Innate And Adaptive Immune Memory: An Evolutionary Continuum in The Host's Response To Pathogens(Part 1)
Jun 08, 2022
To learn more info plz contact david.wan@wecistanche.com
Immunological memory is an important evolutionary trait that improves host survival upon reinfection, Memory is a characteristic recognized within both the innate and adaptive arms of the immune system. Although the mechanisms and properties through which innate and adaptive immune memory are induced are distinct, they collude to improve host defense against pathogens. Here, we propose that innate immune memory, or "trained immunity, "is a primitive form of adaptation in host defense, resulting from chromatin structure re-arrangement, which provides an increased but non-specific response to reinfection. In contrast, adaptive immune memory is more advanced, with an increased magnitude of response mediated through epigenetic changes, as well as specificity mediated by gene recombination. An integrative model of immune memory is important for a broad understanding of host defense, and for identifying the most effective approaches to modulate it for the benefit of patients with infections and immune-mediated diseases.

Click here to learn more about Cistanche
Innate and Adaptive Immunity
Classically, host immunity is divided into innate and adaptive immune responses. The former reacts rapidly and non-specifically to pathogens, whereas the latter responds in a slower but specific manner, with the generation of long-lived immunological memory (Farber et al., 2016). This dichotomy has dictated the last half-century of immunological research, and a vast number of studies have defined the cellular and molecular substrates of each of these two major components of host defense. Innate immunity is mediated by innate immune cell populations such as myeloid cells, natural killer(NK) cells, and innate lymphoid cells (but non-immune cells in specific circumstances as well), as well as by ancient humoral systems such as defensins and complement. Adaptive immunity is a relatively new evolutionary trait based on the immunoglobulin family and cells such as B- and T-lymphocytes in jawed vertebrates (Danilova, 2012)and on variable lymphocyte receptors (VLR) comprised of leucine-rich-repeat (LRR) segments in jawless vertebrates (Boehm et al., 2012).
During an infection, the innate immunity is the first to be triggered (the inflammatory reaction), taking no longer than minutes to hours to be fully activated (Netea et al.,2017). This is crucial for the host defense in the first phase of a new infection. While innate immunity is generally able to eliminate the pathogens efficiently, initial clearance of infection can fail due to the high number or virulence of invading pathogens. In these situations, lymphocytes and adaptive immune mechanisms are activated, which allows specific recognition and elimination of the pathogen. The establishment of adaptive immunity needs 1-2 weeks and is important for host defense during the latter phases of infection and during secondary infections due to its capacity to"remember" and respond more effectively to restimulation (Farber et al.,2016).
The innate and adaptive immune processes were initially seen as relatively compartmentalized responses in time, but research in the last two decades has clearly demonstrated strong links and an efficient network between them. Activation of the adaptive immune response and induction of classical immune memory in lymphocytes are dependent on the innate immune system, in particular on antigen-presenting cells such as dendritic cells. The downstream effects of lymphocyte activation are then exerted by amplifying innate immune responses such as phagocytosis and killing of pathogens by certain innate immune cells. Two properties that discriminate between innate and adaptive immunity are on the one hand specificity, and on the other hand the capacity to build long-lived immunological memory. Innate immune responses are traditionally thought to be non-specific and without the capacity to adapt, whereas adaptive immune responses recognize with great precision different pathogens using gene recombination processes in the immunoglobulin gene family, and subsequently build immunological memory (Danilova, 2012) (Figure 1). The concept that innate immunity is non-specific has been challenged by the discovery of pattern recognition receptors(PRR). These receptors expressed on a variety of cells within the innate immune system recognize specific components of microorganisms, and the combination of PRRs expressed by an immune cell can lead to partially specific recognition of the type of microorganism encountered: for example, innate immune cells recognize the difference between a Gram-negative and a Gram-positive bacteria, but not between two closely related species or strains(Medzhitov and Janeway, 2000). We will not discuss the progress in understanding the pathogen recognition through PRRs in this review, as this is the subject of many excellent recent overviews in the literature (e.g. O'Neill et al., 2013).

Figure 1. Innate versus Adaptive Immune Responses
During the first hours and days of infection, invasion by pathogens induces activation of innate immune cells such as macrophages, monocytes, or NK cells or humoral factors such as complement. These pathways strongly activate an inflammatory reaction and eliminate pathogens (innate immunity). In the few cases when the infection is not eliminated, pathogens are ingested and processed by antigen-presenting cells, followed by antigen presentation and stimulation of specific activation of T- and B-lymphocytes. In turn, this leads to clonal expansion and activation of effector mechanisms (e.g., the release of cytokines, and immunoglobulins) (adaptive immunity).
Here, we will focus on the second property discriminating innate and adaptive host defense mechanisms: induction of immune memory. In contrast to early literature, a growing body of evidence shows that innate immune responses exhibit adaptive characteristics (Bowdish et al.,2007; Netea et al.,2011). In plants and invertebrates that lack adaptive immunity, sustained protection against reinfection has been reported (Kurtz,2005). Studies in mammals have demonstrated that there is cross-protection against infections with different pathogens (Quintin et al, 2014), while experimental studies of mice devoid of functional adaptive immune cells have shown partial protection in certain models of vaccination (Bistoni et al.,1986; can't Wout et al, 1992). It is thus apparent that innate immunity can be modulated by previous encounters with microbes or microbial products, and this property has been termed "trained immunity" and represents a de facto innate immune memory.
However, there are distinct molecular mechanisms that mediate the two types of immune memory. Based on molecular, immunological, and evolutionary arguments, we propose that innate immune memory is a primitive form of immune memory present in all living organisms, while adaptive immune memory is an advanced form of immune memory representing an evolutionary innovation in vertebrates. Based on the complementary properties of the two types of immune memory, and on a range of biological arguments as described below, we argue in this Perspective that the development of innate and adaptive immune memory represents an evolutionary continuum. We also propose that these two forms are two evolutionary steps toward the development of effective mechanisms of adaptation to an environment teeming with potentially pathogenic microorganisms.
Immunological Memory as an Adaptive Evolutionary Trait
The dogma that immunological memory is confined to the adaptive immune system faces a conceptual difficulty when considered from an evolutionary perspective. That immune memory is advantageous from an evolutionary point of view is well illustrated by deadly ancient diseases such as smallpox: while mortality was historically 20%-60% for first infections, individuals became completely immune to the disease thereafter (Riedel, 2005). Therefore, it is difficult to envision that immune memory evolved only in vertebrates, which represent approximately 1%-2% of living species (Gourbal et al.,2018).In contrast, other important advantageous traits, such as vision, evolved independently several times during the evolution of various groups of animals (so-called"convergent evolution")(Gehring, 2004). Interestingly, immunological memory within the vertebrate lineage evolved not just once, but twice. First, the development of VLR-based memory in the jawless fish (such as the lamprey)and, second, the development of the LG-based memory (which is also the basis for the development of B- and T-lymphocyte memory) in all the other jawed vertebrates. That there would be two separate events leading to immune memory in vertebrates (Cooper and Alder,2006), while being completely absent in all other metazoans, is very unlikely.
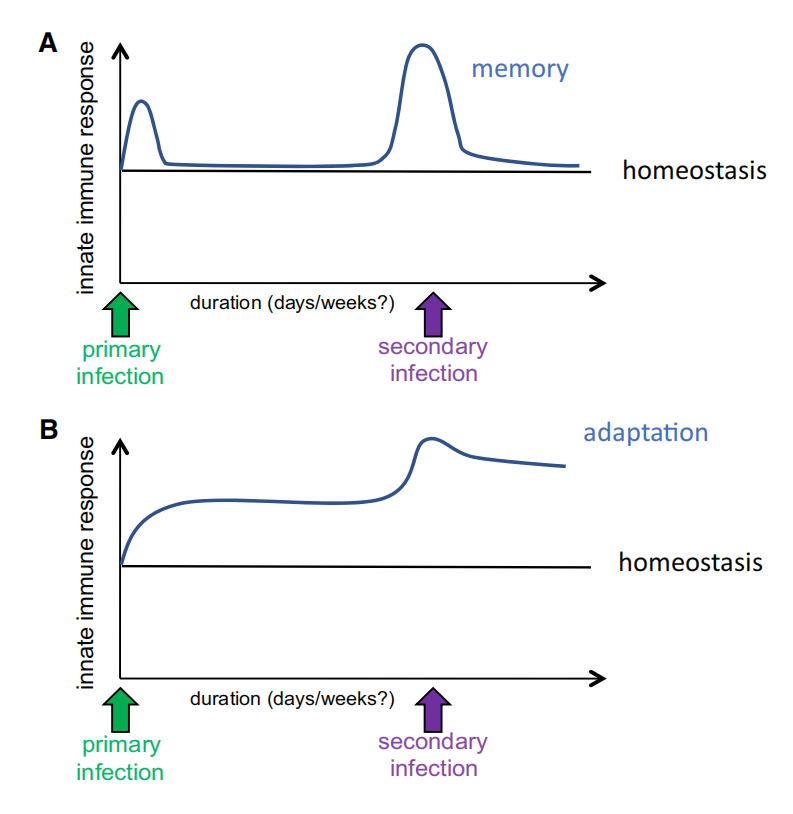
Figure 2. Immune Memory versus Immune Adaptation
Immune memory is defined as a changed status of the immune system of a host after acute infection (or vaccination), leading to a more effective response against reinfection. Importantly, during induction of immune memory, between the first infection and the re-infection, the immune status functionally returns to a low basal state, while the capacity to respond stronger to restimulation is imprinted at the epigenetic level (A). In contrast, adaptation is defined as the long-term change in the immune response determined by a constant change in the environmental conditions, or due to a chronic insult (e.g., a chronic infection), leading to a new functional state. Importantly, the functional immune status during adaptation does not return to the low basal state existing before the insult (B).
In line with this, a large number of studies of plant immunology have demonstrated that plant host defense includes adaptive characteristics, a response termed Systemic Acquired Resistance (SAR)(Jaskiewicz et al.,2011; Kachroo and Robin,2013; Reimer-Michalski and Conrath, 2016). The molecular mechanisms and biochemical mediators of SAR are largely known (Kachroo and Robin, 2013), with epigenetic-based rewiring of host defense being suggested to play a central role(Luna and Ton,2012).In addition, increasing evidence has challenged the belief that invertebrate innate immunity lacks memory traits (Kurtz,2005). Adaptive properties of innate immune responses have been reported in several invertebrate lineages; for example, the microbiota has been shown to induce innate immune memory to protect mosquitoes against Plasmodium(Rodrigues et al, 2010), and the social insect Bombus Terrestris to display innate immune memory against three different pathogens (Sadd and Schmid-Hempel,2006), and the tapeworm Schistocephalus solidus to induce memory in the copepod crustacean (Kurtz and Franz, 2003), and innate immune memory is a defense mechanism in snails (Pinaud et al,2016). These studies have been complemented by data in vertebrates showing that it is possible to induce partial protection from reinfection in experimental murine models even in the absence of functional adaptive immune responses (Bistoni et al.,1986; can't Wout et al.,1992). Moreover, epidemiological studies have shown heterologous protection by human vaccines against a broad spectrum of infections (Benn et al.,2013), with mechanisms likely independent from classical adaptive immune responses. Therefore, it is reasonable to conclude that immunological memory can be found not only in vertebrates but also in plants and lower animals(Kurtz,2005; Ne-tea et al., 2011) that do not harbor a classical adaptive immune system.

Innate and Adaptive Immune Memory: an Evolutionary Process
If these data strongly argue for the presence of immune memory in all groups of higher organisms, how can this be defined? A simple definition is that immunological memory refers to the ability of the immune system to respond more rapidly and effectively to a pathogen. This definition encompasses all processes described as immune memory in plants, invertebrates, and vertebrates, although it does not discriminate between the differences in action observed within various organismal groups. In a recent review, Pradeu and Du Pasquier (2018) propose a multidimensional model of immune memory, in which they discriminate no less than six varieties of immune memory: classical adaptive memory in vertebrates, NK-cell immune memory, trained immunity in myeloid cells, priming in invertebrates, immunological memory in plants (e.g., SAR), and Crispr/Cas-based memory in bacteria and archaea (Pradeu and Du Pasquier,2018). These forms of immunological memory differ in their properties based on a number of basic characteristics of the response: strength, speed, extinction (reversal to the basal state), duration, and specificity. However, while this classification is useful for understanding the ubiquitous presence of immune memory in all living organisms, we believe that this compartmentalization of immune memory does not reflect the common molecular, biological, and functional substrate of the memory characteristics in different organisms.
In this Perspective, we propose a unifying model of immune memory that is based on two concepts:
There is a conceptual difference between two forms of immune adaptation: immune memory and immune differentiation.
An evolutionary continuum links innate and adaptive immune memory.
The first concept that needs to be defined is represented by the difference between immune memory and immune adaptation. We define immune memory as a changed status of the immune system of a host after acute infection (or vaccination), leading to a more effective response against re-infection. Importantly, during induction of immune memory, between the first infection and the reinfection, the immune status functionally returns to a low basal state, while the capacity to respond stronger to restimulation is imprinted at the epigenetic level (Figure 2A). In contrast, we define immune differentiation as a form of adaptation through long-term changes in the functional program of a system (including immune response), determined by a constant change in the environmental conditions or due to a chronic insult (e.g., a chronic infection), leading to a new functional state. Importantly, the functional immune status during differentiation does not return to the low basal state existing before the insult (Figure 2B). There are many biological situations characterized by adaptation through immune differentiation: one such situation may be induced for example by stable changes in gut microbiota, which is known to induce long-term effects on the local immune responses. The process of immune differentiation is the subject of many other excellent reviews, and it will not be the focus of this Perspective.
Another important distinction to make is between priming and memory. Priming is also a term that has been used to describe increased responses to a secondary stimulus, however, this is often an acute process that does not involve long-term memory effects. For example, during acute malaria infection, there is a well-known hyper-responsiveness of immune cells to Gram-negative microorganisms that can lead to severe acute symptoms: the immune cells are primed to respond acutely with higher cytokine release. However, that does not imply a memory response that would persist for months or years.
Here, we will focus on the concept of immune memory, which we believe characterizes all living organisms. We propose that there is an evolutionary continuum between innate and adaptive immune memory based on two fundamental properties mediated by distinct mechanisms:
● Increased magnitude and kinetics of the immune re-
responses upon reinfection, which is mediated by epigenetic processes and characterizes both innate and adaptive immune memory (present in all organisms)
● Specificity of the memory responses, mediated by gene
recombination, a property-specific to adaptive immune memory (described until now only in vertebrates)

Innate Immune Memory
While in vertebrate immunity it has long been assumed that innate immune responses cannot adapt after an infection, and upon reinfection, an identical response is elicited each time, this assumption was never followed in plants or invertebrate immunity. In addition to studies on plants and invertebrate host defenses described above (see Immunological Memory as an Adaptive Evolutionary Trait), several mechanisms have been proposed to account for the induction of innate immune memory in invertebrates, including upregulation or "training" of regulatory pathways such as Toll or Imd (Boutros et al,2002) and quantitative and phenotypic changes in immune cell populations (Ro-drigues et al.,2010). Some investigators have also proposed the presence of diversity-generating mechanisms in insects. Such candidate mechanisms may involve fibrinogen-related proteins with high rates of diversification at the genomic level (Zhang et al.,2004) or upregulation of expression of specific receptors such as peptidoglycan recognition molecules and lectins (Steiner, 2004).
Important clues that vertebrate innate immunity also has adaptive characteristics have been reported in experimental studies in mice. Several such studies have shown that priming or training mice with microbial ligands can protect against lethal infection. For example, trained immunity induced by β-glucan (derived from fungi) induces protection against bacterial infection with Staphylococcus aureus (Di Luzio and Williams, 1978; Marakalala et al,2013). Similarly, the bacterial peptidoglycan component muramyl dipeptide induces protection against Toxoplasma (Krahenbuhl et al,1981), and CpG oligodeoxynucleotide administration protects against sepsis and Escherichia coli meningitis (Ribes et al,2014). Furthermore, flagellin from Gram-negative bacteria can induce protection against the Gram-positive bacterium Streptococcus pneumoniae (Munoz et al.,2010)and rotavirus (Zhang et al.,2014a). In addition to microbial ligands, there is evidence that certain pro-inflammatory cytokines may induce trained immunity: infection of mice with one dose of recombinant IL-1 three days before infection with Pseudo-monas aeruginosa protected mice against mortality(van der Meer et al.,1988). The nonspecific character of the trained immunity effects argues against a classical memory effect mediated by adaptive immunity and suggests activation of nonspecific innate immune mechanisms. An important aspect that will need to be investigated is the duration of the protective effects of trained immunity. Studies in mice and humans have shown effects after 3 months and even one year (Kleinnijenhuis et al, 2014), although epidemiological data based on the protective heterologous effects of vaccines suggests that they will be functional for at least 3-to 5 years.
Trained immunity may mediate at least some of the protective effects of vaccination. Compelling evidence comes from studies showing that vaccination with the tuberculosis vaccine bacillus Calmette Guerin (BCG), the most commonly used vaccine worldwide, induces T cell-independent protection against secondary infections with Candida albicans, Schistosoma mansoni, or influenza in animals (Spencer et al.,1977; Tribouley et al.1978; can't Wout et al.,1992). Human data complete these studies: BCG vaccination in human volunteers protects against an experimental infection with the yellow fever vaccine virus (Arts et al,2018), while large epidemiological studies have reported protective heterologous effects for BCG and measles vaccination(Benn et al,2013; Goodridge et al, 2016). In addition, herpesvirus latency increases resistance to the bacterial pathogens Listeria monocytogenes and Yersinia pestis (Barton et al.,2007) with protection achieved through enhanced production of IFNy and systemic activation of macrophages. Similarly, infection with the helminthic parasite Nip-strongyles brasiliensis induces a long-term macrophage phenotype that damages the parasite and induces protection from reinfection independently of T and B lymphocytes (Chen et al.,2014).
The main cell populations that have been reported to be responsible for innate immune memory are monocytes, monocyte-derived macrophages, and natural killer (NK) cells. Whereas macrophage-dependent secondary protection from infection is nonspecific, NK cell-mediated immune memory may provide increased specificity. The first evidence of NK cell memory comes from observations of different T and B cell-deficient mice (RAG knockout, SCID, and nu/nu) being able to mount robust recall responses to hapten-based con-tact sensitizers (O'Leary et al,2006). NK cells can mediate hapten-specific contact hypersensitivity (CHS) in these animals, yet the NK cell memory is restricted to liver-resident cells of the NK.1.1+DX5-CXCR6+CD49a+ phenotype (OLeary et al,2006; Paust et al,2010; Peng et al.,2013). Mouse liver-resident NK cells can develop specific memory toward a variety of haptens and other antigens, including virus-like particles (VLPs); however, the antigen recognition mechanism is not known (Paust et al.,2010). Antigen-specific and long-lasting (splenic and hepatic) NK memory cell responses were also observed in rhesus macaques, suggesting that antigen-specific memory NK cells may also exist in humans (Reeves et al.,2015)
NK cells can be activated by cytomegalovirus: after infection with murine cytomegalovirus (mCMV)(Nabekura et al,2015; Schlums et al.,2015; Sun et al,2009; Sunset al.,2012), NK cells bearing the Ly49H receptor proliferate, persisting in lymphoid and non-lymphoid organs during the contraction phase of the NK cell response. Upon reinfection, the"memory" NK cells undergo a secondary expansion, rapidly degranulating and releasing cytokines, thus inducing a protective immune response (Sun et al,2009). A number of possible mechanisms have been put forward to explain the memory properties of NK cells, involving either the IL-12/IFNy axis (Sun et al,2012) or the activation of the co-stimulatory molecule DNAM-1 (DNAX accessory molecule-1, CD226) (Nabekura et al.,2014).

CMV-induced NK memory appears to be specific: mouse studies demonstrated no enhanced responsiveness of mCMV-induced NK memory cells against other infections such as influenza or Listeria(Min-Oo and Lanier,2014). Similarly, human studies show no responsiveness of NKG2C+CD57+ NK cell population expanded in human (H)CMV+ individuals(Hendricks et al,2014). Cytokine-primed NK cells have been suggested also to develop memory-like properties. Mouse as well as human NK cells stimulated with a combination of IL12, IL15, and IL18 showed enhanced IFNy production in response to the secondary stimulation with cytokines or tumor cells weeks after cytokine priming (Cooper et al.,2009; Keppel et al,2013; Romee et al.,2012).
Innate lymphoid cells group 2 (ILC2) that share the common lymphoid progenitor with NK, B, and T cells, do not possess antigen receptors but can be activated by cytokines. They also show the potential to"remember" their activation status and generate enhanced responses upon secondary stimulation. In the lung, inhaled allergens stimulate ILC2s to produce IL-5 and IL-13 in an IL-33-dependent manner (Halim et al.,2014). After allergen-induced activation, lung ILC2s undergo expansion followed by a contraction phase in which they do not produce cytokines. A population of allergen-experienced ILC2s persists in the lung and lymph nodes. Upon a secondary challenge with unrelated allergens, memory-likeILC2s mount a more robust immune response. Sensitization of mice with IL-33 was sufficient to generate memory ILC2s responsive to allergen secondary stimulation indicating the non-antigen specific character of ILC2s memory (Martinez-Gonzalez et al.,2016).
These observations may also highlight the unique positioning of ILCs and NK cells at the evolutionary crossroads between innate and adaptive immunity.
Taken together, these complementary murine and human studies demonstrate that innate immune responses have the capacity to be"trained" and thereby exert a new type of immunological memory upon reinfection, for which the term trained immunity has been proposed (Netea et al,2016; Netea et al., 2011). An extension of the trained immunity concept has been recently proposed to contain also non-immune cell types, such as epithelial cells (Cassone,2018).






