrane potential, as estimated by the retention of rhodamine 123 within the organelles(Figure 2B), and ATP depletion (Figure 2C). This suggests a possible association between the toxic iron-mediated effects on the mitochondria and cell viability. Gos prevented the iron-induced dose-dependent impairment of the mitochondria, following the patterns of neuron viability which showed that it may prevent iron-induced HT-22 cell death through mitochondrial protection.

Figure 2.Gos protection against iron-induced neuronal cell damage.Effects of Gos on HT-22 mouse hippocampal neuron survival (A), cellular mitochondrial membrane potential (B), and cellular ATP levels(C). Gos(0-100 μM) or Trolox(500 μM) were co-incubated for 24 h (viability) or 2h (mitochondrial membrane potential and ATP levels) with 100 μM FeCl2/1 mM citrate. The assay conditions are described in Section 4. Damaged control cells(without Gos) contained DMSO (0.001%)plus the iron-citrate mixture. The results are expressed as the percentage of cell death in relation to the undamaged control(containing DMSO0.001%as the vehicle). Values are the means ± S.D. of three different experiments. Different symbols indicate statistical differences at p<0.05 when comparing all pairs of experimental groups according to the ANOVA and post hoc Newman-Keuls tests. (If the symbols are different among groups indicate statistical differences, when they are equal, there is not).

Cistanche can improve immunity
2.2. Gos Rescues Rat-Brain Mitochondria from Iron-Induced Mitochondrial Lipoperoxidation Brain mitochondria incubated with Fe()-citrate (50 μM) underwent a marked lipoperoxidation expressed by an increase in the formation of the thiobarbituric acid reactive substance(Figure 3A). Gos inhibited the iron-mediated mitochondrial lipid peroxidation in a concentration-dependent manner (IC50 value of 12.45±1.24 μM) (Figure 3A, inset). The effects of Gos on tert-butyl hydroperoxide(300 μM)-induced mitochondrial lipid peroxidation(IC50 value>100 μM) shows that it is more effective in preventing iron-induced lipid peroxidation than peroxyl induced lipid peroxidation, suggesting its ability to act directly on iron (Figure 3B).

Figure 3. Gos inhibits malondialdehyde formation in the mitochondria, induced by 50 uM Fe()-citrate(A)or 300μM tert-butyl hydroperoxide(B). Ratbrain mitochondria(1 mg/mL)were incubated in a reaction medium containing 125 mM sucrose,65 mM KCl,10mM HEPES buffer(pH 7.4),2 mM succinate, and 2.5 uM rotenone, with or without Gos(1-50 uM). The experiments started by the addition of 50μM Fe(II) or 300 μM tert-butyl hydroperoxide, except for the undamaged Control(gray bars). The incubation period was 20 min at 28℃C. Values are the means ±S.D. (n=6). Different symbols indicate statistical differences at p<0.05 when comparing all experimental groups according to the ANOVA and posthoc Newman-Keuls tests. (If the symbols are different among groups indicate statistical differences, when they are equal, there is not).
2.3.Gos Prevents Iron-Mediated Mitochondrial Swelling Dissipation, and ATP Depletion

Figure 4A shows that 50μM Fe(I)-citrate complex induced mitochondrial swelling as expressed by a decrease in the absorbance at 540nm(line f versus line a). It was associated with a spontaneous and complete △Y depolarization (Figure 4B, linef versus line a), and ATP depletion (Figure 4C, Fe()/citrate column). Gos inhibited the iron-induced swelling process and AY dissipation in a concentration-dependent manner (Figure 4A,B lines b-e). The absorbance values (mean ± SD at 7 min) from Panel A were: 0.482±0.013 (line a), 0.459± 0.011 (line b),0.445± 0.007 (line c),0.435± 0.015(line d), 0.360±0.011(line e), and 0.338±0.012(line f). The fluorescence values from Panel B(mean ± SD at 7 min)were 15.03±1.71(line a),19.41±2.07 (line b),22.29±2.58 (line c),45.505±4.47(line d), 118.468±5.76 (line e), and 146.201±5.83 (line f).Statistically significant differences were found between line f and the other lines, at p<0.05. The naturally occurring flavonoids also prevented iron-mediated ATP depletion in a concentration-dependent manner(Figure 4C). A strong interaction of Gos with iron is suggested as the key mechanism for neuroprotection against iron-induced damage, both in HT-22 neuronal cells and in brain mitochondria. To further characterize this Gos-iron interaction, different experiments on a cell/mitochondria-free system were performed.

Figure 4. (A) Mitochondrial permeability transition induction, (B)mitochondrial membrane potential (AH)loss, and (C) reduction of ATP levels induced by 50 μM Fe(II)-citrate. A was estimated using the fluorescence probe safranine (5 uM). The inhibitory effects of Gos 50,25, 10, and 1μM are shown in lines (b),(c), (d), and (e), respectively. Lines (a) and(f)represent undamaged controls (without iron), and damaged controls (without Gos), respectively. Rat brain mitochondria (RBM-0.5 mg/mL), Fe(II) and1 μM CCCP were added where indicated by the arrows. Different symbols in the graph(C)indicate statistical differences atp<0.05 when comparing all pairs of experimental groups according to the ANOVA and post hoc Newman-Keuls tests.(If the symbols are different among groups indicate statistical differences, when they are equal, there is not).

2.4. Gos Induces Fe(II)-Citrate Autoxidation and O, Consumption
Gos decreased Fe() concentration (1-min preincubation) in a reaction medium containing 2 mM citrate (without mitochondria) in a dose-dependent manner (Figure 5A, black circles).A 5-min preincubation period (Figure5A, red boxes)decreased Fe(II)concentration even more. Gos also increased the rate of O2 consumption, possibly due to oxidation of Fe(ll) to Fe(ⅡII)(Figure 5B,C). These results suggest that Gos could be removing Fe(II) from the citrate complex and oxidizing it to the ferric form in a process that requires O2 as the electron acceptor. Accordingly, Gos could be hindering the formation of "OH radicals by inactivating the Fe(Il) necessary for the Fenton-Haber-Weiss-type reaction, which can initiate the lipid peroxidation of the mitochondrial membrane.
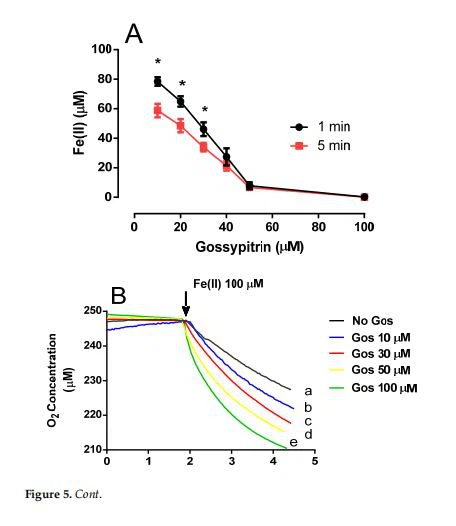
Figure 5. (A)Gos stimulates Fe(Ⅲl) autoxidation in the absence of rat brain mitochondria in a standard medium supplemented with 1 mM citrate, and 5 mM1,10-phenanthroline.1,10-phenanthroline was added 1 or 5 min after the Gos-Fe(II) incubation period, and the absorbances were read at 510nm. Values are the average of three determinations. * statistical difference at p<0.05 when comparing 1 vs. 5min incubation. (B)Effects of Gos on O, consumption mediated Fe(Il) autoxidation in a standard medium supplemented with 1 mM citrate under the following conditions:(a) No Gos,(b)10 uM Gos, (c) 30μM Gos, (d)50 μM Gos,and (e)100μM Gos. Fe(II) (50μM) was added where indicated by the arrow. Results are representative of three experiments. (C) Oxygen consumption rate (nmol O2/min/mL)1 min after Fe() addition.Legends are the same as in(B). Different symbols indicate statistical differences at p<0.05 when comparing all pairs of experimental groups (in the same cell type) according to the ANOVA and post hoc Newman-Keuls tests. (If the symbols are different among groups indicate statistical differences, when they are equal, there is not).
This article is extracted from Molecules 2021, 26, 3364. https://doi.org/10.3390/molecules26113364 https://www.mdpi.com/journal/molecules






