Part 2: The current gaps and future of the quality of care for Acute Kidney Disease
Mar 03, 2022
Part 2: Quality of Care for Acute Kidney Disease: Current Knowledge Gaps and Future Directions
Contact: emily.li@wesictanche.com
For Part 1. please click here.
Weight Assessment
In contrast to the paradigm of dry weight challenges for ESRD patients, acute kidney injury-D patients may require a mild permissive hypervolemia approach that emphasizes avoiding intradialytic hypotension (IDH). The dangers of IDH and high ultrafiltration rates (>13 ml/kg/h) in ESRD patients have been well-described.42 In acute kidney injury-D, IDH may exacerbate ischemic kidney injury and decrease the chances of recovery.43 Two separate retrospective studies found that higher ultrafiltration rates and more frequent IDH were associated with a lower likelihood of recovery in outpatients with acute kidney injury- D.43,44 At a minimum, patients with acute kidney injury-D should have a regular (weekly) reassessment of target weight and fluid removal goals. In non-anuric patients, diuretics may be helpful to limit interdialytic weight gain and decrease ultrafiltration rates.45 The role of specific measures to prevent IDH, such as cooled dialysate or sodium modeling, has not been formally assessed in the acute kidney injury setting.
Access
Nearly all patients with acute kidney injury-D will have a central venous catheter as primary access. Furthermore, the placement of arteriovenous access should be delayed (appropriately) while monitoring for kidney function recovery.46 These patients and their families will be at significant risk for infectious complications, and they should receive proper central venous catheter care and education before hospital discharge, which should continue as an outpatient. Although limited data exist regarding central venous catheter issues in those with acute kidney injury-D, much can be learned from investigations in other populations with central venous catheters.47,48 Patients with acute kidney injury-D should receive training in vein preservation. The delivery of these educational components is an excellent example of an important and easy quality measure for health systems and dialysis facilities to track.

Cistanche protects kidneys and improves kidney functions
Teaching
By definition, acute kidney injury-D is a potentially reversible condition, and studies suggest that upward of 40% of patients with acute kidney injury-D who are discharged on dialysis may recover to dialysis independence.44,49,50 Patients and their caregivers need to be educated to monitor for kidney function recovery. Many patients with acute kidney injury-D will not have had the benefit of nephrology care before the acute illness, so broader education regarding kidney health should be included when appropriate.14,46 In the dialysis facility, patients with acute kidney injury-D should be identifiable separately from ESRD patients. Patients with dialysis-requiring acute kidney injury should undergo a regular care plan meeting (similar to the ESRD process) that articulates the individual's clinical trajectory and personalized expectations for kidney recovery.
Clearance
We believe dialysis facilities caring for patients with acute kidney injury-D need to have the appropriate processes to allow for more frequent blood and urine testing, assessing for kidney recovery.46 This can be done through blood work (e.g., trending predialysis SCr) and timed urine collections, and should be pursued at least weekly during the outpatient transition.51
Dialysis clearance should also be measured and used to guide prescription. Evidence from the large-scale, randomized controlled acute tubular necrosis trial suggests that for patients with acute kidney injury-D, a delivered Kt/tv urea of 1.2 thrice weekly can be considered adequate dialytic clearance.52 However, patients with acute kidney injury-D may have more individualized needs, such as higher catabolism requiring more dialysis for symptomatic control of uremia. Conversely, patients who begin to recover kidney function may tolerate a tapering of dialysis to less than 3 weekly treatments.
Cistanche avoids acute kidney disease.
Hypotension
Intradialytic hypotension is associated with adverse outcomes and may decrease the likelihood of kidney function recovery in patients with acute kidney injury-D.43,44 Emphasizing the importance of limiting interdialytic weight gain is essential, as is the careful adjustment of antihypertensive medications.
Medications
As with any transition in health care settings, the move to outpatient acute kidney injury-D management should be accompanied by a review and reconciliation of medications.53,54 The review and reconciliation process needs to be frequently repeated, especially when kidney function begins to recover, to ensure adequate and appropriate dosing of medications.20 Patients should be educated regarding the potential impact of kidney failure and dialysis on drug clearances, but they should also be instructed that as kidney function begins to recover, drug clearances may be improved and medication dosing may need to be increased or even discontinued. A specific review of common nephrotoxic medications and KENDS should also be performed.20
In addition to these potential measures that focus on processes of care, ideally, outcomes measures such as hospital readmission rates and dialysis independence recovery rates should be monitored. However, there are currently no established benchmarks, and these rates will vary based on patient characteristics. Given the dearth of data in this area, monitoring these rates can help to identify trends and potential quality improvement opportunities.

Cistanche deserticola protect kidneys and improve kidney functions
Conclusions
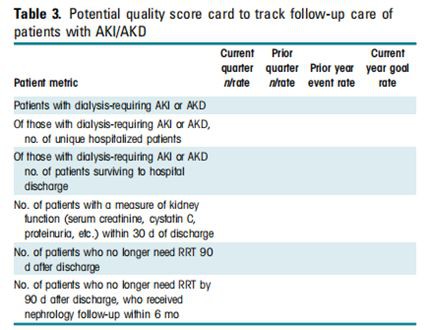
Quality measures are needed for the management of acute kidney injury and acute kidney disease patients after the index hospitalization to standardize care and improve patient outcomes. Relatively few patients receive follow-up laboratory testing of kidney function or post-acute kidney injury and acute kidney disease care. Table 3 provides a potential initial quality scorecard for a health care system to begin to monitor and improve acute kidney injury and acute kidney disease care. Although this tool has not been validated, it is based on limited published data and expert opinion. It captures the basic numbers an institution will need to track in the short and long term to care for patients with acute kidney injury and acute kidney disease. Similarly, Supplementary Appendix A and Supplementary Figure S1 provide a hypothetical quality improvement project related to the care of patients with acute kidney injury and acute kidney disease.
In the future, outcomes such as 30-day readmission rates, catheter-associated infection rates, and short- and long-term mortality after an episode of acute kidney injury or acute kidney disease may be quality metrics that will be reported. For now, health care institutions should be aware of the number of patients who warrant post-acute kidney injury or acute kidney disease care as well as track those who go on to receive some form of follow-up. This focused follow-up care could be provided by either nephrology or non-nephrology providers, and this may be an important metric to track. Future quality improvement work and research could focus on the optimal management strategies and clinical effectiveness of the KAMPS and Weight Assessment, Access, Teaching, Clearance, Hypotension, and Medications bundle components and the potential development and validation of new bundles.
DISCLOSURE
JLK is a consultant for Astute Medical, Sphingotec, and Baxter and receives research support from NxStage, Astute Medical, and Satellite Health Care. KDL is a consultant for Durect, Quark, and Portero; is on the advisory board for AstraZeneca; is a speaker for Baxter, and is a stockholder of Amgen. LGF receives research support from Baxter, La Jolla Pharmaceuticals, and Ortho-Clinical-Diagnostics and receives honoraria from Baxter, La Jolla Pharmaceuticals, Ortho-Clinical-Diagnostics, MediBeacon, and Fresenius. JAK is a consultant for Adrenomed, Astute Medical, Baxter, bio- Mérieux, Bioporto, Davita, Fresenius, Mallinckrodt, Novartis, NxStage, Potrero, RenalSense, and Sphingotech and receives grant support from Astute Medical, Baxter, bioMérieux, and RenalSense. MH is a consultant and lecturer for Abbott, Alere, Baxter, Novartis, Siemens, Roche, and FastBiomedical and receives grant support from Abbott Diagnostics. RLM is on the advisory board for Astute Medical, Baxter, Mallinckrodt, and Indalo Therapeutics and received research funds from Fresenius, Fre-series-Kabi, Grifols, and Relypsa. MHR reports honoraria from the American Society of Nephrology and Baxter and serves on data safety monitoring boards for Retrophin and Reata Pharmaceuticals. All other authors declared no competing interests.
SUPPLEMENTARY MATERIAL
Supplementary File (PDF)
Acknowledgments: The ADQI XXII Participants.
Appendix A. Sample Quality Improvement Project
Figure S1. Pareto chart for analysis of the lack of appropriate medication list review at discharge and communication with primary physicians. acute kidney injury; Cum, Cumulative.

Cistanche is a kidney fatigue supplement.
REFERENCES
1. Chertow GM, Burdick E, Honour M, et al. Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol. 2005;16:3365–3370.
2. Hobson C, Ozrazgat-Baslanti T, Kuxhausen A, et al. Cost and mortality associated with postoperative acute kidney injury. Ann Surg. 2015;261:1207–1214.
3. Meersch M, Schmidt C, Hoffmeier A, et al. Prevention of cardiac surgery-associated acute kidney injury by implementing the KDIGO guidelines in high-risk patients identified by biomarkers: the
Private kidney injury randomized controlled trial. Intensive Care Med. 2017;43:1551–1561.
4. Gocze I, Jauch D, Gotz M, et al. Biomarker-guided intervention to prevent acute kidney injury after major surgery: the prospective randomized BigpAK study. Ann Surg. 2018;267: 1013–1020.
5. Selby NM, Casula A, Lamming L, et al. An organizational-level program of intervention for acute kidney injury: a pragmatic stepped wedge cluster randomized trial. J Am Soc Nephrol. 2019;30:505–515.
6. Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. KDIGO Clinical Practice Guideline for Acute Kidney Injury. Kidney Int. 2012;(suppl):1–138.
7. Chawla LS, Bellomo R, Bihorac A, et al. Acute kidney disease and renal recovery: consensus report of the Acute Disease Quality Initiative (ADQI) 16 Workgroup. Nat Rev Nephrol. 2017;13:241–257.
8. Kashani K, Rosner MH, Haase M, et al. Quality improvement goals for acute kidney injury. Clin J Am Soc Nephrol. 2019;14: 941–953.
9. Sawhney S, Marks A, Fluck N, et al. Post-discharge kidney function is associated with subsequent ten-year renal progression risk among survivors of acute kidney injury. Kidney Int. 2017;92:440–452.
10. Siew ED, Peterson JF, Eden SK, et al. Outpatient nephrology referral rates after acute kidney injury. J Am Soc Nephrol. 2012;23:305–312.
11. Karsanji DJ, Pannu N, Manns BJ, et al. Disparity between nephrologists’ opinions and contemporary practices for community follow-up after acute kidney injury hospitalization. Clin J Am Soc Nephrol. 2017;12:1753–1761.
12. Mishra PK, Luckraz H, Nandi J, et al. Long-term quality of life post-acute kidney injury in cardiac surgery patients. Ann Card Anaesth. 2018;21:41–45.
13. James MT, Pannu N, Hemmelgarn BR, et al. Derivation and external validation of prediction models for advanced chronic kidney disease following acute kidney injury. JAMA. 2017;318:1787–1797.
14. Harel Z, Wald R, Bargman JM, et al. Nephrologist follow-up improves all-cause mortality of severe acute kidney injury survivors. Kidney Int. 2013;83:901–908.
15. Sawhney S, Marks A, Fluck N, et al. Acute kidney injury as an independent risk factor for unplanned 90-day hospital readmissions. BMC Nephrol. 2017;18:9.
16. Silver SA, Harel Z, McArthur E, et al. 30-day readmissions after an acute kidney injury hospitalization. Am J Med. 2017;130, 163.e164–172.e164.
17. Lee BJ, Hsu CY, Parikh RV, et al. Non-recovery from dialysis- requiring acute kidney injury and short-term mortality and cardiovascular risk: a cohort study. BMC Nephrol. 2018;19:134.
18. Bansal N, Matheny ME, Greevy RA Jr, et al. Acute kidney injury and risk of incident heart failure among US veterans. Am J Kidney Dis. 2018;71:236–245.
19. Hsu CY, Chinchilli VM, Coca S, et al. Post-acute kidney injury proteinuria and subsequent kidney disease progression: the Assessment, Serial Evaluation, and Subsequent Sequelae in Acute Kidney Injury (ASSESS-acute kidney injury) study. JAMA Intern Med. 2020;180:402–410.
20. Ostermann M, Chawla LS, Forni LG, et al. Drug management in acute kidney disease – report of the Acute Disease Quality Initiative XVI meeting. Br J Clin Pharmacol. 2018;84:396–403.
21. Wu VC, Wu CH, Huang TM, et al. Long-term risk of coronary events after acute kidney injury. J Am Soc Nephrol. 2014;25:595–605.
22. Wu VC, Yang SY, Lin JW, et al. Kidney impairment in primary aldosteronism. Clin Chim Acta. 2011;412:1319–1325.
23. Lai CF, Wu VC, Huang TM, et al. Kidney function decline after a non-dialysis-requiring acute kidney injury is associated with higher long-term mortality in critically ill survivors. Crit Care. 2012;16:R123.
24. Wu VC, Shiao CC, Chang CH, et al. Long-term outcomes after dialysis-requiring acute kidney injury. Biomed Res Int. 2014;2014:365186.
25. Shiao CC, Wu PC, Huang TM, et al. Long-term remote organ consequences following acute kidney injury. Crit Care. 2015;19:438.
26. US Renal Data System. Acute kidney injury. Available at: https://www.usrds.org/2016/view/v1_05.aspx. Accessed April 15, 2019.
27. Matheny ME, Peterson JF, Eden SK, et al. Laboratory test surveillance following acute kidney injury. PloS One. 2014;9: e103746.
28. Cheng SY, Chou YH, Liao FL, et al. Losartan reduces ensuing chronic kidney disease and mortality after acute kidney injury. Sci Rep. 2016;6:34265.
29. Brar S, Ye F, James MT, et al. Association of angiotensin-converting enzyme inhibitor or angiotensin receptor blocker use with outcomes after acute kidney injury. JAMA Intern Med. 2018;178:1681–1690.
30. Lin YF, Chen L, Lin SL, et al. Potential target-organ protection of mineralocorticoid receptor antagonist in acute kidney disease. J Hypertens. 2019;37:125–134.
31. Hsu CY, Hsu RK, Yang J, et al. Elevated BP after acute kidney injury. J Am Soc Nephrol. 2016;27:914–923.
32. Ftouh S, Thomas M. Acute kidney injury: summary of NICE guidance. BMJ. 2013;347:f4930.
33. Lapi F, Azoulay L, Yin H, Nessim SJ, Suissa S. Concurrent use of diuretics, angiotensin-converting enzyme inhibitors, and angiotensin receptor blockers with non-steroidal anti-in- flammatory drugs and risk of acute kidney injury: a nested case-control study. BMJ. 2013;346, e8525.
34. Silver SA, Goldstein SL, Harel Z, et al. Ambulatory care after acute kidney injury: an opportunity to improve patient outcomes. Can J Kidney Health Dis. 2015;2:36.
35. Wu CL, Kor CT, Chang CC, et al. Association of statin use with mortality after dialysis-requiring acute kidney injury: a population-based cohort study. Mayo Clinic Proc. 2018;93: 1474–1483.
36. Huang TM, Wu VC, Lin YF, et al. Effects of statin use in advanced chronic kidney disease patients. J Clin Med. 2018;7:285.
37. Chen CY, Wu VC, Lin CJ, et al. Improvement in mortality and end-stage renal disease in patients with type 2 diabetes after acute kidney injury who are prescribed dipeptidyl peptidase-4 inhibitors. Mayo Clinic Proc. 2018;93:1760–1774.
38. Tsai IJ, Lai TS, Shiao CC, et al. Proton-pump inhibitors augment the risk of major adverse cardiovascular events and end-stage renal disease in patients with acute kidney injury after temporary dialysis. Clin Pharmacol Ther. 2020;107: 1434–1445.
39. Mehboob A, Zimmerman R, Abramson S, Parker MG. Quality measures in acute kidney injury. Curr Opin Nephrol Hyper- tens. 2018;27:130–135.
40. Rewa O, Mottes T, Bagshaw SM. Quality measures for acute kidney injury and continuous renal replacement therapy. Curr Opin Crit Care. 2015;21:490–499.
41. Heung M, Faubel S, Watnick S, et al. Outpatient dialysis for patients with acute kidney injury: a policy approach to improving care. Clin J Am Soc Nephrol. 2015;10:1868–1874.
42. Chou JA, Kalantar-Zadeh K, Mathew AT. A brief review of intradialytic hypotension with a focus on survival. Semin Dial. 2017;30:473–480.
43. McAdams M, Ortiz-Soriano V, Jordan M, et al. Kidney recovery in patients discharged to an acute rehabilitation facility with acute kidney injury requiring hemodialysis. Clin Nephrol. 2019;92:15–24.
44. Pajewski R, Gipson P, Heung M. Predictors of posthospitalization recovery of renal function among patients with acute kidney injury requiring dialysis. Hemodial Int. 2018;22:66–73.
45. Sibbel S, Walker AG, Colson C, et al. Association of continuation of loop diuretics at hemodialysis initiation with clinical outcomes. Clin J Am Soc Nephrol. 2019;14:95–102.
46. Cerdá J, Liu KD, Cruz DN, et al. Promoting kidney function recovery in patients with acute kidney injury requiring RRT. Clin J Am Soc Nephrol. 2015;10:1859–1867.
47. Tan SH, Lee KC, Chao YF, Hsu LF, Lin PC. Effects of a family involvement program in patients with central-line insertion. Clin Nurs Res. 2015;24:253–268.
48. DeLa Cruz RF, Caillouet B, Guerrero SS. Strategic patient education program to prevent catheter-related bloodstream infection. Clin J Oncol Nurs. 2012;16:E12–E17.
49. Hickson LJ, Chaudhary S, Williams AW, et al. Predictors of outpatient kidney function recovery among patients who initiate hemodialysis in the hospital. Am J Kidney Dis. 2015;65:592–602.
50. Gautam SC, Brooks CH, Balogun RA, et al. Predictors and outcomes of post-hospitalization dialysis-dependent acute kidney injury. Nephron. 2015;131:185–190.
51. Milutinovic J, Cutler RE, Hoover P, Meijsen B, Scribner BH. Measurement of residual glomerular filtration rate in the patient receiving repetitive hemodialysis. Kidney Int. 1975;8: 185–190.
52. VA/NIH Acute Renal Failure Trial Network, Palevsky PM, Zhang JH, et al. Intensity of renal support in critically ill patients with acute kidney injury. N Engl J Med. 2008;359: 7–20.






