Part 2:Independent Dynamics Of Low, Intermediate, And High Frequency Spectral Intracranial EEG Activities During Human Memory Formation
Mar 18, 2022
Contact: Audrey Hu audrey.hu@wecistanche.com
In general, the total induced power was significantly different be- tween the left and the right hemisphere. We investigated this laterality effect by estimating the temporal profiles of the mean power change in each ROI. While there was a significant laterality effect in at least one frequency band of every ROI (Fig.3A; t-statistic, p < 0.05), the timing and magnitude of this effect was specific to a particular brand and ROI. The temporal profiles of the induced power in the left and the
right hemisphere (Fig.3A; red and blue, resp.) provide another spectral signature of particular cortical areas. Hemisphere had an effect on the induced power in time (Fig.3B; repeated measures ANOVA, encoding phase as within-subjects factor, F = 17.01, p < 0.001, df = 3). When the total induced power was compared between the brain regions (posthoc Tukey-Kramer, p < 0.05) only the prefrontal, inferior temporal, and visual cortex showed a significant laterality effect (Fig. 2C). The effect in these three areas was significant regardless of the frequency band analyzed. Significantly more power in the left prefrontal and inferior temporal areas was expected given the role of these areas in processing verbal information.
We also observed a laterality effect in the difference between re-called and forgotten words, also known as the subsequent memory effect (Fig. 3C). Induced power was higher on trials with words that were subsequently recalled than on those that were forgotten. In the left hemisphere, this subsequent memory effect (SME) difference was increasing going from the most posterior to the anterior brain regions, reaching the peak magnitude in the anterior prefrontal cortex. The differences in the right hemisphere were relatively low and showed a peak memory effect in the MTL. Therefore, the SME confirms the hemispheric differences and overlap in the same brain regions that showed the largest laterality effect, including the inferior temporal cortex. This also highlights the importance of these higher association areas in memory function. This trend was not found in the right hemisphere, however, it is important to note that the highest right hemisphere SME was found in the MTL.

Electrodes are separated by location (including hemisphere) and frequency band of activity. The number of participants that these came from is included in bold. (FP – Frontal Pole, PFC – Prefrontal Cortex, Br – Broca’s Area, LT – Lateral Temporal, MTL – Mesial Temporal Lobe, Par – Lateral Parietal, Pre – Precuneus, IT – Inferior Temporal, V – Visual, L – Left Hemisphere, R – Right Hemisphere).

3.3. Low, intermediate, and high-frequency activities show distinct spatiotemporal dynamics
In agreement with the previous studies ( Burke et al., 2014a; Burke et al., 2013; Kucewicz et al., 2014, 2019), the spectral power induced in this task followed a temporal sequence of brain regions, showing enhanced or suppressed power at specific times of word encoding (Fig. 4). We compared these sequences across the nine ROI in the six frequency bands concerning the four phases of word encoding (PRE - before the word appears on the screen, EARLY - the first 800 ms of the word presentation, LATE - the second 800 ms the presentation, and POST - after the word disappears from the screen). First, we con- firmed, while accounting for ROI, hemisphere, and frequency band, that the spectral power was significantly modulated by the phase of memory encoding (repeated measures ANOVA, F = 729.45, p < 001, df = 3). In each band, we found that the power alternates between relative in- duction and suppression, but the timing and the ROI sequences were different across the frequency spectrum, as revealed by the different orders of ROI labels in the y-axis ofFig.4A. The low (theta), intermediate (alpha/beta), and high (gamma) frequency activities each showed a distinct pattern (Fig.4A). The intermediate frequency (alpha and beta) power was enhanced mainly in the pre-and the post-encoding phases. In contrast, the low and the high-frequency band power was induced in the early encoding phase in two sequences of activation - first at word onset in the theta bands and then following the onset in the gamma frequencies. We saw similar patterns for forgotten word trials, but at lower magnitudes of power change (Supplementary Fig. 2). There was a significant effect of the frequency band (repeated measures ANOVA F = 18.06, p < 0.001, df=5), of the brain region (ANOVA F = 15.57, p < 0.001,df = 37), and of the hemisphere location (ANOVA F = 72.72, p < 0.001, df = 1) on the peak power latency. In the gamma bands, earlier latencies were observed in the visual compared to the prefrontal cortical areas (post-hoc Tukey-Kramer, p < 0.05), which was not the case for the theta bands (Fig.4B). The high gamma power peaks occurred significantly later than the theta peaks (post-hoc Tukey-Kramer, p < 0.05) in the pre-frontal cortical areas (Fig.4C left) but not in the visual areas (Fig.4C right), where the alpha/beta peaks followed later than the high gamma peaks. Therefore, the sequences of the induced power were different, both between the low and high-frequency activities in the same cortical areas and between the cortical areas in the same frequency bands. On a more granular level, we investigated the peak latency time of each electrode plotted on brain surfaces (Fig.5A) to confirm the general posterior-to-anterior sequence (see Fig.4A), particularly in the gamma frequency bands. This general pattern is only applied to a subset of electrodes in a given cortical area. The mosaic-like pattern (seeFig. 2) of various peak latencies was observed across the cortex. Significant correlations between the peak latency and anatomical position (Pearson’s correlation) were predominantly observed in the anterior-posterior axis (Fig.5B), providing additional evidence for the anatomical sequences of the induced spectral activities. This anatomical sequence, as well as the mosaic-like pattern of individual electrode activations, was most evident in the low theta and the high gamma bands (Video 1).

Two sequences of induced power during successfully encoded words appear “moving” across the cortical space. Time bar at the top and bottom changes from black to red to indicate word display.
Finally, to provide a complete picture of this spatiotemporal dynamics across the frequency spectrum we interpolated the induced power values from all active electrodes on an average brain surface. Spectral power was predominantly induced in the posterior visual and anterior prefrontal cortical areas at the encoding phases specific for the low, intermediate, and high-frequency activities (Fig. 6 top). Induced power was first observed in the theta activities of the visual areas before word onset and then right after the onset in the anterior prefrontal ar- eas. This early posterior-to-anterior sequence of theta power induction was followed by a secondary induction of the high-frequency gamma power in the same sequence later into the word encoding. These two sequences of induced power are “moving” across the anatomical space in both hemispheres (Video 1). There was no analogous sequence observed in the alpha or the beta bands (Suppl. Video 1). The alpha/beta power was induced in the same posterior and anterior areas but predominantly before and afterword presentation, suggesting a more preparatory or in- inhibitory role in our proposed model of human memory encoding (Fig. 6 bottom). During preparation for encoding of the incoming words, the intermediate frequency oscillations would prevail and be followed by induction of the low-frequency power in the theta bands across the brain at the time of word onset. The theta rhythms would, in turn, entrain the posterior and the anterior areas for final induction of the high frequency

Fig. 4. Low, intermediate, and high-frequency activities are temporally ar- ranged into distinct sequences of cortical brain regions. (A) Mean power changes estimated from correct recall trials of all active electrodes in a given brain region of the left hemisphere are ordered from the earliest to the latest peak latency (dashed lines separate different phases of memory encoding). Notice consistent sequences of activation in the theta, the alpha/beta, and the gamma frequency bands. Areas listed on the y-axes are sorted in order of the earliest peak and thus are specific to each frequency band. (B) Summary of the mean latency ranges (post hoc ANOVA mean comparison) in the time of word presentation (0 corresponds to onset) shows significantly earlier gamma power induction in the most anterior FP areas (black) relative to the more posterior areas (red; p < 0.05, 8 Tukey-Kramer test), and an opposite pattern of the theta power induced earlier in the posterior V area relative to the more anterior brain regions (seeFig. 2for labels). (C) Anterior and posterior brain regions show analogous patterns of gamma activities during the time of word encoding, first in the visual, and then progressively later in the higher-order prefrontal cortex ar- eas (Burke et al., 2014a; Kucewicz et al., 2019). The model is proposed to link the dynamics of particular spectral activities with the associated cognitive processes involved in anticipation, attention, and encoding of the remembered stimuli.
4. Discussion
Separate anatomical and temporal distributions of the low, intermediate, and high-frequency spectral activities in the human cortex are the main findings of this work, along with the resultant simplified holistic model of the spectral power dynamics associated with human memory encoding. Because of the large number of participants and electrodes, implanted throughout all cortical regions, recording from discrete and spatially distinct sites across 39 Brodmann areas as possible. With exception of the visual cortex, within any one cortical area, a majority of the electrode sites showed task-induced activity only in one or two bands of the frequency spectrum. At the same time, activities in all six bands were found among all sites within a given area, producing a “mosaic-like” pattern of theta, alpha, beta, and gamma activities recorded at specific cortical sites. Each site could be viewed as one small tile of the mosaic with a specific color corresponding to a particular band as visualized in the Fig. 2 brain surface plots. Our results are in agreement with the spectral fingerprint view (Siegel et al., 2012) that processing in a given cortical site is characterized by specific neural activities, as previously demonstrated in the MEG and EEG studies (Fellner et al., 2019; Keitel and Gross, 2016). Various spectral activities were sparsely distributed in any one cortical region in more or less equal proportions, despite subtle differences, e.g., relatively more gamma band activities in the prefrontal cortex. This general pattern of heterogeneous, sparse distribution would explain the observation of a broadband "spectral tilt"of power across all frequencies (Burke et al., 2015; Voytek and Knight 2015; Kilner et al., 2005; Miller et al., 2014; Herweg et al., 2020) when activities from multiple sites in a given brain area were averaged together. As a result, low-frequency power (<30 Hz) is typically decreased and high-frequency power (>30 Hz) is increased upon activation of the cortical area in a given task. This broadband tilt in the spectral power can be resolved into specific spectral fingerprints at the level of individual electrode sites. One could go even further to ask how resolved this spectral distribution is and whether it can still be observed on the level of micro-electrode sites. Our study was limited to multiple macro-contacts that were grouped from different subjects - high-density recordings from
multiple macro- and micro-electrodes implanted in individual subjects (Kucewicz, Berry, Worrell chapter in (Lhatoo et al., 2019)) would be required to address these questions further and elucidate the neuronal activities underlying our reported mosaic of spectral fingerprints.
The six frequency bands were similarly represented in the cortex,as quantified by the relative proportion of electrode sites recording theta, alpha/beta, and gamma activities. Even though cortical task activation was typically associated with spectral changes in the theta and gamma frequency bands (Miller et al., 2014; Greenberg et al., 2015; Osipova et al., 2006; Solomon et al., 2017; Burke et al., 2013; Kucewicz et al., 2014), we found the same or higher number of active electrode sites in the alpha and beta bands. Oscillations in these frequency bands were previously implicated with anticipatory or even inhibitory processes preceding task activations (Spitzer and Haegens, 2017; Engel and Fries, 2010). Compared with post-stimulus gamma activities related to sensory processing, alpha and beta activities were reported before stimulus onset and modulated by attention (van Ede et al., 2014; Bauer et al., 2014). Our results confirm the differ- ent timing of alpha and beta power induction, occurring mainly before and after the period of word presentation on the screen. These were observed in anatomical areas that were overlapping with the gamma and theta power induced during word presentation. Hence, the spatiotemporal pattern of spectral power confirmed distinct roles played by the theta and gamma, as well as the alpha and beta activities.
Theta frequency bands were also found to show a profile of spatiotemporal changes that was distinct from the gamma activities. Task-induced power in the high gamma band is known to arrange into a hi hierarchical sequence of brain regions activated first in the posterior sensory areas and then in the more anterior associational temporal and pre-frontal cortex (Kucewicz et al., 2019, 2014,). This posterior-to-anterior sequence of high gamma power inspired a two-stage model of memory encoding (Burke et al., 2014a) to distinguish the early sensory and the late semantic phases of processing words. Theta power was also reported in the same areas preceding the gamma activation (Burke et al., 2013). Our recent analysis showed that this hierarchical activation from the sensory posterior areas to the anterior semantic systems propagates continuously along the ventral visual and semantic processing streams, ending in the ventrolateral prefrontal cortex of the Broca’s speech area and the frontal pole (Kucewicz et al., 2019). Here, we observed a “mosaic-like” pattern of induced power observed across the cortex in the posterior-to-anterior sequence that was different for the theta and the gamma activities (Video 1). First, a temporal sequence of theta peaks was induced at the time of stimulus onset, in contrast to the sequence of gamma peaks following the onset. In addition, the shortest latencies of the induced theta power peaks were localized in the Broca’s and pre-frontal areas even before the visual cortex. The gamma power peaks, on the other hand, revealed the shortest latencies in the visual cortex and the longest in the Broca’s area, prefrontal cortex, and the frontal pole. Hence, both the timing and the anatomical sequence of cortical areas were different for the power induced in the theta and the gamma bands. This suggests that the low and high-frequency activities are independently induced, playing different roles in stimulus processing. It remains an open question whether this sequence of peak power in our study is in any way related to the actual traveling waves evident in the spectral phase analysis (not power) of theta and alpha oscillations(Zhang et al., 2018 ), which confirmed the general posterior-to-anterior directionality of the local, cortical waves. That study focused on more local discrete cortical locations of either slow theta, high theta, or alpha phase propagation, which is different from more global sequences of power across a broad frequency range studied here. Another possible way of analyzing the directionality of spectral amplitude or power changes would be to use cross-correlations between pairs of electrode sites (Adhikari et al., 2010). Here, we focused on the independent mosaic-like pattern of sequential power inductions in different frequency bands for the benefit of a simplified holistic model.
We summarized this holistic picture of the low, intermediate, and high-frequency neural activities in a simplified model of the relative spatiotemporal dynamics during memory encoding (see Fig. 6). Alpha and beta oscillations dominated in the initial pre-stimulus presentation phase potentially related to anticipatory and attentional processes in both the posterior sensory and the anterior association areas. Theta activities are then induced in these areas, we propose, in preparation for the expected stimulus processing around the time of presentation.
Gamma activities are finally induced in response to the stimulus presentation in a sequence of the processing stream from the visual cortex to the prefrontal areas. This comprehensive view of the relative dynamics of these activities can be best captured using all dimensions of the spectral scale, anatomical space, and time of stimulus processing (see Suppl. Video 1). Functional roles of these distinct spectral activations are not the subject of this study, although they are congruent with anticipatory, attentional, preparatory, and perceptual processes proposed in the literature. In general, the lower frequency spectrum dominates in the early phases of preparation and receiving stimulus information, whereas the late phase of encoding the presented information is characterized by gamma activities when the low-frequency power is back at baseline or even suppressed. The observation that the low and high-frequency activities were not induced at the same time would not need to preclude a possible cross-frequency interaction. For instance, bursts of induced gamma power that were previously described in humans and non-human primates (Kucewicz et al., 2014, 2017; Lundqvist et al., 2016, 2018), understood in frames of various models like the theta-gamma memory buffer (Lisman and Jensen, 2013), still would be reconciled with our reported independent spatiotemporal dynamics. Our results show no evidence for amplitude-amplitude inter- actions. However, phase-phase or phase-amplitude interactions between the low and the high-frequency activities (Canolty et al., 2006) are still possible and could explain the capacity limit of the remembered items (Kamiński et al., 2011), which was observed also in our study of free recall. In conclusion, our model shows that spectral power in the three frequency ranges is induced independently in anatomical space and time of memory formation, but this does not preclude phase inter-actions between activities in these ranges, which were beyond the scope of this study.
Neural dynamics differed between the two hemispheres, revealing different profiles of spectral power, particularly in the prefrontal and the temporal cortex. The lateral prefrontal and inferotemporal cortices showed the greatest laterality effect and SME with more power in the left hemisphere, while the visual cortex had more power in the right hemisphere. In general, this hemispheric asymmetry was found to various degrees and in at least one frequency band in each brain region. Spectral fingerprints were thus specific to the hemisphere. One possible reason for this asymmetry is a differential engagement in the encoding and retrieval processes. According to the HERA (hemispheric encoding/retrieval asymmetry) model, we would expect brain activities during encoding to be stronger in the left hemisphere than in the right hemisphere and for the opposite to be true for retrieval (Buckner et al., 1996; Rugg et al., 1996; Fletcher et al., 1998b, 1998a; Grady et al., 1998). Therefore, it is not surprising that higher induced power was found in the left lateral prefrontal and inferior temporal cortex, especially in a verbal task with words. While a complimentary analysis of the induced power during retrieval of recalled words was beyond the scope of this study, others reported in the recall epoch of the same task more theta power in the right temporal and more gamma power in the left prefrontal and temporal cortex areas(Burke et al., 2014a). Previous studies with different tasks (Tulving et al., 1994; Fletcher et al., 1997) associated the right prefrontal cortex with episodic memory retrieval, and the left prefrontal cortex with encoding. Therefore, the preferential activation of the left prefrontal cortex during encoding in our task can be explained both within the HERA model and also by the serializability of the stimuli used (Golby, 2001). The latter could be further studied with the determination of language dominance for every subject, which was not possible in our study with information available for only 4 subjects (Supplementary Table 1). Obtaining and sharing this information in large, multi-center projects is challenging, and future studies will have to address this limitation. Nevertheless, in terms of the hypotheses tested in our study, these effects provide further support for independent spectral activities in the two hemispheres of particular cortical areas.

Influence of the pathophysiology of epilepsy on the laterality effect or the estimates of spectral power, in general, can only be minimized but not eliminated in this patient population. Electrode channels that showed epileptiform activities in the seizure-generating areas were assumed to be excluded from the analysis during the automated selection of active electrodes – an added benefit of the selection method (Saboo et al., 2019). Although the occurrence of epileptiform activities was shown to be related to memory processing at distinct phases and anatomical locations (Matsumoto et al., 2013; Horak et al., 2017), it is highly unlikely that these pathological activities would consistently occur at specific phases of memory encoding and thus bias the power estimates used for selecting active electrodes. Hence, the laterality or the memory effects reported here are presumed to be only minimally affected.
of verbal memory tasks. In our verbal task, the cortical areas with the greatest laterality effect were also the ones to reveal a high subsequent memory effect. As discussed previously, the strong laterality effect may be due to language lateralization in the left hemisphere, as would be expected in the majority of the general population. In the left temporal and prefrontal cortex, spectral power induced on trials with words that were subsequently recalled was greater than on the ones with words that were forgotten. This subsequent memory effect was gradually increased in magnitude with successively more anterior cortical areas. Previous- ous neuroimaging and electrophysiological studies found this memory effect to be the highest in the lateral prefrontal and the temporal cortices during similar, verbal memory tasks (Wagner et al., 1998; Long et al., 2010, 2014; Kucewicz et al., 2019; Kim, 2011; Burke et al., 2013; Sederberg et al., 2003). Our recent investigation of the memory effect in the same task localized the highest magnitude in the left ventrolateral prefrontal cortex close to the Broca’s area, and the left occipitotemporal cortex (Kucewicz et al., 2019). Altogether, the cortical areas identified with the greatest induced power and hemispheric laterality in the task overlap with localization of the memory effect in this verbal task. This pattern may be specific to verbal memory tasks, suggesting it is driven by the lateralization of language processing but not necessarily memory formation.
Despite its known involvement in declarative memory function (Beason-Held et al., 1999; Zola-Morgan et al., 1994; Parkinson et al., 1988; Squire and Zola-Morgan, 1991), MTL and the hippocampus had a relatively low number of active electrodes, the magnitude of the induced spectral power, and laterality effect. One reason for this finding is a sparser distribution of hippocampal activities than in the neocortex, which could result in effectively less induced power recorded on any one macro-contact electrode. Sparse micro-contact neural activities would be averaged out by the surrounding non-active areas, compared to more topographically mapped cortical areas. Another reason is that the task itself may engage the MTL less than a more spatial or episodic memory task. Remembering the sequence of words on the list would be expected to increase hippocampal involvement in the task. A greater neocortical contribution to this task would explain our reports of enhanced performance in this task with stimulation in the lateral temporal cortex (Kucewicz et al., 2018a; Ezzyat et al., 2018). Stimulation in MTL was
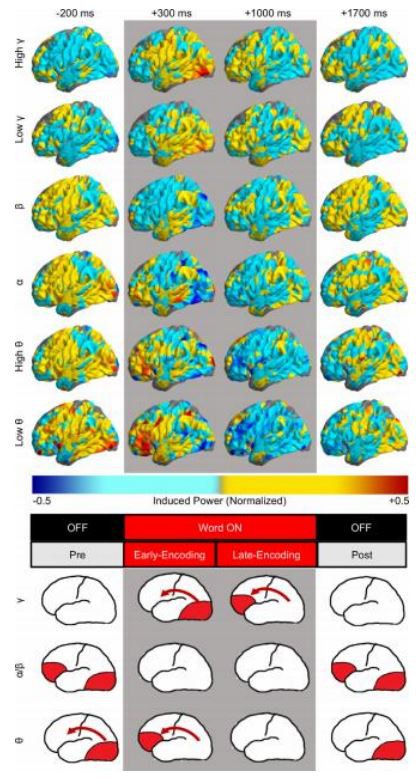
Fig. 6. Anatomical spread of low and high-frequency activities reveals independent posterior-to-anterior temporal sequences of spectral power. (Top) Average brain surface plots summarize interpolated power from all active electrodes at selected four-time points of the pre-, early, late, and post-word encoding periods (gray background indicates word presentation on the screen). Notice analogous patterns of the anatomical spread for the low (theta), intermediate (alpha/beta), and high (gamma) frequency activities, especially in posterior visual and the interior prefrontal areas. (Bottom) Model of independent activation of the low and high-frequency power “moving” from the posterior to the anterior cortical areas during word presentation, preceded and followed by the intermediate frequency alpha and beta band activation outside of the word encoding period. Red arrows indicate directions of the sequences (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article.).
found to have an opposite effect (Kucewicz et al.,2018b; Jacobs et al., 2016), suggesting different roles for the two structures in this task. And given reports of the contrary, where performance improved with MTL stimulation (Suthana and Fried, 2014; Fell et al., 2013), the roles and outcomes may differ with subtle changes within the structure as well.
Mapping neural activities that are critical for memory encoding guides the development and targeting of brain modulation, e.g. with the direct electrical stimulation, for therapeutic and research purposes. Out of the multitude of spectral activities that are induced at various times and anatomical locations, it is necessary to identify prospective targets for modulation. Our holistic map and model of independent spectral activities and their various derivatives, including the laterality or the memory effect, offer simple and accessible biomarkers for new brain-computer interface technologies. Such biomarkers can be rapidly computed to map the brain regions, times, and states for modulating brain ac- activities underlying memory and other cognitive functions. For instance,they can be used to determine the neural activities correlated with memory enhancement (Kucewicz et al.,2018b), to predict memory states for brain stimulation and rescue poor encoding trials (Ezzyat et al., 2017), or trigger modulation online in a closed-loop design for responsive electrical stimulation (Ezzyat et al., 2018). Hence, the spectral biomarkers can be useful for developing and optimizing the emerging technologies and new therapies for mapping and restoring memory functions.
VSM wrote the manuscript and analyzed the results. KVS pre-processed the data and developed the methodology. CT contributed to data processing and analysis. ML and TPT processed and analyzed the brain mapping imaging data. PN and VK contributed to data processing and analysis. GAW designed the study. MTK designed the study and wrote the manuscript. All authors contributed to manuscript edition and data analysis and/or interpretation.
Data availability statement
This work was supported by the Defense Advanced Research Project Agency grant called: “Restoring Active Memory (RAM)” under Cooperative Agreement N66001-14-2-4032, as part of the BRAIN initiative (Brain Research through Advancing Innovative Neurotechnologies). The raw data used for this analysis can be requested on the project website (http://memory.psych.upenn.edu/RAM). The views, opinions, and/or findings contained in this material are those of the authors and should not be interpreted as representing the official views or policies of the Department of Defense or the U.S. Government. Code used for identifying active electrodes can be found at https://github.com/kvsaboo/TaskActiveElectrodeIdentification. Violin plots generated using code originally written by Hol- ger Hoffmann and available on MATLAB’s file exchange (https://www.mathworks.com/matlabcentral/fileexchange/45,134- violin-plot). Other codes were available upon request.
Video 1. An early posterior-to-anterior sequence of low theta power induction is followed by a secondary induction of the high gamma power during word encoding.
Acknowledgments
Cindy Nelson and Karla Crockett assisted in participant recruitment and data collection. This work would not be possible without the dedicated effort and participation of participants and their families. Open-access datasets were originally collected as part of a BRAIN Initiative project called Restoring Active Memory (RAM) funded by the Defense Advanced Research Project Agency (DARPA; Cooperative Agreement N66001–14–2–4032). This research was supported from the First Team grant of the Foundation for Polish Science co-financed by the Euro-
pean Union under the European Regional Development Fund (Grant No. POIR.04.04.00-00-4379/17).
Supplementary materials
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.neuroimage.2021.118637.

References
Greenberg, J.A., Burke, J.F., Haque, R., Kahana, M.J., Zaghloul, K.A., 2015. Decreases in theta and increases in high-frequency activity underlie associative memory encoding. Neuroimage 114 (July), 257–263.
Hanslmayr, S., Gross, J., Klimesch, W., Shapiro, K.L., 2011. The role of oscillations in temporal attention. Brain Res. Rev. 67 (1–2), 331–343.
Hanslmayr, S., Staudigl, T., 2014. How brain oscillations form memories — a processing-based perspective on oscillatory subsequent memory effects. Neuroimage doi:10.1016/j.neuroimage.2013.05.121.
Herweg, N.A., Solomon, E.A., Kahana, M.J., 2020. Theta oscillations in human memory.
Adhikari, A.,Sigurdsson, T.,Topiwala, M.A.,Gordon, J.A., 2010. Cross-correlation of instantaneous amplitudes of field potential oscillations: a straightforward method to estimate the directionality and lag between brain areas. J. Neurosci. Methods 191 (2), 191–200.
Alotaiby, T., El-Samie, F.E.A., Alshebeili, S.A., Ahmad, I., 2015. A review of channel selection algorithms for EEG signal processing. EURASIP J. Adv. Signal Process.doi:10.1186/s13634-015-0251-9.
Bauer, M., Stenner, M.P., Friston, K.J., Dolan, R.J., 2014. Attentional modulation of al- alpha/beta and gamma oscillations reflect functionally distinct processes. J. Neurosci. Off. J. Soc. Neurosci. 34 (48), 16117–16125.
Beason-Held, L.L., Rosene, D.L., Killiany, R.J., Moss, M.B., 1999. Hippocampal formation lesions produce memory impairment in the rhesus monkey. Hippocampus 9 (5), 562–574.
Buckner, R.L.,Bandettini, P.A.,O’Craven, K.M.,Savoy, R.L.,Petersen, S.E.,Raichle, M.E., Rosen, B.R., 1996. Detection of cortical activation during averaged single trials of a cognitive task using functional magnetic resonance imaging. Proc. Natl. Acad. Sci. U.S.A. 93 (25), 14878–14883.
Burke, J.F., Zaghloul, K.A., Jacobs, J., Williams, R.B., Sperling, M.R., Sharan, A.D., Ka- Hana, M.J., 2013. Synchronous and asynchronous theta and gamma activity during episodic memory formation. J. Neurosci. doi:10.1523/jneurosci.2057-12.2013.
Burke, J.F.,Long, N.M.,Zaghloul, K.A.,Sharan, A.D.,Sperling, M.R.,Kahana, M.J., 2014a. Human intracranial high-frequency activity maps episodic memory formation in space and time. Neuroimage 85 (Pt 2), 834–843 January.
Burke, J.F.,Ramayya, A.G.,Kahana, M.J., 2015. Human intracranial high-frequency activity during memory processing: Neural oscillations or stochastic volatility? Curr. Opin. Neurobiol. 31 (April), 104–110.
Burke, J.F., Sharan, A.D., Sperling, M.R., Ramayya, A.G., Evans, J.J., Karl Healey, M., Beck, E.N., Davis, K.A., Lucas, T.H., Kahana, M.J., 2014b. Theta and high-frequency activity mark spontaneous recall of episodic memories. J. Neurosci. 34 (34), 11355–11365.
Buzsaki, G.,2006. Rhythms of the Brain. Oxford University Press.Canopy, R.T., Edwards, E., Dalal, S.S., Soltani, M., Nagarajan, S.S., Kirsch, H.E., Berger, M.S., Barbaro, N.M., Knight, R.T., 2006. High gamma power is phase-locked to theta oscillations in the human neocortex. Science 313 (5793), 1626–1628.
Crone, N.E., Sinai, A., Korzeniewska, A., 2006. High-frequency gamma oscillations and human brain mapping with electrocorticography. Prog. Brain Res. doi:10.1016/s0079-6123(06)59019-3.
Düzel, E., Penny, W.D., Burgess, N., 2010. Brain oscillations and memory. Curr. Opin. Neurobiol. doi:10.1016/j.conb.2010.01.004.
van Ede, F.,Szebényi, S.,Maris, E., 2014. Attentional modulations of somatosensory alpha, beta, and gamma oscillations dissociate between anticipation and stimulus processing. Neuroimage 97, 134–141 August.
Engel, A.K., Fries, P., 2010. Beta-band oscillations–signaling the status quo? Curr. Opin. Neurobiol. 20 (2), 156–165.
Engel, A.K., Moll, C.K.E., Fried, I., Ojemann, G.A., 2005. Invasive recordings from the human brain: clinical insights and beyond. Nat. Rev. Neurosci. doi:10.1038/nrn1585.
Izzy, Y., Kragel, J.E., Burke, J.F., Levy, D.F., Lyalenko, A., Wanda, P., O’Sullivan, L., et al., 2017. Direct brain stimulation modulates encoding states and memory performance in humans. Curr. Biol. 27 (9), 1251–1258.
Ezzyat, Y.,Wanda, P.A.,Levy, D.F.,Kadel, A.,Aka, A.,Pedisich, I.,Sperling, M.R.,et al.,2018. Closed-loop stimulation of the temporal cortex rescues functional networks and improves memory. Nat. Commun. 9 (1), 365.
Fell, J., Axmacher, N., 2011. The role of phase synchronization in memory processes. Nat. Rev. Neurosci. 12 (2), 105–118.
Fell, J., Staresina, B.P., Lam, A.T.A.Do, Widman, G., Helmstaedter, C., Elger, C.E., Ax- macher, N., 2013. Memory modulation by weak synchronous deep brain stimulation: a pilot study. Brain Stimul. 6 (3), 270–273.
Fellner, M.-.C., Gollwitzer, S., Rampp, S., Kreiselmeyr, G., Bush, D., Diehl, B., Ax- macher, N., Hamer, H., Hanslmayr, S., 2019. Spectral fingerprints or spectral tilt?
Evidence for distinct oscillatory signatures of memory formation. PLoS Biol. 17 (7), e3000403.
Fletcher, P.C., Frith, C.D., Rugg, M.D., 1997. The functional neuroanatomy of episodic memory. Trends Neurosci. doi:10.1016/s0166-2236(96)01013-2.
Fletcher, P.C., Shallice, T., Dolan, R.J., 1998a. The functional roles of the prefrontal cortex in episodic memory. I am. encoding. Brain J. Neurol. 121 (Pt 7), 1239–1248 July.
Fletcher, P.C., Shallice, T., Frith, C.D., Frackowiak, R.S., Dolan, R.J., 1998b. The functional roles of the prefrontal cortex in episodic memory. II. Retrieval. Brain J. Neurol. 121 (Pt 7), 1249–1256 July.
Fries, P., 2015. Rhythms for cognition: communication through coherence. Neuron 88 (1),220–235.
Fries, P., Nikolić, D., Singer, W., 2007. The gamma cycle. Trends Neurosci. 30 (7),309–316.
Golby, A.J., 2001. Material-specific lateralization in the medial temporal lobe and pre-frontal cortex during memory encoding. Brain doi:10.1093/brain/124.9.1841.
Grady, C.L., McIntosh, A.R., Rajah, M.N., Craik, F.I.M., 1998. Neural correlates of the episodic encoding of pictures and words. Proc. Natl. Acad. Sci. doi:10.1073/pnas.95.5.2703.
Trends Cogn. Sci. doi:10.1016/j.tics.2019.12.006, (Regul. Ed.).
Hoffmann, H., 2015. Violin.M - Simple violin Plot Using Matlab Default Kernel Density estimation. INES. The University of Bonnhhoffmann@uni-bonn.de.
Horak, P.C., Meisenhelter, S., Song, Y., Testorf, M.E., Kahana, M.J., Viles, W.D., Bu- jarski, K.A., et al., 2017. Interictal epileptiform discharges impair word recall in multiple brain areas. Epilepsia 58 (3), 373–380.
Jacobs, J., Kahana, M.J., 2010. Direct brain recordings fuel advances in cognitive electrophysiology. Trends Cogn. Sci. doi:10.1016/j.tics.2010.01.005, (Regul. Ed.).
Jacobs, J.,Miller, J.,Lee, S.Ah.,Coffey, T., Watrous, A.J., Sperling, M.R., Sharan, A., et al., 2016. Direct electrical stimulation of the human entorhinal region and hippocampus impairs memory. Neuron 92 (5), 983–990.
Jerbi, K.,Ossandón, T.,Hamamé, C.M.,Senova, S.,Dalal, S.S.,Jung, J.,Minotti, L.,et al.,2009. Task-related gamma-band dynamics from an intracerebral perspective: review and implications for surface EEG and MEG. Hum. Brain Mapp. 30 (6), 1758–1771.
Johnson, E., Kam, J., Tzovara, A., Knight, R.T., 2020. Insights into human cognition from intracranial EEG: a review of audition, memory, internal cognition, and causality. J. Neural Eng. doi:10.1088/1741-2552/abb7a5, September.
Kahana, M.J.,2014. Foundations of Human Memory. Oxford University Press.
Kamiński, J., Brzezicka, A., Wróbel, A., 2011. Short-term memory capacity (7 ± 2) pre- dictated by theta to gamma cycle length ratio. Neurobiol. Learn. Mem. 95 (1), 19–23.
Keitel, A., Gross, J., 2016. Individual human brain areas can be identified from their characteristic spectral activation fingerprints. PLoS Biol. 14 (6), e1002498.
Kilner, J.M.,Mattout, J.,Henson, R.,Friston, K.J., 2005. Hemodynamic correlates of EEG: a heuristic. Neuroimage 28 (1), 280–286.
Kim, H., 2011. Neural activity that predicts subsequent memory and forgetting: a meta-analysis of 74 fMRI studies. Neuroimage 54 (3), 2446–2461.
Klimesch, W., 1996. Memory processes, brain oscillations, and EEG synchronization. Int. J. Psychophysiol. doi:10.1016/s0167-8760(96)00057-8.
Klimesch, W., Sauseng, P., Hanslmayr, S., 2007. EEG alpha oscillations: the inhibition–timing hypothesis. Brain Res. Rev. doi:10.1016/j.brainresrev.2006.06.003.
Kucewicz, M.T., Berry, B.M., Kremen, V., Brinkmann, B.H., Sperling, M.R., Jobst, B.C., Gross, R.E.,et al., 2017. Dissecting gamma frequency activity during human memory processing. Brain J. Neurol. 140 (5), 1337–1350.
Kucewicz, M.T., Berry, B.M., Kremen, V., Miller, L.R., Khadjevand, F., Ezzyat, Y., Stein, J.M., et al., 2018a. Electrical stimulation modulates the high activity and human
memory performance. eNeuro 5 (1). doi:10.1523/ENEURO.0369-17.2018.
Kucewicz, M.T., Berry, B.M., Miller, L.R., Khadjevand, F., Ezzyat, Y., Stein, J.M., Kre- men, V., et al., 2018b. Evidence for verbal memory enhancement with electrical brain stimulation in the lateral temporal cortex. Brain J. Neurol. 141 (4), 971–978.
Kucewicz, M.T.,Cimbalnik, J.,Matsumoto, J.Y.,Brinkmann, B.H.,Bower, M.R.,Vasoli, V., Sulc, V., et al., 2014. High-frequency oscillations are associated with cognitive processing in human recognition memory. Brain J. Neurol. 137 (Pt 8), 2231–2244.
Kucewicz, M.T., Saboo, K., Berry, B.M., Kremen, V., Miller, L.R., Khadjevand, F., In-man, C.S., et al., 2019. Human verbal memory encoding is hierarchically distributed in
a continuous processing stream. eNeuro 6 (1). doi:10.1523/ENEURO.0214-18.2018.
Lachaux, J.-.P., Axmacher, N., Mormann, F., Halgren, E., Crone, N.E., 2012. High-frequency neural activity and human cognition: past, present and possible future of in- intracranial EEG research. Prog. Neurobiol. 98 (3), 279–301.
Lhatoo, S.D., Kahane, P., Luders, H.O., 2019. Invasive Studies of the Human Epileptic Brain: Principles and Practice. Oxford University Press, USA.
Lin, J.-.J., Rugg, M.D., Das, S., Stein, J., Rizzuto, D.S., Kahana, M.J., Lega, B.C., 2017. Theta band power increases in the posterior hippocampus predict successful episodic memory encoding in humans. Hippocampus 27 (10), 1040–1053.
Lisman, J.E., Jensen, O.,2013. The - neural code. Neuron 77 (6), 1002–1016.
Long, N.M., Burke, J.F., Kahana, M.J., 2014. Subsequent memory effect in intracranial and scalp EEG. Neuroimage 84 (January), 488–494.
Long, N.M.,Oztekin, I.,Badre, D., 2010. Separable prefrontal cortex contributions to free recall. J. Neurosci. 30 (33), 10967–10976.
Lundqvist, M., Herman, P., Warden, M.R., Brincat, S.L., Miller, E.K., 2018. Gamma and beta bursts during working memory readout suggest roles in its volitional control. Nat. Commun. 9 (1), 394.
Lundqvist, M., Rose, J., Herman, P., Brincat, S.L., Buschman, T.J., Miller, E.K., 2016. Gamma and beta bursts underlie working memory. Neuron 90 (1), 152–164.
Matsumoto, J.Y., Stead, M., Kucewicz, M.T., Matsumoto, A.J., Peters, P.A., Brinkmann, B.H., Danstrom, J.C., et al., 2013. Network oscillations modulate
interictal epileptiform spike rate during human memory. Brain J. Neurol. 136 (Pt 8), 2444–2456.
Michalareas, G., Vezoli, J., van Pelt, S., Schoffelen, J.-.M., Kennedy, H., Fries, P., 2016. Alpha-beta and gamma rhythms subserve feedback and feedforward influences among human visual cortical areas. Neuron 89 (2), 384–397.
Miller, KJ.,Honey, C.J.,Hermes, D.,Rao, R.P.N.,denNijs, M.,Ojemann, J.G., 2014. Broadband changes in the cortical surface potential track activation of functionally diverse neuronal populations. Neuroimage 85 (Pt 2), 711–720 January.
Mitra, P., 2007. Observed brain dynamics. Oxford University Press doi:10.1093/prof:oso/9780195178081.001.0001.
Nyhus, E., Curran, T., 2010. Functional role of gamma and theta oscillations in episodic memory. Neurosci. Biobehav. Rev. 34 (7), 1023–1035.
Osipova, D., Takashima, A., Oostenveld, R., Fernández, G., Maris, E., Jensen, O., 2006. Theta and gamma oscillations predict encoding and retrieval of declarative memory. J. Neurosci. 26 (28), 7523–7531.
Parkinson, J.K., Murray, E.A., Mishkin, M., 1988. A selective mnemonic role for the hippocampus in monkeys: memory for the location of objects. J. Neurosci. 8 (11), 4159–4167.
Rugg, M.D., Fletcher, P.C., Frith, C.D., Frackowiak, R.S., Dolan, R.J., 1996. Differential activation of the prefrontal cortex in successful and unsuccessful memory retrieval. Brain J. Neurol. 119 (Pt 6), 2073–2083 December.
Saboo, K.V., Varatharajah, Y., Berry, B.M., Kremen, V., Sperling, M.R., Davis, K.A., Jobst, B.C., et al., 2019. Unsupervised machine-learning classification of electro- physiologically active electrodes during human cognitive task performance. Sci. Rep. doi:10.1038/s41598-019-53925-5.
Sauseng, P., Griesmayr, B., Freunberger, R., Klimesch, W., 2010. Control mechanisms in working memory: a possible function of EEG theta oscillations. Neurosci. Biobehav.Rev. doi:10.1016/j.neubiorev.2009.12.006.
Schmidt, R., Ruiz, M.H., Kilavik, B.E., Lundqvist, M., Starr, P.A., Aron, A.R., 2019. Beta oscillations in working memory, executive control of movement and thought, and sensorimotor function. J. Neurosci. 39 (42), 8231–8238.
Sederberg, P.B., Kahana, M.J., Howard, M.W., Donner, E.J., Madsen, J.R., 2003. Theta and gamma oscillations during encoding predict subsequent recall. J. Neurosci. 23 (34), 10809–10814.
Sheehan, T.C., Sreekumar, V., Inati, S.K., Zaghloul, K.A., 2018. Signal complexity of human intracranial EEG tracks successful associative-memory formation across individuals. J. Neurosci. 38 (7), 1744–1755.
Siegel, M.,Donner, T.H., Engel, A.K., 2012. Spectral fingerprints of large-scale neuronal interactions. Nat. Rev. Neurosci. 13 (2), 121–134.
Singer, W., 1999. Neuronal synchrony: a versatile code for the definition of relations?”.
Solomon, E.A., Kragel, J.E., Sperling, M.R., Sharan, A., Worrell, G., Kucewicz, M., In-man, C.S., et al., 2017. Widespread theta synchrony and high-frequency desynchronization underlie enhanced cognition. Nat. Commun. 8 (1), 1704.
Spitzer, B., Hagens, S., 2017. Beyond the status quo: a role for beta oscillations in endogenous content (Re) activation. eNeuro 4 (4). doi:10.1523/ENEURO.0170-17.2017.
Squire, L.R., Zola-Morgan, S., 1991. The medial temporal lobe memory system. Science 253 (5026), 1380–1386.
Staresina, B.P., Michelmann, S., Bonnefond, M., Jensen, O., Axmacher, N., Fell, J., 2016. Hippocampal pattern completion is linked to gamma power increases and alpha power decreases during recollection. Elife 5 (August). doi:10.7554/eLife.17397.
Susana, N., Fried, I., 2014. Deep brain stimulation for enhancement of learning and memory. Neuroimage 85 (Pt 3), 996–1002 January.
Tallon-Baudry, C.,Bertrand, O., 1999. Oscillatory gamma activity in humans and its role in object representation. Trends Cogn. Sci. 3 (4), 151–162 (Regul. Ed.).
Tulving, E., Kapur, S., Craik, F.I., Moscovitch, M., Houle, S., 1994. Hemispheric encoding/retrieval asymmetry in episodic memory: positron emission tomography findings. Proc. Natl. Acad. Sci. U.S.A. 91 (6), 2016–2020.
Voytek, B., Knight, R.T., 2015. Dynamic network communication as a unifying neural basis for cognition, development, aging, and disease. Biol. Psychiatry 77 (12), 1089–1097.
Wagner, A.D.,Schacter, D.L.,Rotte, M.,Koutstaal, W.,Maril, A.,Dale, A.M.,Rosen, B.R., Buckner, R.L., 1998. Building memories: remembering and forgetting of verbal experiences as predicted by brain activity. Science 281 (5380), 1188–1191.
Zhang, H., Watrous, A.J., Patel, A., Jacobs, J., 2018. Theta and alpha oscillations are traveling waves in the human neocortex. Neuron 98 (6), 1269–1281.
Zola-Morgan, S., Squire, L.R., Ramus, S.J., 1994. The severity of memory impairment in monkeys as a function of locus and extent of damage within the medial temporal lobe memory system. Hippocampus 4 (4), 483–495.






