Part 2: Acute Kidney Injury in Pediatric Patients Hospitalized With Acute COVID-19 And Multisystem Inflammatory Syndrome in Children Associated With COVID-19
Mar 11, 2022
Acute renal artery occlusion in a solitary kidney: An unexpected recovery after late revascularization
for more information:ali.ma@wecistanche.com
Keywords: acute renal artery occlusion, revascularization, acute kidney injury, kidney
For part 1, please click here.
Summary
Acute renal artery occlusion is a rare cause of acute kidney injury. In this case report, we present a patient with an acute renal artery occlusion who had an unexpected recovery after late renal artery revascularization. The patient, who had peripheral artery disease and a solitary kidney, presented with acute renal failure shortly after hospitalization for acute heart failure.
The cause was initially presumed to be acute tubular necrosis but the persistence of high blood pressure and the absence of recovery of the renal function suggested the presence of renal artery stenosis as a possible cause. On the CT angiogram, the right renal artery was occluded, with distal revascularization due to collateral vessels. The revascularization procedure, performed 16 days after admission, led to full recovery of the baseline kidney function.

Cistanche can improve kidney functions and avoid acute renal artery occlusion.
Click to cistanche deserticola ma and Cistanche for acute kidney injury
Background
Acute renal artery occlusion is an uncommon disease, with a variety of clinical presentations ranging from the classic acute flank pain with haematuria and lactate dehydrogenase (LDH) elevation to many other non-specific features such as fever, leucocytosis, nausea, abdominal pain, hypertension, acute renal failure.1, 2 These atypical presentations, sometimes associated with symptoms related to other organs, can delay the diagnosis. Furthermore, the typical presentation of a renal infarction has the same clinical signs of renal colic, a much more common disease. Moreover, the presentation may be different depending on whether the occlusion affects the main trunk or a branch of the renal artery, and whether it affects a solitary kidney or a kidney with a contralateral functioning one. The known causes are different and include thromboembolism to the kidney (atrial fibrillation being the most common cause), thrombosis of the renal artery, renal artery dissection, traumatic and iatrogenic causes. 3
There is very small evidence to support specific management of acute renal artery occlusion, based primarily on individual case reports or case series. In addition, the timing of the revascularization procedure is debated: although there is general agreement that earlier treatment leads to lower ischemic damage, there are limited data on the interventional window. Many studies have reported good outcomes with renal function recovery even after a prolonged ischemic time (with described cases of complete recovery after 31 days of anuria).

Case presentation
An 87-years-old man presented to our emergency department with acute pulmonary edema and acute kidney injury. The patient was in very good physical and cognitive conditions but had multiple comorbidities. His medical history consisted of arterial hypertension, a peripheral vascular disease with previous angioplasty and stenting of the vessels of the inferior limbs, diabetes mellitus controlled only with diet, and nephroangiosclerosis chronic kidney disease. He also had a solitary kidney after left nephrectomy for renal carcinoma treated 20 years before, with a baseline creatinine value of 135 μmol/L, with an estimated Glomerular Filtration Rate (eGFR) of 40 ml/min/1.73 m2 according to Chronic Kidney Disease Epidemiology Collaboration (CKD EPI) formula.
He had recently been admitted to the general medical ward for decompensated heart failure due to a new-onset tachycardic atrial fibrillation. He had been treated with intravenous diuretics, beta-blockers, and a direct oral anticoagulant. His plasma creatinine three days before discharge was 153 μmol/L (eGFR 35 ml/min/1.73 m2).
A day after discharge, the patient returned with acute onset of dyspnea. Oxygen saturation was 90% with 2 L/min of oxygen. The blood pressure (BP) and heart rate (HR) were both normal (BP 135/85 mmHg and HR 75 bpm).
The chest X-Ray showed signs of pulmonary edema, with bilateral pleural effusions. Serum creatinine was elevated at 470 μmol/L (eGFR
9 ml/min/1.73 m2), without electrolyte abnormalities or metabolic acidosis. There was no haematuria, neither micro nor macroscopic. An ultrasound examination of the right kidney and bladder excluded an obstructive cause and described a kidney with an age-appropriate morphology and size (100 × 51 mm).
The patient was admitted to the general medical ward with intravenous diuretic therapy, but despite this treatment, he remained anuric. Direct oral anticoagulation was replaced by intravenous therapeutic anticoagulation with unfractionated heparin. Therefore, the patient was transferred to the Intensive Care Unit (ICU), to start continuous hemofiltration. After 5 days of continuous renal replacement therapy, he was switched to intermittent hemodialysis, with the persistence of anuria. During his stay at the ICU, repeated ultrasound evaluation excluded structural abnormalities of the kidney and described the presence of blood perfusion at the renal hilum, whereas peripheral vascularization was difficult to sample.
The suspected diagnosis was acute tubular necrosis due to prolonged renal hypoperfusion secondary to low cardiac output. The patient was then transferred to the nephrology ward and continued intermittent hemodialysis three times a week, without neither improvement of renal function or recovery of diuresis.
After a couple of days, the patient presented with uncontrolled arterial hypertension (with values of systolic blood pressure up to 190 mmHg), despite the reintroduction of his home antihypertensive regimen, consisting of a calcium antagonist and an alpha-blocker.
Refractory hypertension, associated with acute kidney injury and flash pulmonary edema, suggested the presence of renal artery stenosis. A duplex ultrasound showed suspected severe stenosis of the right renal artery with an ostial acceleration and a distal post stenotic flow, suggesting a significant flow obstruction. The CT angiogram, acquired during the arterial phase only, demonstrated occlusion of the right renal artery at its origin from the aorta ( Fig. 1 ) and showed a normal-sized kidney without signs of edema, absence of perirenal fat infiltration, with a distal arrival of radiocontrast to the hilar artery branches due to collateral vascularization.

The decision to perform the angioplasty was a challenge. The elements supporting the procedure were the evidence of collateral circulation on the CT angiogram and the absence of clinical or indirect radiological evidence of renal infarction. The CT scan was considered mandatory to confirm the diagnosis and to study the vascular anatomy for accurate angioplasty planning. The patient never had any flank pain before or during the hospitalization and serum LDH was always within range. Moreover, due to the actual occlusion of the artery, the risks of a further worsening of the renal function were low, while the benefits of an even partial recovery of renal function would have al- lowed him to stop hemodialysis and would have drastically changed his quality of life. On the other hand, the occlusion of the main trunk of the right renal artery for more than 2 weeks left a very little hope of technical success of the intervention and renal function improvement; also, there was a procedural risk of embolic events.
After interdisciplinary discussions between the interventional radiologists, the nephrologist, the angiologist, the patient, and his family, on day 16 of anuria, we decided to perform the renal angioplasty.

Treatment
The preliminary aortic arteriography confirmed the occlusion of the right renal artery, with distal revascularization due to collateral circulation, with a delayed and weak contrast enhancement in the renal parenchyma ( Fig. 2 ).

After selective catheterization of the right renal artery, the injection of contrast showed a subtle flow of contrast in the artery. The presence of the subtle flux in the stenosis discouraged us to perform a thromboaspiration as it could have destabilized the atherosclerotic plaque. Nevertheless, even if an amount of fresh thrombus was present, it would have been negligible and would not have justified a thromboaspiration attempt. Considering the whole clinical picture, our opinion was that atherosclerotic stenosis was the main aspect in the pathogenesis of the acute renal artery occlusion and that our revascularization attempt had to focus on that.
The severe stenosis was treated with direct stenting with a balloon-expandable 5 × 21 mm stent (Paramount Mini GPS Stent, Medtronic, Minneapolis, Minnesota, USA).
We placed a 6F 45 cm introducer (CORDIS, Brite-Tip introducer sheath), with a 6F RDC guiding catheter (CORDIS, Vista-Brite Guiding Catheter). We used a Terumo, Radifocus® Guide Wire M Standard type 0035 ″ to deliver the guiding catheter and a 0014 ″ workhorse guidewire (Abbot, Hi-Torque Command ES) to overcome the stenosis and deliver the stent. Hemostasis at the femoral arterial puncture site was obtained using a closing device (Terumo, Angio-Seal 6F).
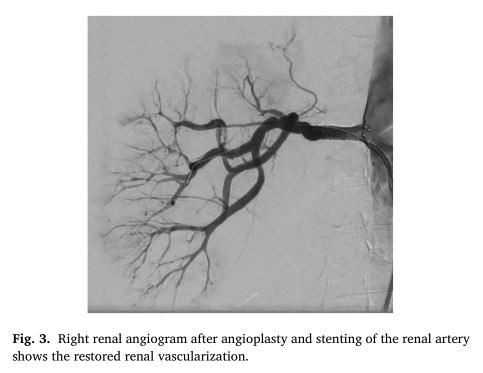
The angiogram performed after the procedure showed a correct positioning of the stent with a normal flow of contrast media through the renal artery and some small filling defects in one of the intraparenchymal branches, probably due to a distal embolus ( Fig. 3 ). Antiplatelet therapy with Clopidogrel was consequently added to oral anticoagulation.
Outcome and follow-up
On the first day after the procedure, the patient recovered an adequate urine output, the serum creatinine started decreasing and intermittent dialysis could be suspended.
On the following days, the blood pressure normalized, allowing us to stop the antihypertensive therapy. The patient was then discharged from the hospital 10 days after the procedure, with a creatinine of 194 μmol/L(eGFR 26 ml/min/1.73 m2).
An outpatient follow-up visit was planned a month after discharge. At the clinical control, he was asymptomatic, with a normal blood pressure without any antihypertensive treatment, and serum creatinine of 170 μmol/L (eGFR 30 ml/min/1.73 m2).
A subsequent follow-up almost six months after the procedure showed creatinine of 149 μmol/L (eGFR 36 ml/min/1.73 m2) and the patient still did not need any anti-hypertensive treatment.
Discussion
Although the role of renal artery stenting in atherosclerotic renovascular disease has been re-evaluated over time, 9–11 reserving the procedure to carefully chosen patients, revascularization procedures in the acute setting still need to be considered when the purpose of kidney salvage is taken into account.
In acute settings, the timing of successful revascularization is unclear, but there are several cases where a late intervention led to a complete recovery. 4 –8 It may be supposed that in the kidney, like in the brain or the heart, there is an area of “ischemic penumbra ”, whose extension and time duration depends on different factors.
In this case, we believe that the pre-existent chronic atherosclerotic disease, with a probable underlying severe renal artery stenosis, has led to a collateral circulation that managed to maintain the viability of the organ until treatment.
Some studies demonstrated that in patients with sudden ischemia of a healthy kidney (e.g. embolic or traumatic occlusion) the renal function loss is irreversible, with rapid progression of ischemia before any treatment can be performed. On the other side, in patients with thrombotic occlusion of a stenosed vessel (in the setting of an atherosclerotic renovascular disease), renal function has more probability to be re-established, regardless of the delay in diagnosis and treatment.13
Thrombotic occlusion most commonly occurs in the settings of pre-existing renal artery stenosis, as it probably happened in our patient. These patients have collateral circulation through per pelvic, periureteric and capsular vessels, that develop during the stenosis progression.14 Therefore, when occlusion occurs, these collaterals maintain a renal blood flow that can improve the tolerance to ischemic damage, preserving renal viability. Another interesting observation is the fact that revascularization after acute renal artery occlusion in kidney transplant patients has a lower rate of success, despite relatively short ischemic time, strengthening the importance of collateral or capsular vascularization.5
Conclusion
In conclusion, interventional radiology in acute renal artery occlusion should be discussed for the goal of kidney salvage, even in the setting of prolonged ischemia. Under selected circumstances, such as the presence of pre-existing stenosis, the kidney can tolerate a prolonged ischemia time and revascularization may re-establish renal function. We suggest discussing every case in a multidisciplinary meeting, balancing the risks and benefits of a revascularization attempt, knowing that there are successful reports after up to 31 days of anuria.

References
The source is by Ilaria Giordani et al on Annals of Vascular Surgery - Brief Reports and Innovations 1 (2021) 100021





