How To Assess Kidney Function in Outpatient Clinics
Mar 03, 2022
Contact: emily.li@wecistanche.com
P. E. Korhonen1,2,3
SUMMARY
Background
In 2002, a new definition and classification of chronic kidney disease were published, and glomerular filtration rate < 60 ml/min/1.73 m2 for 3 months or more was adapted to define chronic kidney disease irrespective of other signs of kidney damage. Aims: To discuss different ways to assess kidney function in outpatient clinics and especially in primary care. Methods: The Pub-Med database was searched for relevant articles. Results: The estimated glomerular filtration rate equations which take into account plasma creatinine, age, sex, race, and body size have been developed to identify patients with chronic kidney disease formerly overlooked if the renal function had been assessed by plasma creatinine alone. Cystatin C-based equations have also been developed to enhance accuracy for individuals with whom creatinine-based estimates for kidney function are acknowledged to be less accurate. Discussion: The characteristics of the patients to whom the diagnostic test is applied can influence the sensitivity of the test. Thus, there is nowadays controversy over the best method to assess kidney function in the general population. Conclusion: In the overwhelming majority of patients currently treated in primary care, the CKD-EPI creatinine equation is suitable for estimating renal function. The CKD-EPIcr-cys equation would provide further reliability in individuals with a CKD-EPI creatinine eGFR of 45–59 ml/min/1.73 m2, but the cost of serum cystatin C analysis limits its use in everyday general practice.

Introduction
In 2002, a definition and classification of chronic kidney disease were introduced by the United States Kidney Foundation (K/DOQI) (Table 1) (1). This framework provided new ‘order’ to the medical community which had for a long time been ignored less severe chronic kidney disease than end-stage renal disease, and used poorly defined terms like renal insufficiency or pre-uremia. Glomerular filtration rate (GFR) < 60 ml/min/1.73 m2 measured repeatedly at least 3 months apart was adapted to define chronic kidney disease irrespective of other signs of kidney damage, because it represents a reduction of more than half of the normal value of 125 ml/min/1.73 m2 in young adults, and this level of GFR is associated with the onset of laboratory abnormalities characteristic of kidney failure, including increased prevalence of several cardiovascular risk factors (2). The GFR is equal to the sum of the filtration rates in all functioning nephrons and can be regarded as a rough measure of the number of them. The new, consistent definition of chronic kidney disease aimed to identify an earlier, often asymptomatic stage where interventions may prevent complications of chronic kidney disease and progression to end-stage kidney disease.
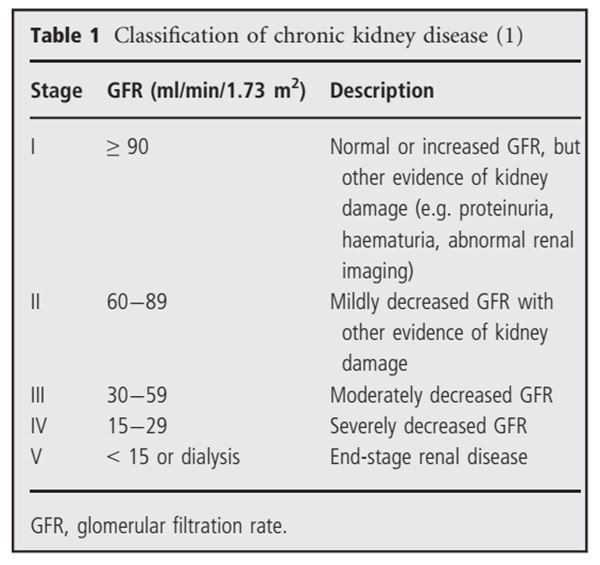
However, as always in medical practice, the success of therapy depends on the accuracy of diagnosis. GFR can be measured (measured GFR, mGFR) quite precisely by the clearance of an infused exogenous substance like inulin or radiopharmaceuticals (125I-iothlamate, 51Cr-EDTA, 99mTc-DTPA), but the difficulty in use and expense prevent applying these methods for large-scale clinical practice. This article reviews available information on assessing kidney function in the stable phase. The PubMed database was searched for relevant articles using the terms 'creatinine', 'creatinine clearance', 'cystatin C', 'Cockcroft-Gault', 'glomerular filtration rate', 'MDRD' 'CKD-EPI'. There were no date restrictions placed on the search. Twenty articles were identified.
This article reviews available information on assessing kidney function in the stable phase. The PubMed database was searched for relevant articles using the terms 'creatinine, 'creatinine clearance', 'cystatin C', 'Cockcroft-Gaul', 'glomerular filtration rate', 'MDRD' 'CKD-EPI'. There were no date restrictions placed on the search. Twenty articles were identified.
Additional papers were identified through the references and related citations of selected articles with a focus on clinically relevant topics.
Serum creatinine
Assessment of kidney function has for decades been based on serum creatinine concentration, which is an inexpensive, common test in clinical practice. However, serum or plasma creatinine is quite an inaccurate test for estimating kidney function: it begins to rise only when GFR has diminished by one-half, and thereafter the rise is exponential, not linear to GRF deterioration (3). Serum creatinine is also affected by age, sex, muscle mass or breakdown, diet, race, tubular secretion, drugs (e.g. amiloride, triamterene, spironolactone, trimethoprim), and laboratory analytical methods (3,4). Fortunately, the implementation of calibration standardization for serum creatinine assays, the isotope dilution mass spectrometry (IDMS)-traceable (gold standard) creatinine assays, has largely resolved the inter-laboratory differences in serum creatinine results (5).
Creatinine clearance
GFR can be assessed indirectly by measuring the clearance of creatinine from serum with a 24-h urine collection.

However, calculating creatinine clearance shares all the limitations of creatinine measurement and requires accurate urine collection which challenges its reliability in routine clinical practice.
Serum cystatin C
Cystatin C is a non-glycosylated protein synthesized and secreted by all nucleated cells, it is freely filtered by the glomerulus and then reabsorbed and catabolized by the proximal tubules (6–8). Thus, GFR is the main determinant of cystatin C serum concentration. In contrast to creatinine, cystatin C concentration is less influenced by age, sex, muscle mass, or diet, but may be influenced by obesity (9), thyroid function (10), smoking (11,12), inflammation (11,13), steroid therapy (14) and viral load in HIV (15). There are nowadays international laboratory reference standards for cystatin C, but the cost of the analysis is approximately twofold higher than measuring serum creatinine concentration.
Equations for estimating glomerular filtration rate
Several approaches have been made to overcome the pitfalls of creatinine measurement in assessing kidney function. The estimated glomerular filtration rate (eGFR) equations use age, sex, race, and body size as surrogates for endogenous creatinine generation and excretion. However, these formulae are still based on serum creatinine and cannot perceive differences in creatinine production, tubular secretion, or extrarenal elimination between individuals. Taking these error components into account, an eGFR within 30% of mGFR is considered acceptable by K/DOQI for clinical interpretation to identify individuals with chronic kidney disease (1)
Cockcroft–Gault equation
Cockcroft and Gault developed an equation for estimating GFR from a small cohort of hospitalized patients in 1976 (16). The equation is mathematically simple:

This equation has important limitations: the development data set included only white subjects (4% females), the reference method used was creatinine clearance (not GFR), and the covariate weight is a potential source of error in obese subjects or patients with low body mass index.
Modification of Diet in Renal Disease equation
In 1999, Levey et al. introduced a new eGFR equation based on GFR values measured by iothalamate clearance (17). The formula does not require a weight variable because it normalizes GFR for a standard body surface area of 1.73 m2. The equation was developed from the database of the Modification of Diet in Renal Disease (MDRD) Study containing subjects with various non-diabetic kidney diseases (mean GFR 40 ml/min/1.73 m2) (17). The simplified four-variable MDRD Study equation for use with IDMS traceable (gold standard) serum creatinine assay (18) is nowadays the most commonly used equation for eGFR:
eGFR(ML/MIN/1.73m2
=175 x (serum creatinine/88.4) -1.154
x (age)-0.203 x (0.742 if female)
x (1.212 if back)
In a recent meta-analysis of general population cohorts, the risk of total mortality became significant around eGFR 60 ml/min/1.73 m2 and was two times higher around eGFR 30–45 ml/min/1.73 m2 compared with optimum eGFR levels 90–104 ml/min/1.73 m2 calculated with the MDRD formula (19).
When the reporting of eGFR using the MDRD Study equation was initially introduced in the UK, the number of referrals from primary care to nephrologists rose 2.7-fold (20). This reflects the increased identification of patients not previously suspected of having chronic kidney disease based on simple creatinine measurement but has also raised concerns about overdiagnosis of chronic kidney disease in the general population (21). In fact, the MDRD Study equation has been shown to systematically underestimate true GFR in subjects with measured GFR ≥ 60 ml/min/1.73 m2 (22–25).
Chronic kidney disease creatinine equation
The Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) developed a new eGFR equation to provide a more accurate estimate of GFR among individuals with normal or mildly reduced GFR (26).
The CKD-EPI equation was derived from studies including 5504 subjects with or without chronic kidney disease, who had a wide range of GFRs (mean GFR 67 ml/min/1.73 m2) measured by iothalamate clearance (27). In the CKD-EPI equation, different exponents are applied to serum creatinine according to creatinine level and gender:
eGFR (ml/min/1.73m2)
=141 x min (serum creatinine/k,1 ) α
x max (serum creatinine /k,1)-1.209
x 0.993 age x 1.018 (if female) x 1.159 ( if back)
min indicates the minimum of serum creatinine/k or 1, and max indicates the maximum of serum creatinine/k or 1
k 0.7 for females, 0.9 for males
a -0.329 for females, -0.411 for males
The CKD-EPI equation has been shown to outperform the MDRD Study equation, especially for individuals with a GFR greater than 60 ml/min/1.73 m2; the median differences between estimated and measured GFR (a measure of bias) were-10.6 ml/min/1.73 m2 for the MDRD Study equation and-3.5 ml/min/1.73 m2 for the CKD-EPI equation (26).
Three large population-based cohorts have recently compared the CKD-EPI and MDRD equations. In the US adult population aged ≥ 20 years, the prevalence of chronic kidney disease was 11.6% using the CKD-EPI equation and 13.1% with the MDRD equation (26). The corresponding figures in the general Australian adult population were 11.5% and 13.4% (28). The prevalence of chronic kidney disease stage 3 (eGFR 30–59 ml/min/1.73 m2) was decreased from 2.5% with the MDRD formula to 1.4% when the CKD-EPI formula was applied in a cohort of the US population aged 45–64 years (29). Importantly, participants of the two last-mentioned studies who were reclassified upward from eGFR 30–59 ml/l/1.73 m2 based on the MDRD formula to eGFR 60–89 ml/min/1.73 m2 using the CKD-EPI formula, had a lower risk of all-cause mortality, major cardiovascular events, and end-stage renal disease compared with those who were not reclassified (28,29).
CKD-EPI cystatin C and CKD-EPI creatinine-cystatin C equations
Recently, the CKD-EPI consortium proposed two new equations for estimating GFR; one using serum cystatin C (CKD-EPIcys) and another using both serum creatinine and serum cystatin C (CKD-EPIcrcys) (30). The CKD-EPI cystatin C equation:
133 x min (serum cystatin C/0.8,1)-0.499
x max (serum cystatin C/0.8,1)-1.328 x 0.996 age
x 0.932 ( if female)
min indicates the minimum of serum cystatin C/k or 1, and max indicates the maximum of serum cystatin C/k or 1. The CKD-EPI creatinine–cystatin C equation:
135 x min (serum creatinine/k,1 ) α
x max (serum creatinine /k,1)-0.601
x min (serum cystatin C/0.8,1)-0.375
x max (serum cystatin C/0.8,1)-0.711 x 0.995 age
x 0.969 ( if famle) x 1.08 ( if back)
k is 0.7 for females and 0.9 for males
α is -0.248 for females and -0.207 for males min indicates the minimum of serum creatinine/k or 1, and max indicates the maximum of serum creatinine/k or 1
Data from 13 cohorts of diverse populations were used for the development of these equations, and GFR measurements were based on urinary or plasma clearance of exogenous filtration markers. The CKD-EPIcys equation performed similarly to the CKD-EPI creatinine equation in different subgroups of patients including those with low body mass index. Interestingly, the combined CKD-EPcr-cys equation performed better than the equations that used creatinine or cystatin C alone. In participants whose eGFR based on CKD-EPI creatinine was 45–59 ml/min/1.73 m2, the CKD-EPIcr-cys equation correctly reclassified 17% of them having a GFR ≥ 60 ml/min/1.73 m2 (30).
In a meta-analysis of 11 general population studies, the prevalence of an eGFR < 60 ml/min/1.73 m2 was 9.7% with the CKD-EPI creatinine equation, 13.7% with the CKD-EPIcys equation, and 10.0% with the combined CKD-cys equation (31). The meta-analysis also included five chronic kidney disease cohorts with 2960 participants. The cystatin C-based calculation of eGFR reclassified 42% of the study participants with a creatinine-based eGFR of 45–59 ml/min/1.73 m2 to > 60 ml/min/1.73 m2. Moreover, the reclassified subjects had a relative reduction of 34% in the risk of death from any cause, as compared with persons in whom the eGFR was not reclassified (31). The risk of death from any cause and from cardiovascular diseases was increased for all eGFR levels when values for CKD-EPIcys and CKD-EPIcr-cys were below a threshold of 85 ml/min/1.73 m2 (31). Thus, eGFR equations based on the measurement of cystatin C can be used to detect increased risks of adverse outcomes that are not detected with the only creatinine-based calculation of the eGFR.
The Kidney Disease Improving Global Outcome (KDIGO) 2012 Clinical Practice Guideline recommends the measurement of serum cystatin C and estimation of GFR by the combined CKD-EPIcr-cys question in adults in whom eGFR based on serum creatinine is within the range of 45–59 ml/min/1.73 m2, but who do not have markers of kidney damage (32).
In a study of a European ancestry population aged 74 years and older, the three CKD-EPI equations appeared to be more accurate than the MDRD Study equation, but the difference was significant only for individuals with an eGFR ≥ 60 ml/min/1.73 m2 (33). Thus, the MDRD Study equation performs quite well in clinical practice among elderly people, whose muscle mass, dietary protein intake, and GFR are more akin to the diseased development cohorts of the MDRD Study (33). Moreover, the effect of aging on cystatin C levels is strong, especially in men (34).
Summary
The characteristics of the patients to whom the diagnostic test is applied and the stage or severity of the disease (Table 2) can influence the sensitivity of the test (35). In the overwhelming majority of patients currently treated in primary care, the CKD-EPI creatinine equation is suitable for estimating renal function. The CKD-EPIcr-cys equation would provide further reliability in individuals with a CKD-EPI creatinine eGFR of 45–59 ml/min/1.73 m2 (31), but the cost of serum cystatin C analysis and the accuracy in elderly subjects still limits its use in everyday general practice.

The patient described in the vignette has an eGFR of 58 and 62 ml/min/1.73 m2 according to the MDRD Study and the CKD-EPIcreatinine equation, respectively. Because of her obesity, the Cockcroft–Gault formula was regarded as biased. Serum cystatin C was not measured and thus, the CKD-EPIcys and CKD-EPIcr-cys equations could not be used.
Although the patient has no albuminuria or other signs of kidney damage, she is diagnosed with chronic kidney disease according to the MDRD Study equation. This might trigger her physician to reduce the dosage of really excreted drugs ACE inhibitor and metformin. However, the diagnosis of chronic kidney disease requires two eGFR measurements at least 3 months apart (32). It should be emphasized that follow-up of the kidney function is very important in clinical decision-making. If our patient will have stable kidney function during follow-up, she is just likely to have suffered from the cumulative effects of hypertension, diabetes, and dyslipidemia over the years with ensuing ischemia and renal dysfunction. In that case, she will continue to benefit from the prescription of heart and kidney protective drugs such as ACE inhibitors and metformin. The KDIGO guideline defines chronic kidney disease progression based on the decline in the eGFR category (Table 1) accompanied by a≥ 25% drop in eGFR from baseline, or a sustained decline in eGFR > 5 ml/min/1.73 m2 per year (33).
However, our patient with at least mildly decreased kidney function is at risk of acute or chronic kidney injury at times of physiological stress. Therefore, it is recommended that potentially nephrotoxic and really excreted drugs (ACE inhibitors, angiotensin-receptor blockers, aldosterone inhibitors, renin inhibitors, diuretics, NSAIDs, metformin, lithium, digoxin) should be discontinued at least temporarily during acute illness, for example, serious infection, vomiting, diarrhea or surgical procedures (33).
Management of blood pressure is highly significant in the prevention of chronic kidney disease progression. The KDIGO 2012 Clinical Practice Guideline (33), the European Society of Hypertension and European Society of Cardiology 2013 Guidelines for the management of arterial hypertension (36), and the Eighth Joint National Committee (JNC 8) Guideline 2014 (37) recommend that in patients with chronic kidney disease and without albuminuria, target blood pressure should be < 140/90 mmHg and initial drug treatment option would be an ACE inhibitor or an angiotensin-receptor blocker. Thus, the patient described in the vignette should have her blood pressure lowered to consistently achieve this level.
![]() 2014 John Wiley & Sons Ltd
2014 John Wiley & Sons Ltd
Int J Clin Pract, February 2015, 69, 2, 156–161. DOI: 10.1111/ijcp.12516
References
1 National Kidney Foundation. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Executive summary. Am J Kidney Dis 2002; 39: S17–31.
2 Sarnak MJ, Levey AS, Schoolwerth AC et al. Kidney disease as a risk factor for the development of cardiovascular disease. A statement from the American Heart Association Councils on kidney in cardiovascular disease, high blood pressure research, clinical cardiology, and epidemiology and prevention. Hypertension 2003; 42: 1050–65.
3 Shemesh O, Golbety H, Kriss JP, Myers BD. Limitations of creatinine as a filtration marker in glomerulopathic patients. Kidney Int 1985; 28: 830–8.
4 Perrone RD, Madias NE, Levey AS. Serum creatinine as an index of renal function: new insights into old concepts. Clin Chem 1992; 38: 1933–53.
5 Pieroni L, Delanaye P, Boutten A, et al. A multicentric evaluation od IDMS-traceable creatinine enzymatic assays. Clin Chem Acta 2011; 412: 2070–5.
6 Abrahamson M. Structure and expression of the human cystatin C gene. Biochem 1990; 268: 287–94.
7 Jacobsson B, Lignelid H, Bergerheim US. Transthyretin and cystatin C are catabolized in proximal tubular epithelial cells and the proteins are not useful as markers for renal cell carcinomas. Histopathology 1995; 26: 559–64.
8 Tenstad O, Roald AB, Grubb A, Aukland K. Renal handling of radiolabelled human cystatin C in the rat. Scand J Clin Lab Invest 1996; 56: 409–14.
9 Naour N, Fellahi S, Renucci J-F et al. Potential contribution of adipose tissue to elevated serum cystatin C in human obesity. Obesity 2009; 17: 2121–6.
10 Fricker M, Wiesli P, Brandle M, Schwegler B, Schmid C. Impact of thyroid dysfunction on serum cystatin C. Kidney Int 2003; 63: 1944–7.
11 Knight EL et al. Factors influencing serum cystin C levels other than renal function and the impact on renal function measurement. Kidney Int 2004; 65:1416–21.
12 Galteau MM, Guyon M, Gueguen R, Siest G. Determination of cystatin C; biological variation and reference values. Clin Chem Lab Med 2001; 39: 850–7.
13 Stevens LA, Schmid CH, Greene T et al. Factors other than glomerular filtration rate affect serum cystatin C levels. Kidney Int 2009; 75: 652–60.
14 Risch L, Huber AR. Glucocorticoids and increased serum cystatin C concentrations. Clin Chim Acta 2002; 320: 133–4
15 Gagneux-Brunon A, Mariat C, Delaney P. Cystatin C in HIV-infected patients: promising but not yet ready for prime time. Nephrol Dial Transplant 2012; 27: 1305–13.
16 Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron 1976; 16: 31–41.
17 Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med 1999; 130: 461–70.
18 Levey AS, Coresh J, Greene T et al. Expressing the MDRD study equation for estimating GFR with IDMS traceable (gold standard) serum creatinine values. J Am Soc Nephrol 2005; 16: 69A.
19 Chronic Kidney Disease Prognosis Consortium. Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: a collaborative meta-analysis. Lancet 2010; 375: 2073–81.
20 Richards N, Harris K, Whitfield M, et al. The impact of population-based identification of chronic kidney disease using estimated glomerular filtration rate (eGFR) reporting. Nephrol Dial Transplant 2008;
21 Moynihan R, Glassock R, Doust J. Chronic kidney disease controversy: how expanding definitions are unnecessarily labeling many people as diseased. BMJ 2013; 347: f4298.
22 Stevens LA, Coresh J, Feldman HI et al. Evaluation of the Modification of Diet in Renal Disease Study equation in a large diverse population. J Am Soc Nephrol 2007; 18: 2749–57.
23 Rule AD, Larson TS, Bergstralh EJ, Slezak JM, Jacobsen SJ, Cosio FG. Using serum creatinine to estimate glomerular filtration rate: accuracy in good health and in chronic kidney disease. Ann Intern Med 2004; 141: 929–37.
24 Rule AD, Gussak HM, Pond GR et al. Measured and estimated GFR in healthy potential kidney donors. Am J Kidney Dis 2004; 43: 112–9.
25 Stevens LA, Coresh J, Greene T, Levey AS. Assessing kidney function – measured and estimated glomerular filtration rate. N Engl J Med 2006; 354: 2473–83.
26 Levey AS, Stevens LA, Schmid CH et al. A new equation to estimate glomerular filtration rate. Ann Intern Med 2009; 150: 604–12.
27 Levey AS, Coresh J, Balk E et al. National Kidney Foundation Practice Guidelines for Chronic Kidney Disease: Evaluation, Classification, and Stratification. Ann Intern Med 2003; 139: 137–47.
28 White SL, Polkinghorne KR, Atkins RC, Chadban SJ. Comparison of the prevalence and mortality risk of chronic kidney disease in Australia using the chronic kidney disease Epidemiology Collaboration (CKD-EPI) and Modification of Diet in Renal Disease (MDRD) Study GFR estimating equations: The AusDiab (Australian Diabetes, Obesity, and Lifestyle) Study. Am J Kidney Dis 2010; 55: 660–70.
29 Matsushita K, Selvin E, Bash LD, Astor BC, Coresh J. Risk implications of the new Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation compared with the MDRD Study equation for estimated GFR: The Atherosclerosis Risk in Communities (ARIC) Study. Am J Kidney Dis 2010; 55: 648–59.
30 Inker LA, Schmid CH, Tighiouart MS et al. Estimating glomerular filtration rate from serum creatinine and cystatin C. N Engl J Med 2012; 367: 20–9.
31 Shlipak MG, Matsushita K, Arnel € of J et al. Cysta- € tin C versus creatinine in determining risk based on kidney function. N Engl J Med 2013; 369: 932–43.
32 Kidney disease: Improving Global Outcomes (KDIGO) chronic kidney disease Work Group. KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int 2012; 2013(Suppl. 3): 1–150.
33 Kilbride HS, Stevens PE, Eaglestone G et al. Accuracy of the MDRD (Modification of Diet in Renal Disease) Study and CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration) equations for estimation of GFR in the elderly. Am J Kidney Dis 2013; 61: 57–66.
34 Werner KB, Elmstahl S, Christensson A, Pihlsgard M. Male sex and vascular risk factors affect serum cystatin C–derived renal function in older people without diabetes or overt vascular disease. Age and Aging 2013; 43: 411–7.
35 Machin D, Campbell MJ, Walters SJ. Medical Statistics. A Textbook for the Health Sciences. 4th edn. Chichester: John Wiley & Sons Ltd, 2007.
36 The Task Force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). ESH/ESC Guidelines for the management of arterial hypertension. J Hypertens 2013; 2013(31): 1281–357.
37 Report from the Panel Members Appointed to the Eighth Joint National Committee (JNC 8). Evidence-Based Guideline for the management of high blood pressure in adults. JAMA 2014; 311(5): 507–20.










