Part Ⅱ:Impact Of Obesity in Kidney Diseases
Apr 12, 2023
Clinical Consequences
The clinical consequences of obesity on the kidney, with or without metabolic abnormalities, involve renal function and structure, Figure 2. Higher rates of urinary albumin excretion, subnephrotic syndrome, kidney stones, increased risk of CKD, and ESKD progression have been determined to be associated with obesity and have relevant clinical implications. In renal replacement therapy and kidney transplantation, obesity also affects donor availability and graft survival. The presence of sarcopenia (a not uncommon condition) may be misleading when estimating the association and impact of obesity with renal disease, as it may lead to an underestimation of obesity. Therefore, parameters other than BMI should be considered.
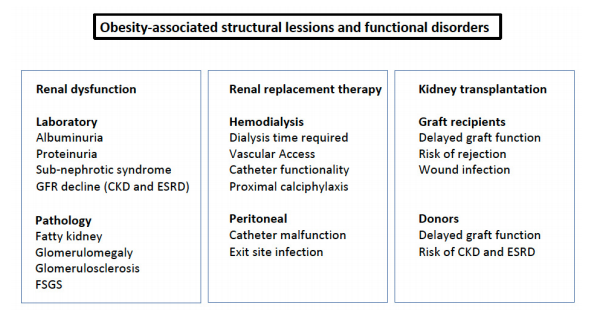
Figure 2. Obesity-associated structural lesions and dysfunction. FSGS: focal segmental glomerulosclerosis.
1. Urinary Albumin Excretion and Proteinuria
Proteinuria is more common in obese subjects. Proteinuria has been reported to be significantly associated with either obesity or central obesity, with proteinuria being higher in the presence of central obesity. The presence of cardiovascular risk factors increases the risk.
Proteinuria associated with obesity has been observed in children and adolescents. In moderately obese adolescents, the prevalence was 2.4%; however, in severe obesity, 3% exhibited proteinuria, 14% exhibited microalbuminuria, and 3% had a GFR <60 mL/min/1.73 m2. In addition, Goknar et al. reported a higher number of markers of ureteral damage in severely obese children, such as n -acetyl- β - d -amyloglucosidase (NAG) and renal injury molecule (KIM)-1.
Although the prevalence of proteinuria is well established in obese patients, this condition remains underdiagnosed due to the lack of clinical signs and the lack of specific search for low-purity proteinuria.
2. Sub-Nephrotic Syndrome
Obesity-associated glomerulopathy is a characteristic syndrome that can be divided into subnephrotic proteinuria, glomerulopathy, and loss of renal function. In 30% of subjects, patients usually do not have nephrotic syndrome levels of proteinuria, in the absence of edema, hypoproteinemia, and much less hyperlipidemia. The reason for the differences between this evidence and typical nephrotic syndrome is the delayed development of compensatory mechanisms over the years. These mechanisms reduce or limit the systemic and metabolic effects and increase the hepatic synthesis of albumin and other proteins. This is in contrast to a nephrotic syndrome caused by other etiologies. Biopsies of obese patients show glomerulomegaly, and some of them also develop adaptive focal segmental glomerulosclerosis, increasing the risk of progression to renal dysfunction.

Click here to get Cistanche supplements to relieve kidney failure
3. Progression to CKD and ESRD
Obesity is associated with a higher prevalence of CKD, defined as proteinuria and/or GFR <60 mL/min/1.73 m2 compared to the non-obese population. The effect of obesity on the progressive decline in renal function has been highlighted. Subjects with low birth weight children, low renal endowment, reduced renal mass due to different origins, or primary or secondary renal injury are at increased risk of progression to CKD and ESKD in the presence of obesity. The role of obesity-associated metabolic abnormalities in increased risk has received attention. Although some studies support that the risk of developing CKD and ESKD is not increased or even decreased in metabolically healthy obese individuals (MHO). However, other studies prefer that MHO is the first stage of obesity and that the development of metabolic abnormalities is a matter of time, thereby increasing the risk of developing renal dysfunction.
The risk of end-stage renal disease (ESKD) is more than three times higher in obese individuals than in those of normal weight. In a large Austrian cohort study, the prevalence of obesity was 11.8%, and 0.3% developed ESKD over 22 years of follow-up, with a 56% increase in risk for every 5-point increase in BMI. 320,252 subjects in the Kaiser permanent registry were followed for more than 21 years, and the risk of ESKD compared to normal weight subjects was 3.57, 6.10, and 7.07 for obesity class, respectively, 6.10 and 7.07 in obese subjects 1 to III, respectively. However, controversial data have been reported when assessing the rate of decline in renal function to ESKD in CKD. While some studies have reported a more rapid decline in the presence of obesity, others have not confirmed it.
4. Nephrolithiasis
The prevalence and incidence of kidney stones are increased in obese populations. Lower urinary pH, increased urinary oxalate, sodium and phosphate excretion, and uric acid promote this association. Other factors, such as the effect of insulin resistance on tubular HNa exchangers and the promotion of urinary acidification by ammonification, are also involved in the pathogenesis. Notably, this risk is increased after certain weight loss therapies. Indeed, the absorption of intestinal oxalic acid by gastric bypass is substantially increased after Roux-en-Y treatment, requiring prevention of the risk of kidney stones by reducing dietary oxalic acid intake and oral calcium supplementation.
5. Renal Replacement Therapy
The increased prevalence of obesity in hemodialysis and peritoneal dialysis presents a challenge to the optimal care of patients on renal replacement therapy. In the case of hemodialysis, at 3 years, obesity in the subcutaneous tissue leads to problems with vascular access and reduced catheter function. Moreover, in obese subjects, increasing the duration or frequency of dialysis is necessary and makes it more difficult to achieve dry weight. Proximal calcification reactions are more common in obese patients than in lean patients. In peritoneal dialysis patients, catheter dysfunction and exit site infections are more common in obese subjects. In some severely obese patients, prophylactic omentectomy may be useful. In addition, patients with advanced CKD, especially those on dialysis, often have severe nutritional disorders and protein-energy wastage, and the presence of obesity may be better in this population, an obesity paradox.
6. Kidney Transplantation
In the past, obesity was a contraindication to kidney transplantation if there was no weight loss. Despite the fact that the cutoff limit has increased, even with a BMI of 40 kg/m2, obesity remains one of the main reasons for inactivity on the transplant list. The reason for this is the delayed graft function, wound infection, and increased incidence of rejection in obese recipients of transplants.
The effect of obesity on living kidney donors and the acceptance of organs by obese subjects are also relevant. In the former, both donors and recipients are at risk, as a substantial reduction in obese subjects puts them at risk for future ESKD, while in the latter, delayed graft function is more common if the donor is obese. According to KDIGO recommendations, the BMI of the living donor>30 kg/m2 may lead to hypertension, diabetes mellitus, and ESKD.

Herba Cistanche
7. Renal Cancer
Obesity is associated with an increased risk of kidney malignancies. Several studies have concluded that increased risk is associated with obesity It is estimated that 20% of kidney cancer patients are obese. Compared to normal-weight subjects, the risk of kidney cancer was increased by 35% in overweight subjects and 76% in obese subjects, independent of sex. This association is consistent across both sexes and populations; however, the pathogenesis is not clearly explained to date.
8. Fatty Kidney
The accumulation of ectopic fat in the kidney has received increasing attention in recent years and will increase with the development of techniques that allow better estimation than conventional ultrasound and CT scans. In addition to intrarenal accumulation in the proximal tubule and small-grade accumulation in the glomerulus, fat in the renal sinus and around the perinephric membrane appears to play a role in renal dysfunction in obese patients. The Framingham Heart Study found an association between renal sinus fat and CKD. In addition, perirenal fat appears to exert a lipotoxic effect on the kidney, increasing glomerular hydrostatic pressure and renin-angiotensin-aldosterone system activity and promoting the progression of renal damage.
9. Other Obesity-Associated Conditions and Renal Damage
Two common complications of obesity appear to further increase the risk of renal damage. The first is sleep apnea and nocturnal hypoxemia, which are associated with loss of renal function through activation of the renin-angiotensin system. The second is a non-alcoholic fatty liver disease (NAFLD). In a meta-analysis involving 33 studies, NAFLD, non-alcoholic steatohepatitis, and advanced fibrosis were associated with an increased risk of CKD prevalence and morbidity, with a graded risk from the present to the severity of NAFLD.
Treatment of obesity and kidney damage
Weight loss is key to limiting the effects of obesity on the kidney by reducing proteinuria/albuminuria, decreasing the rate of eGFR deterioration, delaying the progression of CKD and ESKD, and improving kidney transplant outcomes. The impact due to weight loss is multifaceted. In addition to lowering blood pressure and controlling other CV risk factors, reduced leptin, glomerular hyperfiltration, RAAS activity, inflammation, and oxidative stress appear to be the most relevant. Considering the characteristic hyperfiltration hemodynamic profile and the relevance of hyperfiltration-mediated conditions in obesity-induced renal injury, the reduction in filtration rate is the main mechanism providing beneficial effects in subjects with weight loss. In addition, a decrease in RAAS activity was also observed. Weight loss also contributes to the proper control of cardiometabolic risk factors such as hypertension, metabolic syndrome, diabetes mellitus, and dyslipidemia, which may have a protective effect not only against renal damage but also against cardiovascular disease.
1. Life Style Intervention
Obese patients, especially those with markers of renal injury (proteinuria/tubular markers or eGFR <60 mL/min/1.73 m2), need to be encouraged to lose weight through a combination of diet and physical activity. If addressed early, a low-calorie diet, with or without physical activity, can reduce proteinuria to a degree proportional to weight loss. Weight loss achieved through a combination of diet and exercise also has a beneficial effect on reducing urinary protein excretion. A reduction in UAE can be observed within a few weeks after the introduction of the diet. In a controlled trial lasting 5 months, a 4% weight loss reduced proteinuria in approximately 50% of subjects. However, data on slowing the progression of CKD are scarce due to the difficulty of assessing outcomes and the short-term duration of the study.
A low-calorie diet with salt restriction is recommended, as it helps to lower blood pressure. If proteinuria is present, salt intake should be further reduced. The dietary addition of fiber to promote the growth of short-chain fatty acid-producing bacteria has been shown to reduce all-cause mortality in CKD and appears to have promising results in preclinical CKD risk. High-protein diets are not recommended because they increase GFR and UAE.
A recent manuscript reviewed randomized clinical trials of lifestyle interventions in patients with CKD. Dietary interventions low in calories and salt restriction reduced body weight and albuminuria; however, there are no convincing data for other specific dietary patterns, such as low-fat, low-carbohydrate, or Mediterranean diets. Studies on the effects of physical activity have shown reductions in blood pressure, BMI, and improvements in exercise capacity and quality of life; however, no reduction in proteinuria was seen. The limitation of lifestyle weight loss is that the maximum weight loss effect is 3 ~ 4%, with poor maintenance time, thus requiring the implementation of other additional actions.

Cistanche benefits for Kidney function
2. Medications
RAAS blockers
In the setting of proteinuria or proteinuria, RAAS blockers may reduce not only systemic overactivity, but also sympathetic overactivity, HTN, insulin resistance, and hypo-inflammation. The most important effect is a reduction in filtration rate and thus proteinuria; however, patients with CKD should be monitored for a reduction in eGFR after initiation of therapy.
Antiobese-drugs
Among the drugs approved for the treatment of obesity, phentermine-topiramate, GLP-1 receptor agonists, and acetone-naltrexone, data on the effects of GLP1 agonists on renal function are mainly available. These drugs have been tested for renal protection in diabetic patients. Liraglutide, a GLP1 agonist, was originally introduced as a weight-affecting hypoglycemic agent capable of reducing body weight, and in a recent trial, LEADER demonstrated a reduced CV risk. In diabetic patients, there was a significant reduction in proteinuria, new persistent proteinuria, and no progression in eGFR decline. MAINTAIN -6 in combination with Semaglutide (another member of the GLP1 agonists) reduced the risk of a composite renal outcome caused mainly by persistent proteinuria. However, in AWARD-7, no difference was found in the reduction of proteinuria with dulaglutide. Topiramate did not demonstrate a beneficial effect on renal outcomes in type 2 diabetes in one study. lorcaserin, a selective serotonin 2C receptor, was associated with a reduced rate of renal damage compared to a placebo in patients at high cardiovascular risk. the beneficial effect of GLP1 is that it protects the kidney from developing CKD and/or ESKD.
Sodium-glucose cotransporter 2 inhibitors
Sodium-glucose cotransporter 2 inhibitors (SGLT2i) are a class of drugs released in recent years with mechanisms that produce various beneficial effects in patients with diabetes, obesity, and cardiac and renal protection. Inhibition of glucose reabsorption in the proximal tubule produces glycosuria, lower caloric burden, lower sodium content, reduced blood volume, increased sodium reaching the glomerular body, inhibition of renal angiotensin system hyperactivity, lower filtration rate, and renal protection. Thus, a slight reduction in body weight, lower blood pressure, and GFR results in the protection of renal function. other mechanisms of NH3, sympathetic nerve activity, and oxidative stress have beneficial effects and this is a very useful drug. Several outcome trials support the beneficial effects of the drug on cardiovascular and renal outcomes. The European Society of Cardiology (ESC) and the European Society of Diabetes (ESC/EASD) recommend SGLT2i as a first step in patients with diabetes at very high risk or with previous cardiovascular events. option to protect renal function. This also applies to obese subjects with increased urinary albumin excretion or proteinuria. However, in patients with reduced GFR, the efficacy of the drug is reduced and the protective effect is diminished. As GFR <45 ml/min/1.73 m2 is almost negligible, this is a challenge for their use. In the trial, beneficial effects were observed in patients with GFR between 30-45 mL/min/1.73 m2 and no side effects, suggesting their use outside the indication.
Conclusions
The impact of obesity on the kidney has received attention because it is recognized that BMI is the second most important indicator of ESKD development after proteinuria and one of the most relevant indicators for CKD, as obesity is commonly associated with hypertension, metabolic syndrome, and diabetes. It also has important implications for subjects on renal replacement therapy and renal transplantation. Pathological lesions include characteristic glomerulopathies with cellular fat load and perivascular fat deposits, as well as so-called adipose kidneys with fat deposits in the perinephric and renal sinuses. The mechanisms linking obesity and renal damage are well understood and include several interrelated mechanisms of effect. In the setting of increased urinary albumin excretion, action must be taken to reduce overweight and to control hypertension, diabetes mellitus, and dyslipidemia to further prevent a decrease in GFR.
Standardized Cistanche
Cistanche extract is well-tolerated by the body
One of the best things about Cistanche is that it is well-tolerated by the body. This means that it is unlikely to cause any significant side effects, even when taken in high doses. This makes it a safe and effective option for those who want to improve their kidney health without the risk of adverse effects.
Cistanche extract is an excellent supplement for those who suffer from kidney disease or want to improve their kidney health. Its ability to improve kidney function, protect against oxidative stress, reduce inflammation, and have a mild diuretic effect make it a valuable tool in the fight against kidney-related conditions. As with any supplement, it is essential to consult with a healthcare professional before starting to take Cistanche.
References
1. Sharma, D.; Hawkins, M.; Abramowitz, M.K. Association of sarcopenia with eGFR and misclassification of obesity in adults with CKD in the United States. Clin. J. Am. Soc. Nephrol. 2014, 9, 2079–2088.
2. Chintam, K.; Chang, A.R. Strategies to Treat Obesity in Patients with CKD. Am. J. Kidney Dis. 2021, 77, 427–439.
3. Chen, B.; Yang, D.; Chen, Y.; Xu, W.; Ye, B.; Ni, Z. The prevalence of microalbuminuria and its relationships with the components of metabolic syndrome in the general population of China. Clin. Chim. Acta 2010, 411, 705–709.
4. Thoenes, M.; Reil, J.C.; Khan, B.V.; Bramlage, P.; Volpe, M.; Kirch, W.; Böhm, M. Abdominal obesity is associated with microalbuminuria and an elevated cardiovascular risk profile in patients with hypertension. Vasc. Health Risk Manag. 2009, 5, 577–585.
5. Chandie Shaw, P.K.C.; Berger, S.P.; Mallat, M.; Frölich, M.; Dekker, F.; Rabelink, T.J. Central Obesity Is an Independent Risk Factor for Albuminuria in Nondiabetic South Asian Subjects. Diabetes Care 2007, 30, 1840–1844.
6. Du, N.; Peng, H.; Chao, X.; Zhang, Q.; Tian, H.; Li, H. Interaction of obesity and central obesity on elevated urinary albumin-to-creatinine ratio. PLoS ONE 2014, 9, e98926.
7. Lurbe, E.; Torro, M.I.; Alvarez, J.; Aguilar, F.; Fernandez-Formoso, J.A.; Redon, J. Prevalence and factors related to urinary albumin excretion in obese youths. J. Hypertens. 2013, 31, 2230–2236.
8. Xiao, N.; Jenkins, T.M.; Nehus, E.; Inge, T.H.; Michalsky, M.; Harmon, C.M.; Helmrath, M.A.; Brandt, M.L.; Courcoulas, A.; Moxey-Mims, M.; et al. Kidney function in severely obese adolescents undergoing bariatric surgery. Obesity 2014, 22, 2319–2325.
9. Goknar, N.; Öktem, F.; Özgen, I.T.; Torun, E.; Kucukkoc, M.; Demir, A.D.; Cesur, Y. Determination of early urinary renal injury markers in obese children. Pediatr. Nephrol. 2014, 30, 139–144.
10. Lakkis, J.I.; Weir, M.R. Obesity and Kidney Disease. Prog. Cardiovasc. Dis. 2018, 61, 157–167.
11. Hernández-Conde, M.; Llop, E.; Carrillo, C.F.; Tormo, B.; Abad, J.; Rodriguez, L.; Perelló, C.; Gomez, M.L.; Martínez-Porras, J.L.; Puga, N.F.; et al. Estimation of visceral fat is useful for the diagnosis of significant fibrosis in patients with non-alcoholic fatty liver disease. World J. Gastroenterol. 2020, 26, 6658–6668.
12. D’Agati, V.D.; Chagnac, A.; de Vries, A.; Levi, M.; Porrini, E.; Herman-Edelstein, M.; Praga, M. Obesity-related glomerulopathy: Clinical and pathologic characteristics and pathogenesis. Nat. Rev. Nephrol. 2016, 12, 453–471.
13. Fox, C.S.; Larson, M.; Leip, E.P.; Culleton, B.; Wilson, P.W.F.; Levy, D. Predictors of new-onset kidney disease in a community-based population. JAMA 2004, 291, 844–850.
14. Ejerblad, E.; Ford, C.M.; Lindblad, P.; Fryzek, J.; McLaughlin, J.K.; Nyrén, O. Obesity and risk for chronic renal failure. J. Am. Soc. Nephrol. 2006, 17, 1695–1702.
15. Chang, A.R.; Grams, M.E.; Ballew, S.; Bilo, H.; Correa, A.; Evans, M.; Gutierrez, O.M.; Hosseinpanah, F.; Iseki, K.; Kenealy, T.; et al. Adiposity and risk of decline in glomerular filtration rate: Meta-analysis of individual participant data in a global consortium. BMJ 2019, 364, k5301.
16. . Gurusinghe, S.; Brown, R.D.; Cai, X.; Samuel, C.S.; Ricardo, S.D.; Thomas, M.C.; Kett, M.M. Does a nephron deficit exacerbate the renal and cardiovascular effects of obesity? PLoS ONE 2013, 8, e73095.
17. Hashimoto, Y.; Tanaka, M.; Okada, H.; Senmaru, T.; Hamaguchi, M.; Asano, M.; Yamazaki, M.; Oda, Y.; Hasegawa, G.; Toda, H.; et al. Metabolically Healthy Obesity and Risk of Incident CKD. Clin. J. Am. Soc. Nephrol. 2015, 10, 578–583.
18. Song, Y.-M.; Sung, J.; Lee, K. Longitudinal relationships of metabolic syndrome and obesity with kidney function: Healthy Twin Study. Clin. Exp. Nephrol. 2015, 19, 887–894.
19. Panwar, B.; Hanks, L.J.; Tanner, R.M.; Muntner, P.; Kramer, H.; McClellan, W.M.; Warnock, D.G.; Judd, S.E.; Gutiérrez, O.M. Obesity, metabolic health, and the risk of end-stage renal disease. Kidney Int. 2015, 87, 1216–1222.
20. Chen, S.; Zhou, S.; Wu, B.; Zhao, Y.; Liu, X.; Liang, Y.; Shao, X.; Holthöfer, H.; Zou, H. Association between metabolically unhealthy overweight/obesity and chronic kidney disease: The role of inflammation. Diabetes Metab. 2014, 40, 423–430.
21. Hsu, C.-Y.; McCulloch, C.E.; Iribarren, C.; Darbinian, J.; Go, A.S. Body mass index and risk for end-stage renal disease. Ann. Intern. Med. 2006, 144, 21–28.
22. Zitt, E.; Pscheidt, C.; Concin, H.; Kramar, R.; Lhotta, K.; Nagel, G. Anthropometric and metabolic risk factors for ESRD are disease-specific: Results from a large population-based cohort study in Austria. PLoS ONE 2016, 11, e0161376.
23. Fritz, J.; Brozek, W.; Concin, H.; Nagel, G.; Kerschbaum, J.; Lhotta, K.; Ulmer, H.; Zitt, E. The Triglyceride-Glucose Index and Obesity-Related Risk of End-Stage Kidney Disease in Austrian Adults. JAMA Netw. Open 2021, 4, e212612.
24. Lu, J.L.; Molnar, M.Z.; Naseer, A.; Mikkelsen, M.K.; Kalantar-Zadeh, K.; Kovesdy, C.P. Association of age and BMI with kidney function and mortality: A cohort study. Lancet Diabetes Endocrinol. 2015, 3, 704–714.
25. Swartling, O.; Rydell, H.; Stendahl, M.; Segelmark, M.; Lagerros, Y.T.; Evans, M. CKD Progression and Mortality among Men and Women: A Nationwide Study in Sweden. Am. J. Kidney Dis. 2021, 78, 190–199.e1.
26. Siener, R.; Glatz, S.; Nicolay, C.; Hesse, A. The Role of Overweight and Obesity in Calcium Oxalate Stone Formation. Obes. Res. 2004, 12, 106–113.
27. Diwan, T.S.; Cuffy, M.C.; Linares-Cervantes, I.; Govil, A. Impact of obesity on dialysis and transplant and its management. Semin. Dial. 2020, 33, 279–285.
28. Kittiskulnam, P.; Johansen, K.L. The obesity paradox: A further consideration in dialysis patients. Semin. Dial. 2019, 32, 485–489.
29. Martin, W.P.; White, J.; López-Hernández, F.J.; Docherty, N.G.; Le Roux, C.W. Metabolic Surgery to Treat Obesity in Diabetic Kidney Disease, Chronic Kidney Disease, and End-Stage Kidney Disease; What Are the Unanswered Questions? Front. Endocrinol. 2020, 11, 289.
30. Rysz, J.; Franczyk, B.; Ławi ´nski, J.; Olszewski, R.; Gluba-Brzózka, A. The Role of Metabolic Factors in Renal Cancers. Int. J. Mol. Sci. 2020, 21, 7246.
31. Foster, M.C.; Hwang, S.-J.; Porter, S.A.; Massaro, J.; Hoffmann, U.; Fox, C.S. Fatty Kidney, Hypertension, and Chronic Kidney Disease: The Framingham Heart Study. Hypertension 2011, 58, 784–790.
32. Praga, M.; Morales, E. The Fatty Kidney: Obesity and Renal Disease. Nephron 2016, 136, 273–276.
33. Hanly, P.J.; Ahmed, S.B. Sleep Apnea and the Kidney: Is sleep apnea a risk factor for chronic kidney disease? Chest 2014, 146, 1114–1122.
34. Musso, G.; Gambino, R.; Tabibian, J.H.; Ekstedt, M.; Kechagias, S.; Hamaguchi, M.; Hultcrantz, R.; Hagström, H.; Yoon, S.K.; Charatcharoenwitthaya, P.; et al. Association of Non-alcoholic Fatty Liver Disease with Chronic Kidney Disease: A Systematic Review and Meta-analysis. PLoS Med. 2014, 11, e1001680.
35. Camara, N.; Iseki, K.; Kramer, H.; Liu, Z.-H.; Sharma, K. Kidney disease and obesity: Epidemiology, mechanisms, and treatment. Nat. Rev. Nephrol. 2017, 13, 181–190.
36. DeBoer, M.D.; Filipp, S.L.; Musani, S.K.; Sims, M.; Okusa, M.D.; Gurka, M.J. Metabolic Syndrome Severity and Risk of CKD and Worsened GFR: The Jackson Heart Study. Kidney Blood Press. Res. 2018, 43, 555–567.
37. Morales, E.; Valero, M.A.; León, M.; Hernández, E.; Praga, M. Beneficial effects of weight loss in overweight patients with chronic proteinuric nephropathies. Am. J. Kidney Dis. 2003, 41, 319–327.
38. Afshinnia, F.; Wilt, T.J.; Duval, S.; Esmaeili, A.; Ibrahim, H.N. Weight loss and proteinuria: Systematic review of clinical trials and comparative cohorts. Nephrol. Dial. Transplant. 2009, 25, 1173–1183.
39. Navaneethan, S.D.; Yehnert, H.; Moustarah, F.; Schreiber, M.J.; Schauer, P.R.; Beddhu, S. Weight Loss Interventions in Chronic Kidney Disease: A Systematic Review and Meta-analysis. Clin. J. Am. Soc. Nephrol. 2009, 4, 1565–1574.
40. Krishnamurthy, V.M.R.; Wei, G.; Baird, B.C.; Murtaugh, M.; Chonchol, M.B.; Raphael, K.; Greene, T.; Beddhu, S. High dietary fiber intake is associated with decreased inflammation and all-cause mortality in patients with chronic kidney disease. Kidney Int. 2012, 81, 300–306.
41. Vaziri, N.D.; Liu, S.-M.; Lau, W.L.; Khazaeli, M.; Nazertehrani, S.; Farzaneh, S.H.; Kieffer, D.A.; Adams, S.H.; Martin, R.J. High Amylose Resistant Starch Diet Ameliorates Oxidative Stress, Inflammation, and Progression of Chronic Kidney Disease. PLoS ONE 2014, 9, e114881.
42. Marso, S.P.; Daniels, G.H.; Brown-Frandsen, K.; Kristensen, P.; Mann, J.F.E.; Nauck, M.A.; Nissen, S.E.; Pocock, S.; Poulter, N.R.; Ravn, L.S.; et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 311–322.
43. Verma, S.; McGuire, D.K.; Bain, S.C.; Bhatt, D.L.; Leiter, L.A.; Mazer, C.D.; Fries, T.M.; Pratley, R.E.; Rasmussen, S.; Vrazic, H.; et al. Effects of glucagon-like peptide-1 receptor agonists liraglutide and liraglutide on cardiovascular and renal outcomes across body mass index categories in type 2 diabetes: Results of the LEADER and SUSTAIN 6 trials. Diabetes Obes. Metab. 2020, 22, 2487–2492.
44. Tuttle, K.; Lakshmanan, M.C.; Rayner, B.; Zimmermann, A.G.; Woodward, D.B.; Botros, F.T. Body weight and eGFR during dulaglutide treatment in type 2 diabetes and moderate-to-severe chronic kidney disease (AWARD-7). Diabetes Obes. Metab. 2019, 21, 1493–1497.
45. Scirica, B.M.; Bohula, E.A.; Dwyer, J.P.; Qamar, A.; Inzucchi, S.E.; McGuire, D.K.; Keech, A.C.; Smith, S.R.; Murphy, S.A.; Im, K.; et al. Lorcaserin and Renal Outcomes in Obese and Overweight Patients in the CAMELLIA-TIMI 61 Trial. Circulation 2019, 139, 366–375.
46. Janež, A.; Fioretto, P. SGLT2 Inhibitors and the Clinical Implications of Associated Weight Loss in Type 2 Diabetes: A Narrative Review. Diabetes Ther. 2021, 12, 2249–2261.
47. Wanner, C.; Inzucchi, S.E.; Lachin, J.; Fitchett, D.; Von Eynatten, M.; Mattheus, M.; Johansen, O.E.; Woerle, H.J.; Broedl, U.C.; Zinman, B.; et al. Empagliflozin and Progression of Kidney Disease in Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 323–334.
48. Furtado, R.H.; Bonaca, M.P.; Raz, I.; Zelniker, T.A.; Mosenzon, O.; Cahn, A.; Kuder, J.; Murphy, S.A.; Bhatt, D.L.; Leiter, L.A.; et al. Dapagliflozin and Cardiovascular Outcomes in Patients With Type 2 Diabetes Mellitus and Previous Myocardial Infarction. Circulation 2019, 139, 2516–2527.
49. Neal, B.; Perkovic, V.; Mahaffey, K.W.; De Zeeuw, D.; Fulcher, G.; Erondu, N.; Shaw, W.; Law, G.; Desai, M.; Matthews, D.R. Canagliflflozin and Cardiovascular and Renal Events in Type 2 Diabetes. N. Engl. J. Med. 2017, 377, 644–657.
50. Perkovic, V.; Jardine, M.J.; Neal, B.; Bompoint, S.; Heerspink, H.J.L.; Charytan, D.M.; Edwards, R.; Agarwal, R.; Bakris, G.; Bull, S.; et al. Canagliflflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy. N. Engl. J. Med. 2019, 380, 2295–2306.
51. Cosentino, F.; Grant, P.J.; Aboyans, V.; Bailey, C.J.; Ceriello, A.; Delgado, V.; Federici, M.; Filippatos, G.; Grobbee, E.D.; Hansen, T.B.; et al. 2019 ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD. Eur. Heart J. 2020, 41, 255–323.
52. Li, J.; Fagbote, C.O.; Zhuo, M.; Hawley, C.E.; Paik, J.M. Sodium-glucose cotransporter 2 inhibitors for diabetic kidney disease: A primer for deprescribing. Clin. Kidney J. 2019, 12, 620–628.
53. Scheurlen, K.M.; Probst, P.; Kopf, S.; Nawroth, P.P.; Billeter, A.T.; Müller-Stich, B.P. Metabolic surgery improves renal injury independent of weight loss: A meta-analysis. Surg. Obes. Relat. Dis. 2019, 15, 1006–1020.
54. Shulman, A.; Peltonen, M.; Sjöström, C.D.; Andersson-Assarsson, J.C.; Taube, M.; Sjöholm, K.; Le Roux, C.W.; Carlsson, L.M.S.; Svensson, P.-A. Incidence of end-stage renal disease following bariatric surgery in the Swedish Obese Subjects Study. Int. J. Obes. 2018, 42, 964–973.
55. Funes, D.R.; Blanco, D.G.; Gómez, C.O.; Frieder, J.S.; Menzo, E.L.; Szomstein, S.; White, K.P.; Rosenthal, R.J. Metabolic Surgery Reduces the Risk of Progression From Chronic Kidney Disease to Kidney Failure. Ann. Surg. 2019, 270, 511–518.
56. Friedman, A.N.; Wahed, A.S.; Wang, J.; Courcoulas, A.P.; Dakin, G.; Hinojosa, M.W.; Kimmel, P.L.; Mitchell, J.E.; Pomp, A.; Pories, W.J.; et al. Effect of Bariatric Surgery on CKD Risk. J. Am. Soc. Nephrol. 2018, 29, 1289–1300.
57. Kuo, J.H.; Wong, M.S.; Perez, R.V.; Li, C.-S.; Lin, T.-C.; Troppmann, C. Renal Transplant Wound Complications in the Modern Era of Obesity. J. Surg. Res. 2012, 173, 216–223.
58. Abou-Mrad, R.M.; Abu-Alfa, A.K.; Ziyadeh, F.N. Effects of weight reduction regimens and bariatric surgery on chronic kidney disease in obese patients. Am. J. Physiol. Renal Physiol. 2013, 305, F613–F617.
Vasilios Kotsis 1, Fernando Martinez 2, Christina Trakatelli 1 and Josep Redon 2,3,4.
1. 3rd Department of Internal Medicine, Hypertension-24h ABPM ESH Center of Excellence, Papageorgiou Hospital, Aristotle University of Thessaloniki, 564 29 Pavlos Melas, Greece; vkotsis@auth.gr (V.K.); ctrak@auth.gr (C.T.)
2. Internal Medicine Hospital Clínico de Valencia, 46010 Valencia, Spain; fernandoctor@hotmail.com
3. Cardiovascular and Renal Research Group, INCLIVA Research Institute, the University of Valencia, 46010 Valencia, Spain
4. CIBERObn Carlos III Institute, 28029 Madrid, Spain






