Part 2:Extracts Of Cistanche Deserticola Can Antagonize Immunosenescence And Extend Life Span in Senescence-Accelerated Mouse Prone 8 (SAM-P8) Mice
Mar 12, 2022
Contact: Audrey Hu Whatsapp/hp: 0086 13880143964 Email: audrey.hu@wecistanche.com
3.5. Extracts df Cistanche deserticola (ECD)Promote.
Apoptosis and Inhibit Necrosis of Lymphocytes in SAM-P8 Mice. The levels of necrosis and apoptosis in blood lymphocyte populations were analyzed by double staining with Annexin V-FITC V/PI. This revealed that the proportion of necrotic lymphocytes in SAM-P8micwas significantly higher than in SAM-R1 control animals, while the proportion of apoptotic lymphocytes was lower(Figure 7). Supplementation of the diet of SAM-P8 mice with three different doses of ECD was able to inhibit the levels of necrosis seen in peripheral lymphocyte populations, whilst only the two higher levels of ECDsupplementationproduceda significant change in the level of apoptotic lymphocytes(Figure 7).

3.6.Extracts of Cistanche deserticola(ECD) Decrease the Proinflammatory Cytokine IL-6 in SAM-P8 Mice.
As a further indicator of ECD's capacity to influence age-related changes in the immune system, the effect of ECD diet supplementation on the level of a number of cytokines(IFN-, TNF-, IL-2.IL-6.IL-10, GM-CSF, and IL-3) was analyzed using cytometric bead array analysis(CBA)which involves staining with fluorescent-dye-labeled antibodies coupled with capture cytometric beads. The results(Figure8) showed that the inflammatory cytokine IL-6 was not increased in 8-month-old SAM-P8 mice compared with control SAM-R1 mice of the same age, but it was significantly increased when com-pared with6-month-old SAM-R1 mice(results not shown). However, dietary supplementation with high and medium doses of ECD was able to produce a statistically significant decrease in plasma IL-6 level(Figures 8(a) and 8(b),<0.05
(IFN-,TNF-,IL-2,IL-10, GM-CSF,and IL-3)showedno significant variationsbetweenSAM-P8,SAM-R1,andECD treated groups of mice (data not shown).

4. Discussion
The immune defense against newly invasive microorganisms or endogenous tumor cells depends on the diversity of T cell repertoire, which in turn relies on the generation and maintenance of naive T-cells[19]. During the aging process, the diversity of the T-cell repertoire has been shown to shrink dramatically due to the progressive depletion of naive T cells in the peripheral reserve pool. It is this shortage of responsive naive T-cells that have been thought to be responsible for the susceptibility of elderly people to infection, cancer, and poor outcomes following vaccination [20]. Various interventions, including caloric restriction [7], exercise [21], and vitamin E supplementation [8], have all been used to successfully replenish naive T cells and thereby extend life span and reduce the onset of infection and cancer among the elderly. In this study, we have demonstrated that ECD is able to increase the level of naive T cells in the peripheral pool and there was a concomitant extension of life span and reduction in the frequency of tumor formation in senescence-accelerated mice(ECDtherapeutic groupversusSAM-P8,0 versus 1/6). Therefore, it seems reasonable to conclude that ECD's capacity to reverse the age-dependent depletion of peripheral naive T-cells may contribute to its known effects in reducing age-related disease which derives from long-term clinical observations.

5. Materials and Methods
5.1.Materials.
Inthenormalimmuneresponse, following the containment and elimination of a foreign antigen, most of the clonally expanded T-cells undergo apoptosis with only a small fraction being retained as memory T cells. Consequently, as immune-competent organism ages, peripheral memory T cells accumulate as a result of a lifetime of exposure to foreign anti-gens converting naive T-cells into memory cells[22,23]. A number of studies have suggested that defects of lymphocyte apoptosis in older animals and humans also contribute to the enlargement of the memory T-cell compartment [24,25]. In addition, a recent study has shown that an overabundance of peripheral memory T-cells can inhibit the homeostatic proliferation of naive T cells [26]. These recent studies have also suggested that memory T cells may secrete high levels of proinflammatory cytokines which may contribute to persistent inflammations in various tissues of the elderly. Consequently, reducing the level of memory T-cells should be beneficial both for the homeostatic proliferation of naive T cells and also in alleviating the excess inflammatory state seen in older organisms. In this study, dietary supplementation with ECD was shown to be able to reduce the level of peripheral memory T cells and increase levels of naive T cells. In addition, ECD supplementation increased apoptosis space for new naive lymphocytes. Necrosis in peripheral lymphocytes, which is mainly induced by oxidative stress in senile animals, was also inhibited.
Extensive evidence exists to indicate that aging in an organism is not only characterized by immune deficiency but also by an increase in chronic inflammation[27. Proinflammatory status is most clearly manifested in increased, and IL-1, in the circulation coupled with increased frequencies of chronic inflammatory diseases associated with aging such as Alzheimer's disease, Parkinson's diseases, and atherosclerosis. One of the most prominent inflammatory cytokinesis is IL-6 and its plasma concentration increases with aging and age-related diseases [18,28,29], and epidemiological studies suggest IL-6 is a good biomarker of longevity as it is closely related to mortality in elderly patient cohorts [30,31]. Consistent with this, a number of therapeutic interventions that decrease the IL-6 concentration are able to

Figure6:The effects of ECDdietary supplementation on stem cell antigen-1 (Sca-1) in the peripheral blood lymphocytes ofSAM-P8mice. Eight-month-old maleSAM-P8 and control SAM-RI mice were divided into 5 groups. The 3 treatment groups of SAM-P8 animals were fed for four weeks with diets supplemented with low (100mg/kg), medium 500mg/kg), and high 250 mg/kg)doses of ECDand the two control animals groups were fed on the same diet without supplementation. Following fasting for12hours, peripheral blood was collected from anesthetized animals and subjected to FACS analysis all as described in Section 5. (a)Representative FACSplots of the different groups of mice generated by gating on stem cell antigen-1 (Sca-1)positive cells.(b) Histogram showing the percentage of stem cell antigen-1 (Sca-1)positive cells in peripheral blood of the different animal groups.Barsrepresentthemeat SD(in each group,= 10).w* <0.01 SAM-P8 versus SAM-R1;** <0.01 each treated group versus SAM-P8 group (by one-way ANOVA with post hoc test).
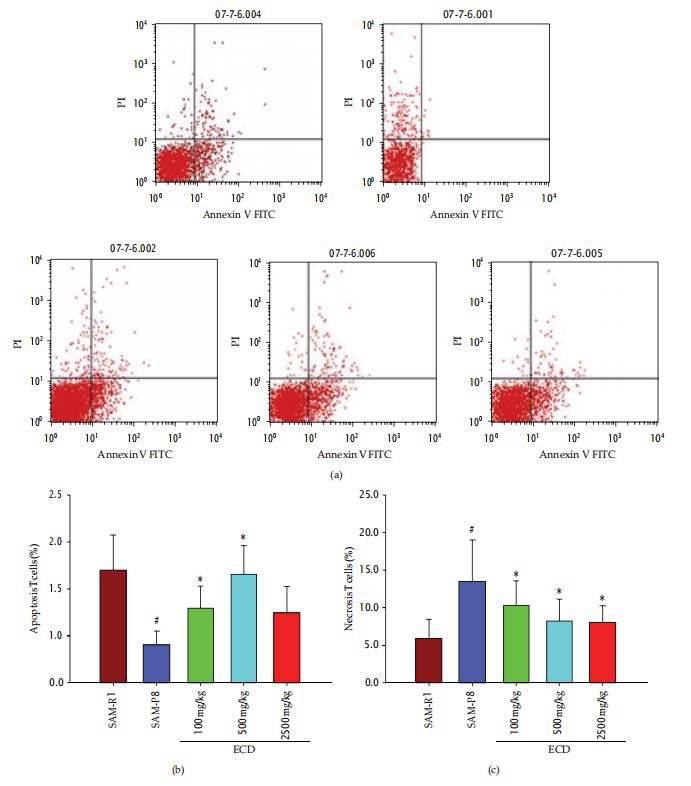
Figure 7:The effects of ECD dietary supplementation on apoptosis and necrosis of lymphocytes in aged SAM-P8 mice. Eight-month-old maleSAM-P8 and control SAM-RI mice were divided into 5 groups. The3 treatment groups of SAM-P8 animals were fed for four weeks with diets supplemented with low (100mg/kg), medium (500mg/kg), and high (2500mg/kg)doses of ECD and the two control animal groups were fed on the same diet without supplementation. Following fasting for 12 hours, peripheral blood was collected from anesthetized animals and subjected to FACS analysis all as described in Section 5. (a)Representative FACSplots of the different groups of mice generated by gating on double-stained blood lymphocytes with Annexin V-FITC/PI. (b)Histogram showing the percentage of apoptotic lymphocytes (Annexin V-FITC positive cells) in peripheral blood of the different animal groups. (c) Histogram showing the percentage of necrotic lymphocytes (PI-positive cells)in peripheral blood of the different animal groups. Bars represent the mear SD(in each group,= 10)."<0.05 SAM-P8 versusSAM-R1;*<0.05each treated groupversusSAM-P8group(by one-way ANOVA with post hoc test).

Figure 8:Effects of the ECD dietary supplementation on serum proinflammatory cytokine L6 level of SAM-P8 mice. Eight-month-old male SAM-P8 and control SAM-RI mice were divided, into 5 groups.The3 treatment groups of SAM-P8 animals were fed for four weeks with diets supplemented with low(100mg/kg), medium (500mg/kg), and high (2500mg/kg)doses of ECD, and the two control animal groups were fed on the same diet without supplementation. Following fasting for12 hours, the plasmas were collected and plasma cytokines in the different animal groups were determined by cytometric bead array immunoassay all as described in Section 5.(a) Representative EACSplots of the different groups of mice generated by gatingonIL-6cytometric bead array. (b)Histogram showing the concentrations of cytokine IL6inplasmaof the different animal groups.Barsrepresentthemean±SD(in eachgroup,= 10). <0.001each treated group versus SAM-P8 group (by one-way ANOVA with post hoc test).
alleviate age-related diseases [18]. Against this background, it was significant that in this study dietary supplementation with ECD was able to reduce peripheral IL-6 concentrations, with the implication that this effect may contribute to the underlying mechanisms of reducing the frequency of age-related diseases that have been obtained through the long-term use of ECD in clinical practice.
Alongside the changes in the adaptive immune response that occurs with age, there are also major changes in innate immunity as an organism age. Amongst these, changes in natural killer [18] cells, the first line of cellular components to provide direct cytotoxic lysis of tumor cells and virus infected cells, have been extensive since the elderly people show increased susceptibility to both tumor development and virus infections [32]. This study indicated that CD3+Ly49+ NK cells were present at significantly lower levels in senile SAM-P8 mice compared to SAM-R1 control animals, which may be another underlying factor accounting for the increased susceptibility of elderly mice to cancer and virus infection. The increase in the level of NK cells seen in this study following dietary supplementation with ECD could therefore be beneficial to elderly organisms in terms of increased resistance to tumor development and virus infection. However, further studies are required to clarify the role of the effect of ECD on NK cells in aged organisms.
In conclusion, this study has shown that treatment of the senescence-accelerated mouse prone substrain, SAM-P8, with ECD can induce a significant reversal of age-related immunosenescence alterations. Deficiencies in both peripheral and spleen cell populations of naive T-cells and NK cells were reduced, while levels of redundant memory cells were also reduced. In addition, dietary supplementation with ECD was able to suppress both necroses in peripheral lymphocytes and the levels of the proinflammatory cytokine, IL-6. Finally, in addition, to bringing about changes in the senile immune system, ECD dietary supplementation significantly prolonged the life span of the senileSAM-P8 mic.
References
[1] Y.Jiang and P-F.Tu, "Analysis of chemical constituents Cistanche species, "Journal of Chromatography A, vol.1216, no. 11, pp.1970-1979,2009.
[2] Q.Xiong K. Hase Y.Tezuka,T.Tani,T.Namba,andSKadota. "Hepatoprotective activity of phenylethanoids from Cistanche deserticola," PlartaMedica,vol.64,no.2,pp.120-125,1998.
[3]"Immunology and aging in Europe. Proceeding of the 2nd Conference on Basic Biology and Clinical Impact of Immunosenescence March 22-26, 2001, Cordoba, Spain, "Experimental Gerontology, vol.37, no.2-3, pp.183-473,2002.
[4] G.-D. Xuan and C.-Q. Liu, "Research on the effect of phenylethanoid glycosides(PEG) of the Cistanche deserticola on anti-aging in aged mice induced by D-galactose,"Journal of Chinese Medicinal Materials, vol31, no 9, pp. 1385-1388,2008.
[5] D.Aw, A.B.Silva, and D.B.Palmer, "Immunosenescence emerging challenges for an aging population," Immwmology, vol.120, no.4, pp.435-446,2007.








