Part Ⅱ Efficacy And Safety Of A Low-sodium Diet And Spironolactone in Patients With Stage 1-3a Chronic Kidney Disease: A Pilot Study
May 12, 2023
Results
1. Patient disposition and characteristics
We assessed 452 patients for eligibility. Of these, 368 were excluded because of no signed informed consent (n=42), current steroid or immunosuppressant therapy (n=300), advanced tumors (n sent for review (n =5), remote residence inconven- =11), and other reasons (n=10). A total of 84 patients were randomized into three groups with 28 patients per group and started the run-in period, with seven withdrawing. Finally, 77 patients started the intervention period, including 27, 26, and 24 in the low-sodium+placebo, medium-sodium+SPL, and low-sodium+SPL groups, respectively. During the intervention period, two patients and one patient withdrew from the medium-sodium+SPL group and low-sodium+SPL group, respectively. A total of 74 patients completed the 12 weeks of intervention (Fig. 1).
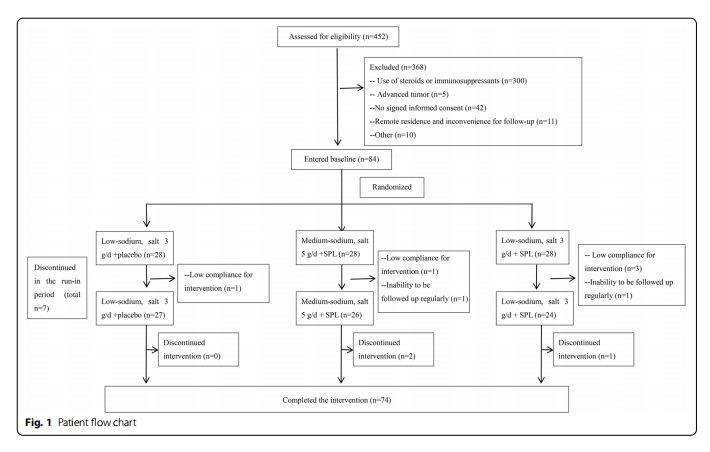
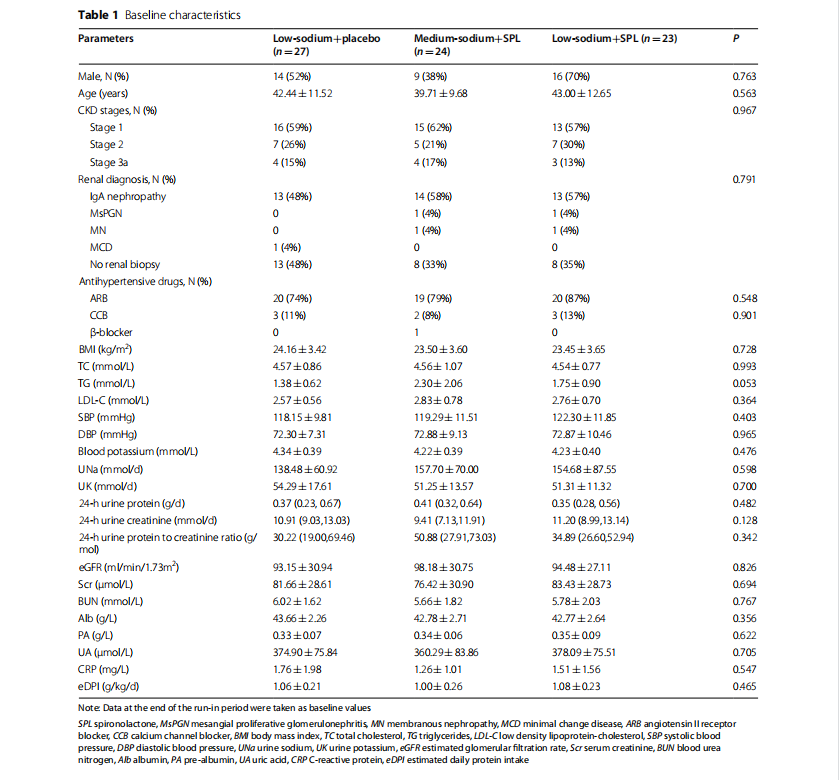
These 74 patients consisted of 39 males and 35 females. Their diagnoses included: 40 patients with IgA nephropathy, two patients with membranous nephropathy, two patients with mesangial proliferative glomerulonephritis, one patient with minimal change disease, and 29 patients with chronic glomerulonephritis not otherwise specified. Sixty-three patients were stage 1–2 CKD, and 11 patients were stage 3a CKD. There were two patients in the lowsodium+SPL group who had diabetes (one patient had a history of diabetes for five years and another for three years). There were no significant differences in sex, age, body mass index (BMI), 24-h urine protein, eGFR, Scr, BUN, blood pressure, nutritional, metabolic, inflammatory, and other biomarkers among the three groups at baseline (Table 1).
As for the salt levels of patients in this study, the median (range) of 24h-UNa levels at 4, 8, and 12 weeks fluctuated between 115 (93,150) mmol/d and 125 (91,156) mmol/d, 145 (110,217) mmol/d and 153 (117,190) mmol/d, and 114 (86,173) mmol/d and 126 (94,169) mmol/d in the low-sodium+placebo, medium-sodium+SPL, and low-sodium+SPL groups, respectively. Supplementary Table S1 shows that there were no differences in 24h-UNa among the three groups at baseline (P=0.598), but that the 24h-UNA was significantly higher in the medium-sodium+SPL group compared with the lowsodium+placebo (P=0.024) and low-sodium+SPL group (P=0.007). Supplementary Table S2 shows that there were no differences in Una/Cr among the three groups at baseline (P=0.303), but that the UNA/Cr was significantly higher in the medium-sodium+SPL group compared with the low-sodium+placebo (P=0.011) and low-sodium+SPL group (P=0.006).


Click here to buy Cistanche supplements
2. Efficacy
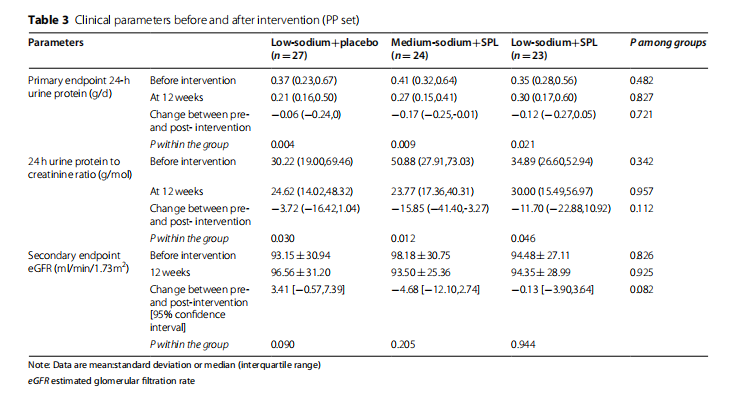
After 12 weeks of intervention, as the primary endpoint (ITT set), 24-h urine protein decreased from 0.37 (0.23, 0.70) to 0.23 (0.16, 0.51) in the low-sodium+placebo group (P=0.004). Te 24-h urine protein decreased from 0.44 (0.33, 0.71) to 0.29 (0.17, 0.50) in the mediumsodium+SPL group (P=0.020). Te 24-h urine protein decreased from 0.35 (0.26, 0.73) to 0.31 (0.22, 0.60) in the low-sodium+SPL group (P=0.013). There were no significant differences among the three groups in 24-h urine protein amount changes after intervention from pretreatment values (P=0.760), as shown in Table 2.

In the low-sodium+placebo group, the 24-h urine protein to creatine ratio was decreased from 30.54 (18.83, 77.38) to 25.40 (14.12, 50.71) (P=0.012). Meanwhile, the 24-h urine protein to creatine ratios were decreased from 54.00 (28.77, 81.30) to 24.72 (18.49, 44.10), and from 33.12 (25.90, 58.68) to 29.20 (15.69, 62.39) in the medium-sodium+SPL (P=0.011) and low-sodium+SPL (P=0.026) groups, respectively. As for the changes pre and post-intervention, there were no differences among the three groups (P=0.118). The secondary endpoint eGFR also showed no significant differences among the three groups. In addition, the results of the 24-h urine protein by using PP set analysis were similar to the ITT set (PP set; Table 3).
3. Safety and other indexes
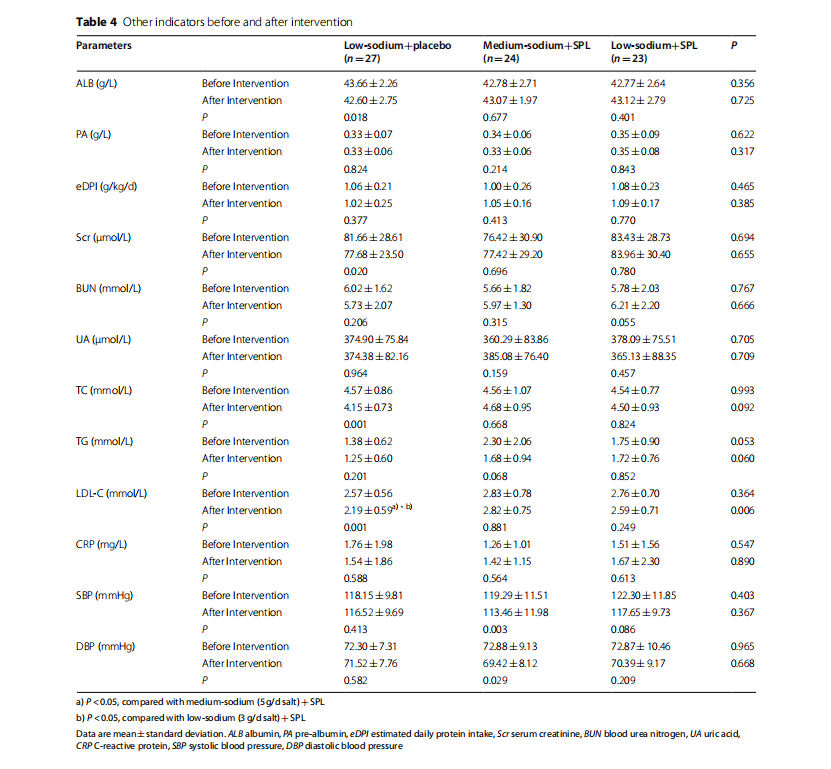
During the intervention period of this clinical trial, hyperkalemia was not observed in any of the three groups. Average blood potassium levels ranged from 4.26±0.28mmol/L in the low-sodium+placebo group, 4.29±0.26mmol/L in the medium-sodium+SPL group, and 4.35±0.32mmol/L in the low-sodium+SPL group (P>0.05). However, three patients (one patient with stage 2 CKD and two patients with stage 3a CKD, respectively) with increased blood potassium levels were observed in the low-sodium+SPL group. The range of blood potassium increase was 5.18–5.24mmol/L. Dietary reports revealed that these three patients ate high-potassium foods. Their blood potassium decreased to 4.46– 4.87mmol/L after dietary guidance was provided without the need for any medication adjustments.
No gynecomastia or sexual dysfunction was observed in patients in the medium-sodium+SPL group and low-sodium+SPL group. No significant abnormal changes in blood routine, liver function, ECG parameters, and other routine safety indicators were observed in any patient. In addition, no progressive renal dysfunction (eGFR decreased by 30%) or all-cause death was observed during the study.

Herba Cistanche
During the intervention period, nutritional, metabolic, and inflammatory markers, as well as blood pressure, were stable in all three groups. ALB levels in the lowsodium+placebo group were decreased after 12 weeks of intervention, but all fell within the normal range. TC and LDL-C levels in the low-sodium+placebo group decreased significantly (both p=0.001), while LDL-C level in the low-sodium+placebo group after the intervention was significantly lower than the other two groups (both p< 0.05). The SBP and DBP all showed a decrease after intervention with no significant statistical difference among the three groups (Table 4).
Discussion
The key observations of this study are as follows. First, no further benefit exists from the addition of SPL in patients with a low-sodium diet (3.0 g/d salt). Second, small doses of SPL may benefit patients with poor sodium restriction.
Under the condition of basic treatment, 24-h urine protein to creatine ratios were decreased significantly in all three groups of ITT or PP set after 12 weeks of intervention, but there were no significant differences among the three groups (P > 0.05). These results suggested that sodium control and/or SPL might have renal protective effects in patients with stage 1-3a CKD.
Few clinical studies have assessed the relationship between salt intake and MRA administration, and a literature review found no related randomized controlled trials. A post hoc analysis of MRA efficacy stratified by urinary sodium excretion of the Eplerenone Combination Versus Conventional Agents to Lower Blood Pressure on Urinary Antialbuminuric Treatment Efect (EVALUATE) trial was recently conducted [35]. In this study, the population included patients with CKD and hypertension but without diabetes, and the basic treatment was ACEI and/or ARB. The results showed that eplerenone-treated patients in the highest sodium excretion tertile exhibited significantly greater reduction in urinary albumin to creatinine ratio (UACR) compared with the placebo subjects in the same tertile (−22.5% vs. +21.8%, p=0.02). While this disparity was not observed either in the lowest tertile (−10.2% vs. -0.84%, p=0.65) or in the middle tertile (−19.5% vs. +9.5%, p=0.22). These authors concluded that the therapeutic effect of MRA is related to salt intake. In patients with high urinary sodium, the antiproteinuric effect of MRA was more significant than that of a placebo, while in patients with low urinary sodium, the antiproteinuric effect was not significantly different [35]. This conclusion corroborated our findings. The possible mechanism is as follows. Salt loading could enhance the renal MR activation pathway by activating Rac1 without increasing circulating aldosterone, causing hypertension and renal damage; meanwhile, MRA blocks this pathway, inhibits high-salt intake associated renal injury, overcomes salt-associated resistance to routine RAS blockade treatment, and plays a role of renal protection [36]. Similar results have been found in patients with refractory hypertension. The higher the salt intake level, the greater the blood pressure response to SPL treatment, suggesting that MRA could antagonize the increase of blood pressure caused by high salt intake [37].

Cistanche pills
As a simple and cost-effective treatment, dietary salt restriction delays CKD progression. However, patients with CKD show poor adherence to a low-salt diet in their daily life. Data showed that the average 24h urine sodium excretion in patients with CKD ranges between 144 and 200mmol/d [3, 38–41]. It is challenging to achieve salt restriction even in clinical trials. Average daily salt consumption is 9–13g/d in reported CKD clinical studies up to now [42]. Adding SPL to the treatment regimen may be a potential new strategy in patients with CKD that are unable to achieve the ideal salt restriction goal.
Hyperkalemia is the main adverse side effect in patients taking MRA, especially in combination with ACEI and/ or ARB. Previous studies have shown that the risk of hyperkalemia after treatment with MRA combined with ACEI and/or ARB is elevated compared with ACEI and/ or ARB alone [22, 23, 43–46]. Epstein et al. carefully evaluated hyperkalemia incidence in 268 patients, and blood potassium levels showed no significant change in MRA combined with ACEI compared to ACEI alone [47]. During the 12-week intervention period in this study, there was no hyperkalemia in medium-sodium+SPL and low-sodium+SPL groups. Even in patients with ESRD, previous research has suggested that treatment is safe provided that the medication indication is accurate and strict monitoring parameters are applied [48].
Gynecomastia resulting from the combination of aldosterone with androgen and progesterone receptors is often reported as a side effect of SPL, thereby limiting its clinical usefulness. In the RALES study, gynecomastia incidence in patients treated with SPL was 9% versus 1% in the placebo arm [49]. The use of the selective MRA EPL could reduce these risks and other side effects [50]. In addition, furanone, a non-steroidal, selective mineralocorticoid receptor antagonist, reduced the risks of CKD progression and cardiovascular events in patients with CKD and type 2 diabetes compared with placebo in the FIDELIO-DKD trial; meanwhile, the overall rates of adverse events were similar in both groups [51]. In this study, the non-selective MRA SPL was administered at 20mg/day, and no significant adverse events were found in the medium-sodium+SPL and low-sodium+SPL groups throughout the study.
Several limitations in this clinical trial are noted as follows. First, although this was a prospective, randomized controlled study (medication administration was performed in a blinded manner), salt intake could not be blinded. Secondly, based on ethical principles, a high salt group (oral salt tablet) was not established in this study due to the proven high salt-associated renal damage. Thirdly, low-dose ARB was administered to most patients, and the independent effect of salt restriction alone or salt restriction combined with MRA could not be explained. Fourthly, this was a single-center pilot study, with a relatively small sample size, relatively normal kidney function, and short follow-up, which limited the evaluation of the efficacy and long-term safety profile. Fifthly, long-term data for various endpoints were not obtained. Based on these limitations, larger multicenter clinical trials with longer follow-ups are required to further evaluate the effects and safety of sodium restriction and MRAs.

Conclusion
Under low sodium intake conditions, ARB combined with SPL has no additional benefits in lowering urine protein compared with ARB alone. It is unnecessary to add on SPL for patients with satisfying sodium restriction, thus avoiding the adverse events of SPL. In patients with poor sodium restriction, supplementation of low-dose SPL might show a therapeutic benefit in lowering 24-h urine protein. The combination of short-term low-dose SPL and ARB is safe in patients with stage 1-3a CKD, but blood potassium must be carefully monitored.
The effect of Cistanche extract on patients with chronic kidney disease
Cistanche extract is a traditional Chinese herbal medicine that has attracted attention for its potential therapeutic benefits on patients with chronic kidney disease (CKD). CKD is a progressive condition characterized by a decline in renal function, which can lead to complications such as cardiovascular disease and end-stage renal failure. Cistanche extract contains various active constituents such as phenylethanoid glycosides, polysaccharides, iridoids, and echinacoside, which have been found to possess anti-inflammatory, antioxidant, and anti-apoptotic properties.
Studies have shown that Cistanche extract may help improve renal function and reduce the severity of kidney damage, possibly by promoting tissue regeneration and preventing cell apoptosis. In animal models of CKD, Cistanche extract treatment led to significant reductions in serum creatinine and blood urea nitrogen levels while increasing GFR and renal tissue weight.
Furthermore, Cistanche extract has been suggested to possess a protective effect against acute kidney injury and prevent the formation of renal calcium oxalate crystals, which are often observed in patients with nephrolithiasis (kidney stones).
While there is still a need for further research on the safety and efficacy of Cistanche extract in treating CKD, these findings suggest that it could be a promising complementary therapy along with other conventional treatments such as dietary modifications and medication. As always, it's essential to talk to a healthcare professional before adding any new supplement to your regimen.
References
35. Nishimoto M, Ohtsu H, Marumo T, Kawarazaki W, Ayuzawa N, Ueda K, et al. Mineralocorticoid receptor blockade suppresses dietary salt-induced ACEI/ARB-resistant albuminuria in non-diabetic hypertension: a sub-analysis of evaluate the study. Hypertens Res. 2019;42:514–21.
36. Shibata S, Ishizawa K, Uchida S. Mineralocorticoid receptor as a therapeutic target in chronic kidney disease and hypertension. Hypertens Res. 2017;40:221–5.
37. Ghazi L, Dudenbostel T, Lin CP, Oparil S, Calhoun DA. Urinary sodium excretion predicts blood pressure response to spironolactone in patients with resistant hypertension independent of aldosterone status. J Hypertens. 2016;34:1005–10.
38. De Nicola L, Minutolo R, Chiodini P, Zoccali C, Castellino P, Donadio C, et al. A global approach to cardiovascular risk in chronic kidney disease: reality and opportunities for intervention. Kidney Int. 2006;69:538–45.
39. Van Zuilen AD, Wetzels JF, Bots ML, Van Blankestijn PJ, Group MS. MASTERPLAN: a study of the role of nurse practitioners in a multifactorial intervention to reduce cardiovascular risk in chronic kidney disease patients. J Nephrol. 2008;21:261–7.
40. Kutlugun AA, Arici M, Yildirim T, Turgut D, Yilmaz R, Altindal M, et al. Daily sodium intake in chronic kidney disease patients during nephrology clinic follow-up: an observational study with 24-hour urine sodium measurement. Nephron Clin Pract. 2011;118:c361–6.
41. Kang SS, Kang EH, Kim SO, Lee MS, Hong CD, Kim SB. Use of mean spot urine sodium concentrations to estimate daily sodium intake in patients with chronic kidney disease. Nutrition. 2012;28:256–61.
42. Krikken JA, Laverman GD, Navis G. Benefits of dietary sodium restriction in the management of chronic kidney disease. Curr Opin Nephrol Hypertens. 2009;18:531–8.
43. Chrysostomou A, Pedagogos E, MacGregor L, Becker GJ. Double-blind, placebo-controlled study on the effect of the aldosterone receptor antagonist spironolactone in patients who have persistent proteinuria and are on long-term angiotensin-converting enzyme inhibitor therapy, with or without an angiotensin II receptor blocker. Clin J Am Soc Nephrol. 2006;1:256–62.
44. van den Meiracker AH, Baggen RG, Pauli S, Lindemans A, Vulto AG, Poldermans D, et al. Spironolactone in type 2 diabetic nephropathy: Effects on proteinuria, blood pressure, and renal function. J Hypertens. 2006;24:2285–92.
45. Rossing K, Schjoedt KJ, Smidt UM, Boomsma F, Parving HH. Beneficial effects of adding spironolactone to recommended antihypertensive treatment in diabetic nephropathy: a randomized, double-masked, crossover study. Diabetes Care. 2005;28:2106–12.
46. Schjoedt KJ, Rossing K, Juhl TR, Boomsma F, Rossing P, Tarnow L, et al. The beneficial impact of spironolactone in diabetic nephropathy. Kidney Int. 2005;68:2829–36.
47. Epstein M, Williams GH, Weinberger M, Lewin A, Krause S, Mukherjee R, et al. Selective aldosterone blockade with eplerenone reduces albuminuria in patients with type 2 diabetes. Clin J Am Soc Nephrol. 2006;1:940–51.
48. Lyubarova R, Gosmanova EO. Mineralocorticoid Receptor Blockade in End-Stage Renal Disease. Curr Hypertens Rep. 2017;19:40.
49. Pitt B, Zannad F, Remme WJ, Cody R, Castaigne A, Perez A, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N Engl J Med. 1999;341:709–17.
50. Pitt B, Remme W, Zannad F, Neaton J, Martinez F, Roniker B, et al. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med. 2003;348:1309–21.
51. Bakris GL, Agarwal R, Anker SD, Pitt B, Ruilope LM, Rossing P, et al. Effect of Finerenone on Chronic Kidney Disease Outcomes in Type 2 Diabetes. N Engl J Med. 2020;383:2219–29.
Hongmei Zhang, Bin Zhu, Liyang Chang, Xingxing Ye, Rongrong Tian, Luchen He, Dongrong Yu, Hongyu Chen, and Yongjun Wang






