Part One Endothelial Cell Dysfunction And Increased Cardiovascular Risk in Patients With Chronic Kidney Disease
Jun 07, 2023
Abstract
The endothelium is considered to be the gatekeeper of the vessel wall, maintaining and regulating vascular integrity. In patients with chronic kidney disease, protective endothelial cell functions are impaired due to the proinflammatory, prothrombotic, and uremic environment caused by the decline in kidney function, adding to the increase in cardiovascular complications in this vulnerable patient population. In this review, we discuss endothelial cell functioning in healthy conditions and the contribution of endothelial cell dysfunction to cardiovascular disease. Further, we summarize the phenotypic changes of the endothelium in chronic kidney disease patients and the relation of endothelial cell dysfunction to cardiovascular risk in chronic kidney disease. We also review the mechanisms that underlie endothelial changes in chronic kidney disease and consider potential pharmacological interventions that can ameliorate endothelial health.
Keywords
atherosclerosis, cardiovascular diseases, chronic kidney diseases, endothelial cells, vascular stiffness.

Click here to buy the Cistanche supplement
INCREASED CARDIOVASCULAR RISK IN CHRONIC KIDNEY DISEASE
Chronic kidney disease (CKD) is defined by kidney damage or a reduced kidney filtration function (glomerular filtration rate <60 mL/minute per 1.73 m²) for a period beyond 3 months that affects health.1 With a global prevalence of ≈13.4%,2 CKD imposes a serious burden on our socioeconomic and healthcare system. CKD is a progressive disease, classified into stages 1 to 5 based on the reduction in kidney function (Figure 1A).1 In end-stage kidney disease (CKD stage 5: glomerular filtration rate <15 mL/minute per 1.73 m²), patients require kidney support therapy as dialysis or kidney transplantation to replace the failing kidney function.
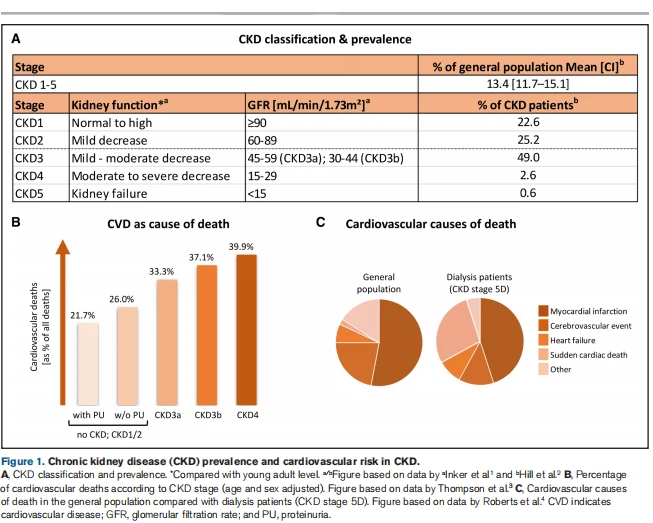
Of note, patients with moderate to advanced CKD are at increased cardiovascular risk compared with the general population and patients with mild CKD, with lower estimated glomerular filtration rate and higher albuminuria identified as risk factors for all-cause and cardiovascular mortality, independent of traditional cardiovascular risk factors.5,6 Overall, 33.3% to 37.1% of CKD3(a/b) patients and 39.9% of CKD4 patients die from cardiovascular diseases (CVD) compared with 21.7% to 26% of the general population (Figure 1B).3 For patients with end-stage kidney disease aged 25 to 34, annual mortality is even increased 500 to 1000 times compared with similarly aged controls with healthy kidney function and is comparable to 85-year-olds in the general population, underscoring the high cardiovascular burden in CKD.6
In the general population, myocardial infarction and cerebrovascular events are the most important cardiovascular causes of death, underlying ≈75% of all CVD-related deaths (Figure 1C).4 Also, in CKD5 patients on dialysis (CKD5D), these CVD types remain important as they account for 45% (myocardial infarction) and 13% (cerebrovascular events) of deaths by cardiovascular causes, though with a joint responsibility of ≈58% of all CVD-associated deaths, they are reduced in relative importance compared with the general population. Instead, CKD5D patients show a relative increase in sudden cardiac death and heart failure, being responsible for 28% and 9% of all CVD-associated deaths compared with 2% and 7% as observed in the general population (Figure 1C).4 This reveals a high increase, especially in the risk of sudden cardiac death in advanced CKD.4
1. Atherosclerosis and Myocardial Infarction
The main underlying cause of both myocardial infarction and stroke is atherosclerosis, a lipid-driven inflammatory disease of medium to large-sized arteries triggering the development of atherosclerotic lesions.7 These lesions gradually grow over time and can ultimately restrict blood flow or trigger thrombosis through plaque rupture or erosion.7,8 Patients with CKD3-5D show an increased prevalence of subclinical atherosclerotic lesions compared with the general population, with a larger increase in more advanced CKD stages after adjustment for sex, age, and diabetes.9,10 Furthermore, compared with patients without CKD progression, patients with CKD progression over 24 months displayed more frequently also a progression of atherosclerotic lesions as detected by ultrasound.10,11 After acute myocardial infarction, patients with CKD show a reduced survival over time compared with non-CKD patients, with an increased risk of death as well as non-fatal cardiac events with increasing CKD stage.12
2. Sudden Cardiac Death and Uremic Cardiomyopathy
In the general population, coronary heart disease is responsible for 80% of sudden cardiac deaths.13 The disproportional increase in sudden cardiac death in patients with advanced CKD suggests differences in its pathophysiology and causes as kidney function declines. Left ventricular hypertrophy is significantly associated with an increased risk of sudden cardiac death in the general population14 and can be caused by cardiac preload (intravascular volume overload), cardiac afterload (pressure overload), or afterload/preload-independent factors.15 Patients with CKD present more frequently with left ventricular hypertrophy, with a prevalence of up to 40% and even 75% in patients in CKD5D.16 Together with cardiac fibrosis, left ventricular hypertrophy is one of the hallmarks of uremic cardiomyopathy and may trigger cardiac electrical disturbances and lethal arrhythmias.15

Cistanche tubulosa
3. Endothelial (Cell) Dysfunction as a Contributor to Cardiovascular Risk
A main contributor to increased cardiovascular risk is endothelial cell dysfunction, which encompasses a whole array of maladaptive alterations in the endothelial cell functional phenotype associated with increased cardiovascular risk. This term was suggested in an excellent review by Gimbrone et al17 to provide a distinction from the more narrow term “endothelial dysfunction,” which typically has been used to refer to endothelial abnormalities triggering a reduction in nitric oxide bioavailability and associated vascular relaxation. Significant endothelial heterogeneity exists across the vascular tree, for example, when comparing arteries versus veins, as well as the microvasculature (including the large elastic as well as muscular conduit arteries) versus the microvasculature (including the capillaries, arterioles, and venules) and both endothelial cell dysfunction at the macro- and microvascular level contribute to increased cardiovascular risk. In this review, we discuss the contribution of endothelial cell dysfunction to CVD with a special focus on patients with CKD. We review findings on molecular mechanisms underlying endothelial cell dysfunction in CKD as well as discuss the impact of pharmacological interventions.
ENDOTHELIAL CELL DYSFUNCTION AS A CONTRIBUTOR TO CARDIOVASCULAR RISK
1. Endothelial Cell Dysfunction and Atherosclerotic Risk
The endothelial cell layer of the vasculature provides a semipermeable barrier enabling a regulated exchange of fluids, molecules, and cells and plays an important role in maintaining vascular health (Figure 2). Macrovascular endothelial cell dysfunction is an early event in the development of atherosclerotic lesions. On the one hand, it is influenced by hemodynamic factors: in atherosclerosis-resistant areas of the arteries, a laminar blood flow contributes to a protective endothelial cell phenotype. However, atherosclerosis-prone regions of the arterial vasculature are exposed to a disturbed, oscillatory blood flow and associated low time-averaged shear stress, which induce oxidative stress, endothelial phenotypic changes, and cell junction alterations as well as endothelial cell turnover (as discussed in more detail in the review by Gimbrone et al17). Furthermore, inflammatory triggers such as proinflammatory cytokines, oxLDL (oxidized low-density protein) as well as different cardiovascular risk factors such as metabolic disturbances and smoking, contribute to endothelial cell dysfunction. Also, excessive stretch on blood vessels can trigger endothelial permeability, inflammatory responses, and oxidative stress.18 Combined, this triggers proinflammatory signaling in endothelial cells with an upregulation of proinflammatory cytokines (eg, IL [interleukin]-1, IL-8), chemokines (eg, C-C motif chemokine ligand 2), and endothelial-leukocyte adhesion molecules (VCAM-1 [vascular cell adhesion molecule 1], ICAM-1 [intercellular adhesion molecule 1], P-selectin), reduces endothelial production of atheroprotective nitric oxide and increases endothelial permeability. As a result, inflammatory leukocytes are recruited, adhere to the inflamed endothelium and infiltrate into the vascular wall, where they together with accumulated lipids contribute to the development and progression of atherosclerotic lesions.7 The atheroprotective phenotype of the endothelium is regulated by master transcription factors as KLF (Kruppel-like factor)-2, KLF-4, and NRF (nuclear factor erythroid 2-related factor)-2, whereas NF-κB (nuclear factor-κB) is a key transcription factor driving endothelial inflammation.17 Furthermore, endothelial cells demonstrate a de-differentiation and increased heterogeneity during atherosclerosis progression, with also signs of endothelial-to-mesenchymal transition. Endothelial-to-mesenchymal transition is characterized by the acquiring of mesenchymal cell functions as ECM (extracellular matrix) production and is mainly driven by the transcription factors Snail, Slug, and Twist1. Its extent has been associated with the severity of atherosclerosis plaques in human arteries,19,20 and animal studies investigating key regulators of endothelial-to-mesenchymal transition suggested an important role in plaque progression20 and calcification,21 as discussed in detail in an excellent review by Souilhol et al.22

Of note, endothelial cell dysfunction does not only contribute to plaque initiation, progression, and destabilization with subsequent plaque rupture but also to atherosclerotic plaque erosion, which is expected to be responsible for one-third of acute coronary syndromes. Here, chronic low-grade endothelial activation by for example TLR (toll-like receptor)-2 ligands combined with endothelial cell apoptosis and catabolism of basement membrane components can trigger endothelial cell detachment with subsequent thrombus formation on the denuded area.8
2. Endothelial Cell Dysfunction and Thrombotic Risk
A healthy, functional endothelial layer at macro- and microvascular levels is crucial in the regulation of hemostasis and interferes both at the level of primary as well as secondary hemostasis to prevent unwanted platelet activation and coagulation (Figure 2). At the level of primary hemostasis, the endothelium elicits strong platelet inhibition by the continuous secretion of nitric oxide (NO) and prostacyclin causing an increase in intraplatelet cGMP (cyclic guanosine monophosphate) and cAMP (cyclic adenosine monophosphate) levels, respectively.23 To prevent platelet activation by extracellular ATP and ADP, the endothelial layer expresses CD39 and CD73, exonucleases that convert ATP and ADP to adenosine, a platelet inhibitor that by increasing platelet cAMP levels elevates the platelet’s activation threshold.24 In addition, the endothelial glycocalyx repels platelets by its negative charge and as such aids in the prevention of platelet adhesion.25,26 At the level of secondary hemostasis, within the glycocalyx, heparan sulfate proteoglycans bind and promote the activity of ATIII (antithrombin III), a potent inhibitor of multiple coagulation factors including thrombin, FIXa, FXa, FXIa, and FXIIa.26,27 Furthermore, endothelial cells express TFPI (tissue factor pathway inhibitor), a serine protease that—as its name suggests—interferes with TF (tissue factor)-induced coagulation and thereby limits the activity of the extrinsic pathway.27 Next to TFPI, endothelial cells constitutively express thrombomodulin, a membrane-bound protein that captures thrombin from the circulation and upon binding increases the affinity of thrombin for the anticoagulant protein C. Together with protein S, activated protein C disables FVa and FVIIIa.27
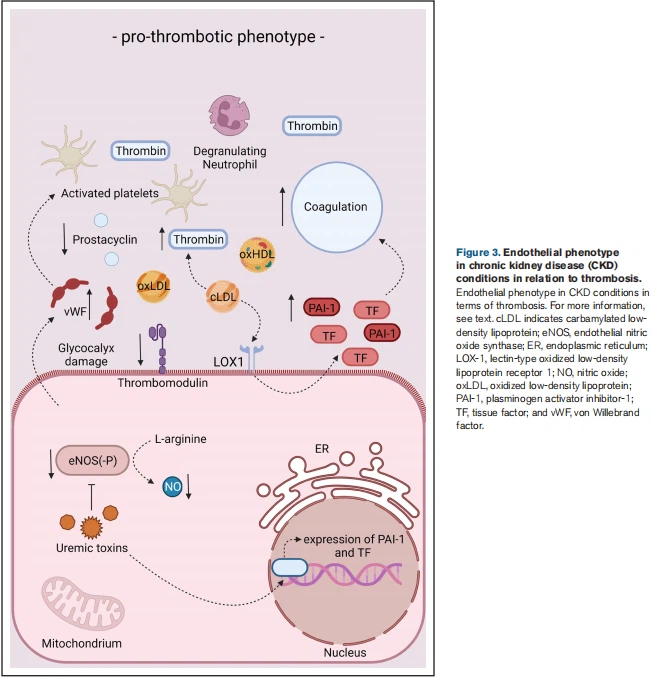
Upon disturbances in lipid metabolism, inflammation, oxidative stress, and pathophysiological shear stress, endothelial cell dysfunction develops, characterized by a diminishment of antithrombotic and anti-inflammatory properties and degradation of the glycocalyx. Concomitantly, the dysfunctional endothelium takes on proinflammatory and prothrombotic characteristics (Figures 3 and 4).27 As a result, the production of NO and prostacyclin decreases, while the secretion of prothrombotic and proinflammatory molecules like vWF (von Willebrand factor) and C-C motif chemokine ligand 2 increases.27 Furthermore, the expression of thrombomodulin is strongly downregulated upon endothelial cell dysfunction resulting in a downregulation of protein C activation, whilst the expression of TF is upregulated favoring the activation of coagulation.27 Already during the early stages of atherogenesis, neutrophil extracellular traps are implicated in endothelial cell dysfunction and fuel the thromboinflammatory response.28

All in all, these prothrombotic and proinflammatory responses of the dysfunctional endothelium spiral out of control, creating a vicious circle in which endothelial cell dysfunction progresses and vascular integrity is lost, resulting in a strongly increased thrombotic risk.
3. Endothelial Dysfunction, Reduced Vasorelaxation, Increased Vascular Stiffness, and Cardiovascular Risk
Aging as well as pathophysiological remodeling of the vascular wall by cardiovascular risk factors (eg, hypertension, diabetes, kidney disease) induces arterial stiffening, which reduces arterial compliance and increases pulsative shear and pressure on the vasculature. On a structural level, arterial stiffness is characterized by collagen deposition and elastin degradation in the ECM.29 Furthermore, vascular smooth muscle cell (VSMC) tone and endothelial dysfunction impact vascular reactivity, with an impaired relaxation capacity of VSMCs as well as a reduced endothelium-dependent vasorelaxation contributing to arterial stiffness. Endothelial cells play an important role in vasorelaxation through NO production by eNOS (endothelial NO synthase; Figure 2), with reduced production and/or bioavailability of NO or other vasodilatory substances reducing the vascular relaxation capacity (Figure 5).

The gold standard for quantifying arterial stiffness is the analysis of blood flow rate, measured as pulse wave velocity (PWV, analyzed as carotid-femoral PWV or brachial-ankle PWV). Alternatively, the shape of the arterial pressure waveform (pulse wave analysis) provides insights into arterial stiffness through quantification of the augmentation index. Endothelial function in terms of vasodilatory responses can be measured either invasively or noninvasively within the epicardial coronary arteries, the peripheral conduit arteries (analyzing flow-mediated dilation [FMD] of the brachial artery as gold standard), or within the coronary or peripheral microvasculature (Figure 6). Peripheral and coronary endothelial dysfunction has been reported to correlate with each other,30,32 although others have reported rather modest correlations between peripheral and coronary endothelial dysfunction and suggested a potential reflection of different pathologies or vascular beds.33 Furthermore, whereas PWV and the augmentation index were shown to correlate well, this was not always the case for the augmentation index and the “reactive hyperemia index” as noninvasive endothelial functional readout in the periphery, potentially due to the impact of other factors than endothelial function on vascular stiffness.34

Vascular stiffness is an early prognostic marker of coronary heart disease.35 Vascular stiffness increases the load on the heart; it urges a greater energy demand for cardiac ejection and triggers cardiac hypertrophy.29 With increased arterial stiffness being associated with a reduced Windkessel function (ie, a reduction in the elastic buffering capacity required to dampen blood pressure fluctuations), arterial stiffness also imposes an increased pulsatility on the microcirculation and enhanced cyclical stretch on endothelial cells. Recently, it was shown that the latter triggers the secretion of protein GAS6 (growth arrest-specific 6) by endothelial cells with subsequent proinflammatory GAS6/Axl signaling in monocytes and their transformation to macrophages and dendritic cells. As shown in a mouse model of Angiotensin II-induced chronic aortic remodeling, this then contributed to vascular and kidney inflammation,36,37 linking arterial stiffness to further end-organ damage.
As endothelial dysfunction is an early event in CVD that precedes macrovascular complications, also endothelial functional analyses in terms of vascular tone regulation have been intensively studied for a predictive capacity of cardiovascular risk beyond traditional cardiovascular risk factors. In multiple studies, macro and microvascular endothelial dysfunction could independently predict cardiovascular events in patients at risk for coronary artery disease.31 For example, in relation to endothelial reactivity of the conduit arteries (microvasculature), medium to low endothelial function recorded by brachial artery FMD was associated with an increased risk of cardiac events in patients with peripheral artery disease undergoing vascular surgery.38 Also in patients with chronic heart failure, endothelial dysfunction analyzed by brachial and radial FMD was associated with future cardiac events or mortality.39–41 Others showed healthy endothelial function recorded by brachial FMD in apparently healthy individuals to result in a significantly higher survival over a 5-year follow-up, even after adjustment for traditional risk factors.42,43 Thus, endothelial cell dysfunction can add important information on cardiovascular risk beyond traditional risk factors.30 Of note, this value of endothelial dysfunction analysis for cardiovascular risk prediction seems to be influenced by the analyzed patient cohort, since others could not confirm a cardiovascular risk prediction value for FMD-based analysis of endothelial dysfunction in an elderly cohort, potentially due to reduced arterial compliance (i.e. the vascular ability to expand upon pressure increases) in elderly subjects.44 Nonetheless, this study did report an association of peripheral microvascular endothelial dysfunction with increased cardiovascular risk, as discussed in the next paragraph in more detail.

Cistanche powder
4. Microvascular Dysfunction and Cardiovascular Risk
Whereas larger arteries are predominantly affected by atherosclerotic changes, dysfunction of the microvasculature—the network of arterioles, capillaries, and venules enabling tissue perfusion—is different. Microvascular dysfunction (MVD) can develop at the level of the coronary microcirculation as well as in the periphery in the absence or presence of obstructive artery disease of the larger vessels. The development of MVD is multifactorial and can be the result of functional or structural changes or a combination thereof, depending on the underlying disease. In terms of functional changes, impaired vasodilatory responses—at least partially endothelium-dependent—underlie MVD. Furthermore, increases in the levels of vasoconstrictive substances in combination with enhanced responsiveness toward these stimuli have been implicated in MVD, contributing to the occurrence of vascular spasms. Structural changes associated with MVD encompass luminal narrowing of the microvasculature due to adverse vascular remodeling and perivascular fibrosis; microvascular compression; microvascular rarefaction resulting in a loss of coherent microvascular trees (capillaries, small arterioles, and venules); and microembolization of atherosclerotic and thrombotic material.45–48
As for macrovascular endothelial dysfunction, also microvascular endothelial dysfunction has been associated with cardiovascular risk.49 This was for example the case in a population-based prospective study, in which peripheral microvascular endothelial dysfunction—but not FMD-mediated endothelial function analysis of conduit arteries—correlated with cardiovascular events (myocardial infarction, stroke, or death) in elderly patients during 5 years of follow-up, even beyond major cardiovascular risk factors from the Framingham risk score.44 Along the same line, peripheral microvascular endothelial dysfunction predicted ischemic heart disease and even performed better in future cardiovascular risk prediction in nonobstructive coronary artery disease compared with other risk scores.50 Also, retinal arteriolar endothelial dysfunction independently predicted MACE in patients with or at high risk of coronary artery disease.51
Also about heart failure, both patients with heart failure with reduced left ventricular ejection fraction as well as patients with heart failure with preserved ejection fraction present with microvascular dysfunction. In failure with reduced left ventricular ejection fraction, peripheral microvascular endothelial function analysis was associated with an increased rate of HF-related events.52 Patients with heart failure with preserved ejection fraction present with both large vessel as well as microvascular dysfunction.53 Furthermore, they display a reduction in cardiac microvascular density54 as well as an increase in markers of endothelial cell dysfunction in myocardial biopsies, including the upregulation of endothelial adhesion molecules (E-selectin, ICAM-1), pro-oxidative regulators (NOX-2) and eNOS uncoupling.55 Coronary microvascular dysfunction has been proposed to contribute to cardiac wall stiffening and diastolic dysfunction in heart failure with preserved ejection fraction patients. However, whether this involves a real causal relation or rather a non-causal association between coronary microvascular dysfunction and heart failure with preserved ejection fraction remains debated.56,57 Of note, coronary microvascular dysfunction may also have a pathophysiological and prognostic role in other types of CVD, for which we refer to an excellent recent review by Del Buono et al.46

Herba Cistanche and Cistanche extract
5. Endothelial Cell Dysfunction and Vascular Aging
Endothelial cell dysfunction with reduced endothelial vasodilation capacity increased inflammation and permeability and enhanced prothrombotic properties, all described above, is an important hallmark of vascular aging. Endothelial cell dysfunction is not only observed in macrocirculation but also contributes to microvascular dysfunction upon aging. Vascular aging is furthermore characterized by functional and structural changes of the vascular wall and adventitia by processes of e.g. inflammation, vascular calcification, and ECM remodeling, which further contribute to increased vascular stiffness and cardiovascular risk.58
REFERENCES
1. Inker LA, Astor BC, Fox CH, Isakova T, Lash JP, Peralta CA, Kurella Tamura M, Feldman HI. KDOQI US commentary on the 2012 KDIGO clinical practice guideline for the evaluation and management of CKD. Am J Kidney Dis. 2014;63:713–735. doi: 10.1053/j.ajkd.2014.01.416
2. Hill NR, Fatoba ST, Oke JL, Hirst JA, O-Callaghan CA, Lasserson DS, Hobbs FDR. The global prevalence of chronic kidney disease - a systematic review and meta-analysis. PLoS One. 2016;11:e0158765. doi: 10.1371/journal.pone.0158765
3. Thompson S, James M, Wiebe N, Hemmelgarn B, Manns B, Klarenbach S, Tonelli M; Alberta Kidney Disease Network. Cause of death in patients with reduced kidney function. J Am Soc Nephrol. 2015;26:2504–2511. doi: 10.1681/ASN.2014070714
4. Roberts MA, Polkinghorne KR, McDonald SP, Ierino FL. Secular trends in cardiovascular mortality rates of patients receiving dialysis compared with the general population. Am J Kidney Dis. 2011;58:64–72. doi: 10.1053/j.ajkd.2011.01.024
5. van der Velde M, Matsushita K, Coresh J, Astor BC, Woodward M, Levey A, de Jong P, Gansevoort RT, Matsushita K, Coresh J, et al; Chronic Kidney Disease Prognosis Consortium. Lower estimated glomerular filtration rate and higher albuminuria are associated with all-cause and cardiovascular mortality. A collaborative meta-analysis of high-risk population cohorts. Kidney Int. 2011;79:1341–1352. doi 10.1038/ki.2010.536
6. Jankowski J, Floege J, Fliser D, Bohm M, Marx N. Cardiovascular disease in chronic kidney disease: pathophysiological insights and therapeutic options. Circulation. 2021;143:1157–1172. doi: 10.1161/CIRCULATIONAHA.120.050686
7. Weber C, Noels H. Atherosclerosis: current pathogenesis and therapeutic options. Nat Med. 2011;17:1410–1422. doi: 10.1038/nm.2538
8. Quillard T, Franck G, Mawson T, Folco E, Libby P. Mechanisms of erosion of atherosclerotic plaques. Curr Opin Lipidol. 2017;28:434–441. doi: 10.1097/MOL.0000000000000440
9. Betrui A, Martinez-Alonso M, Arcidiacono MV, Cannata-Andia J, Pascual J, Valdivielso JM, Fernández E; Investigators from the NEFRONA Study. Prevalence of subclinical atheromatosis and associated risk factors in chronic kidney disease: the NEFRONA study. Nephrol Dial Transplant. 2014;29:1415–1422. doi 10.1093/ndt/gfu038
10. Valdivielso JM, Rodríguez-Puyol D, Pascual J, Barrios C, BermúdezLópez M, Sánchez-Niño MD, Pérez-Fernández M, Ortiz A. Atherosclerosis in chronic kidney disease: more, less or just different?. Arterioscler Thromb Vasc Biol. 2019;39:1938–1966. doi: 10.1161/ATVBAHA.119.312705
11. Gracia M, Betrui A, Martinez-Alonso M, Arroyo D, Abajo M, Fernández E, Valdivielso JM; NEFRONA Investigators. Predictors of subclinical atheromatosis progression over 2 years in patients with different stages of CKD. Clin J Am Soc Nephrol. 2016;11:287–296. doi: 10.2215/CJN.01240215
12. Anavekar NS, McMurray JJV, Velazquez EJ, Solomon SD, Kober L, Rouleau JL, White HD, Nordlander R, Maggioni A, Dickstein K, et al. Relation between renal dysfunction and cardiovascular outcomes after myocardial infarction. N Engl J Med. 2004;351:1285–1295. doi: 10.1056/NEJMoa041365
13. Myerburg RJ, Junttila MJ. Sudden cardiac death caused by coronary heart disease. Circulation. 2012;125:1043–1052. doi: 10.1161/CIRCULATIONAHA.111.023846
14. Haider AW, Larson MG, Benjamin EJ, Levy D. Increased left ventricular mass and hypertrophy are associated with increased risk for sudden death. J Am Coll Cardiol. 1998;32:1454–1459. doi: 10.1016/s0735-1097(98)00407-0
15. Glassock RJ, Pecoits-Filho R, Barberato SH. Left ventricular mass in chronic kidney disease and ESRD. Clin J Am Soc Nephrol. 2009;4:S79–S91. doi: 10.2215/CJN.04860709
16. Middleton R, Parfrey PS, Foley RN. Left ventricular hypertrophy in the renal patient. J Am Soc Nephrol. 2001;12:1079–1084. doi 10.1681/ASN.V1251079
17. Gimbrone MA Jr, Garcia-Cardena G. Endothelial cell dysfunction and the pathobiology of atherosclerosis. Circ Res. 2016;118:620–636. doi: 10.1161/CIRCRESAHA.115.306301
18. Meza D, Musmacker B, Steadman E, Stransky T, Rubenstein DA, Yin W. Endothelial cell biomechanical responses are dependent on both fluid shear stress and tensile strain. Cell Mol Bioeng. 2019;12:311–325. doi 10.1007/s12195-019-00585-0
19. Evrard SM, Lecce L, Michelis KC, Nomura-Kitabayashi A, Pandey G, Purushothaman KR, d’Escamard V, Li JR, Hadri L, Fujitani K, et al. Endothelial to mesenchymal transition is common in atherosclerotic lesions and is associated with plaque instability. Nat Commun. 2016;7:11853. doi: 10.1038/ncomms11853
20. Chen PY, Qin L, Baeyens N, Li G, Afolabi T, Budatha M, Tellides G, Schwartz MA, Simons M. Endothelial-to-mesenchymal transition drives atherosclerosis progression. J Clin Invest. 2015;125:4514–4528. doi: 10.1172/JCI82719
21. Bostrom KI, Yao J, Guihard PJ, Blazquez-Medela AM, Yao Y. Endothelialmesenchymal transition in atherosclerotic lesion calcification. Atherosclerosis. 2016;253:124–127. doi 10.1016/j.atherosclerosis.2016.08.046
22. Souilhol C, Harmsen MC, Evans PC, Krenning G. Endothelial-mesenchymal transition in atherosclerosis. Cardiovasc Res. 2018;114:565–577. doi: 10.1093/cvr/cvx253
23. Mitchell JA, Ali F, Bailey L, Moreno L, Harrington LS. Role of nitric oxide and prostacyclin as vasoactive hormones released by the endothelium. Exp Physiol. 2008;93:141–147. doi: 10.1113/expphysiol.2007.038588
24. Johnston-Cox HA, Ravid K. Adenosine and blood platelets. Purinergic Signal. 2011;7:357–365. doi: 10.1007/s11302-011-9220-4
25. Born GV, Palinski W. Unusually high concentrations of sialic acids on the surface of vascular endothelia. Br J Exp Pathol. 1985;66:543–549.
26. Reitsma S, Slaaf DW, Vink H, van Zandvoort MAMJ, oude Egbrink MGA. The endothelial glycocalyx: composition, functions, and visualization. Pfugers Arch. 2007;454:345–359. doi 10.1007/s00424-007-0212-8
27. Yau JW, Teoh H, Verma S. Endothelial cell control of thrombosis. BMC Cardiovasc Disord. 2015;15:130. doi: 10.1186/s12872-015-0124-z
28. Döring Y, Soehnlein O, Weber C. Neutrophil extracellular traps in atherosclerosis and atherothrombosis. Circ Res. 2017;120:736–743. doi: 10.1161/CIRCRESAHA.116.309692
29. Zieman SJ, Melenovsky V, Kass DA. Mechanisms, pathophysiology, and therapy of arterial stiffness. Arterioscler Thromb Vasc Biol. 2005;25:932– 943. doi 10.1161/01.ATV.0000160548.78317.29
30. Flammer AJ, Anderson T, Celermajer DS, Creager MA, Deanfield J, Ganz P, Hamburg NM, Lüscher TF, Shechter M, Taddei S, et al. The assessment of endothelial function: from research into clinical practice. Circulation. 2012;126:753–767. doi: 10.1161/CIRCULATIONAHA.112.093245
31. Alexander Y, Osto E, Schmidt-Trucksass A, Shechter M, Trifunovic D, Duncker DJ, Aboyans V, Back M, Badimon L, Cosentino F, et al. Endothelial function in cardiovascular medicine: a consensus paper of the European Society of Cardiology Working Groups on Atherosclerosis and Vascular Biology, Aorta and Peripheral Vascular Diseases, Coronary Pathophysiology and Microcirculation, and Thrombosis. Cardiovasc Res. 2021;117:29–42. doi: 10.1093/cvr/cvaa085
32. Lerman A, Zeiher AM. Endothelial function: cardiac events. Circulation. 2005;111:363–368. doi 10.1161/01.CIR.0000153339.27064.14
33. Schnabel RB, Schulz A, Wild PS, Sinning CR, Wilde S, Eleftheriadis M, Herkenhoff S, Zeller T, Lubos E, Lackner KJ, et al. Noninvasive vascular function measurement in the community: cross-sectional relations and comparison of methods. Circ Cardiovasc Imaging. 2011;4:371–380. doi: 10.1161/CIRCIMAGING.110.961557
34. Perrault R, Omelchenko A, Taylor CG, Zahradka P. Establishing the interchangeability of arterial stiffness but not endothelial function parameters in healthy individuals. BMC Cardiovasc Disord. 2019;19:190. doi: 10.1186/s12872-019-1167-3
35. Bonarjee VVS. Arterial stiffness: a prognostic marker in coronary heart disease. Available methods and clinical application. Front Cardiovasc Med. 2018;5:64. doi: 10.3389/fcvm.2018.00064
36. Van Beusecum JP, Barbaro NR, Smart CD, Patrick DM, Loperena R, Zhao S, de la Visitacion N, Ao M, Xiao L, Shibao CA, et al. Growth arrest specific-6 and Axl coordinate inflammation and hypertension. Circ Res. 2021;129:975–991. doi: 10.1161/CIRCRESAHA.121.319643
37. Chen W, Van Beusecum JP, Xiao L, Patrick DM, Ao M, Zhao S, Lopez MG, Billings FT, Cavinato C, Caulk AW, et al. Role of Axl in target organ inflammation and damage due to hypertensive aortic remodeling. Am J Physiol Heart Circ Physiol. 2022;323:H917–H933. doi: 10.1152/ajpheart.00253.2022
38. Huang AL, Silver AE, Shvenke E, Schopfer DW, Jahangir E, Titas MA, Shpilman A, Menzoian JO, Watkins MT, Raffetto JD, et al. Predictive value of reactive hyperemia for cardiovascular events in patients with peripheral artery disease undergoing vascular surgery. Arterioscler Thromb Vasc Biol. 2007;27:2113–2119. doi: 10.1161/ATVBAHA.107.147322
39. Fischer D, Rossa S, Landmesser U, Spiekermann S, Engberding N, Hornig B, Drexler H. Endothelial dysfunction in patients with chronic heart failure is independently associated with increased incidence of hospitalization, cardiac transplantation, or death. Eur Heart J. 2005;26:65–69. doi: 10.1093/eurheartj/ehi001
40. Meyer B, Mörtl D, Strecker K, Hülsmann M, Kulemann V, Neunteufl T, Pacher R, Berger R. Flow-mediated vasodilation predicts outcome in patients with chronic heart failure: comparison with B-type natriuretic peptide. J Am Coll Cardiol. 2005;46:1011–1018. doi 10.1016/j.jacc.2005.04.060
41. Katz SD, Hryniewicz K, Hriljac I, Balidemaj K, Dimayuga C, Hudaihed A, Yasskiy A. Vascular endothelial dysfunction and mortality risk in a patient with chronic heart failure. Circulation. 2005;111:310–314. doi 10.1161/01.CIR.0000153349.77489.CF
42. Yeboah J, Crouse JR, Hsu FC, Burke GL, Herrington DM. Brachial flow-mediated dilation predicts incident cardiovascular events in older adults: the Cardiovascular Health Study. Circulation. 2007;115:2390–2397. doi: 10.1161/CIRCULATIONAHA.106.678276
43. Yeboah J, Folsom AR, Burke GL, Johnson C, Polak JF, Post W, Lima JA, Crouse JR, Herrington DM. Predictive value of brachial flow-mediated dilation for incident cardiovascular events in a population-based study: the multi-ethnic study of atherosclerosis. Circulation. 2009;120:502–509. doi: 10.1161/CIRCULATIONAHA.109.864801
44. Lind L, Berglund L, Larsson A, Sundström J. Endothelial function in resistance and conduit arteries and 5-year risk of cardiovascular disease. Circulation. 2011;123:1545–1551. doi: 10.1161/CIRCULATIONAHA.110.984047
45. Vancheri F, Longo G, Vancheri S, Henein M. Coronary microvascular dysfunction. J Clin Med. 2020;9:2880. doi: 10.3390/jcm9092880
46. Del Buono MG, Montone RA, Camili M, Carbone S, Narula J, Lavie CJ, Niccoli G, Crea F. Coronary microvascular dysfunction across the spectrum of cardiovascular diseases: JACC State-of-the-Art review. J Am Coll Cardiol. 2021;78:1352–1371. doi 10.1016/j.jacc.2021.07.042
47. Ehling J, Babickova J, Gremse F, Klinkhammer BM, Baetke S, Knuechel R, Kiessling F, Floege J, Lammers T, Boor P. Quantitative micro-computed tomography imaging of vascular dysfunction in progressive kidney diseases. J Am Soc Nephrol. 2016;27:520–532. doi 10.1681/ASN.2015020204
48. Prommer HU, Maurer J, von Websky K, Freise C, Sommer K, Nasser H, Samapati R, Reglin B, Guimaraes P, Pries AR, et al. Chronic kidney disease induces systemic microangiopathy, tissue hypoxia, and dysfunctional angiogenesis. Sci Rep. 2018;8:5317. doi: 10.1038/s41598-018-23663-1
49. Querfeld U, Mak RH, Pries AR. Microvascular disease in chronic kidney disease: the base of the iceberg in cardiovascular comorbidity. Clin Sci (Lond). 2020;134:1333–1356. doi 10.1042/CS20200279
50. Matsuzawa Y, Sugiyama S, Sugamura K, Nozaki T, Ohba K, Konishi M, Matsubara J, Sumida H, Kaikita K, Kojima S, et al. Digital assessment of endothelial function and ischemic heart disease in women. J Am Coll Cardiol. 2010;55:1688–1696. doi 10.1016/j.jacc.2009.10.073
51. There JD, Al-Fiadh AH, Amirul Islam FM, Patel SK, Burrell LM, Wong TY, Farouque O. Impaired retinal microvascular function predicts long-term adverse events in patients with cardiovascular disease. Cardiovasc Res. 2021;117:1949–1957. doi: 10.1093/cvr/cvaa245
52. Fujisue K, Sugiyama S, Matsuzawa Y, Akiyama E, Sugamura K, Matsubara J, Kurokawa H, Maeda H, Hirata Y, Kusaka H, et al. Prognostic significance of peripheral microvascular endothelial dysfunction in heart failure with reduced left ventricular ejection fraction. Circ J. 2015;79:2623–2631. doi: 10.1253/circa.CJ-15-0671
53. Maréchaux S, Samson R, van Belle E, Breyne J, de Monte J, Dédrie C, Chebai N, Menet A, Banfi C, Bouabdallaoui N, et al. Vascular and microvascular endothelial function in heart failure with preserved ejection fraction. J Card Fail. 2016;22:3–11. doi: 10.1016/j.cardfail.2015.09.003
54. Mohammed SF, Hussain S, Mirzoyev SA, Edwards WD, Maleszewski JJ, Redfield MM. Coronary microvascular rarefaction and myocardial fibrosis in heart failure with preserved ejection fraction. Circulation. 2015;131:550– 559. doi: 10.1161/CIRCULATIONAHA.114.009625
55. Franssen C, Chen S, Unger A, Korkmaz HI, De Keulenaer GW, Tschöpe C, Leite-Moreira AF, Musters R, Niessen HWM, Linke WA, et al. Myocardial microvascular inflammatory endothelial activation in heart failure with preserved ejection fraction. JACC Heart Fail. 2016;4:312–324. doi: 10.1016/j.jchf.2015.10.007
56. D’Amario D, Migliaro S, Borovac JA, Restivo A, Vergallo R, Galli M, Leone AM, Montone RA, Niccoli G, Aspromonte N, et al. Microvascular dysfunction in heart failure with preserved ejection fraction. Front Physiol. 2019;10:1347. doi: 10.3389/Phys.2019.01347
57. Cornuault L, Rouault P, Duplàa C, Couffinhal T, Renault MA. Endothelial dysfunction in heart failure with preserved ejection fraction: what are the experimental proofs? Front Physiol. 2022;13:906272. doi: 10.3389/Phys.2022.906272
58. Harvey A, Montezano AC, Alves Lopes R, Rios F, Touyz RM. Vascular fibrosis in aging and hypertension: molecular mechanisms and clinical implications. Can J Cardiol. 2016;32:659–668. doi 10.1016/j.cjca.2016.02.070
Constance C.F.M.J. Baaten, Sonja Vondenhoff, Heidi Noels






