Protective Effect Of Cistanche Deserticola On Liver Injury: Alcoholic Liver Injury
Mar 19, 2022
Abstract: Modern pharmacological studies have shown that Cistanche deserticola (C. deserticola) has a protective effect on the liver, but its active fraction and mechanism are not clear. In order to identify the effective fraction of C. deserticola Y. C. Ma, an acute alcoholic liver injury model in mice was established with 56-proof Erguotou and different fractional extracts of C. deserticola Y. C. Ma (total glycosides, polysaccharides, and oligosaccharides) were administered. After 14 days of oral administration, liver pathology and lipid deposition were measured and the expression of nuclear factor E2-related factor (Nrf-2), kelch-like ECH-associated protein-1 (Keap-1), and plasmalemma vesicle-associated protein-1 (PV1) were measured by immunofluorescence. The levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), endotoxin (ET), diamine oxidase (DAO), and D-lactic acid (D-LA) in serum, and superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), catalase (CAT), and malondialdehyde (MDA) in the liver were measured by ELISA. All animal experiments were carried out with the approval of the Experimental Animal Welfare Ethics Committee of the Peking University Health Science Center. The results show that the total glycosides of C. deserticola Y. C. Ma (400 mg·kg-1) could decrease liver pathology, decrease serum endotoxin, diamine oxidase, and D-lactic acid, and reduce hepatic lipid deposition. Total glycosides also promoted Nrf-2 transfer into the nucleus and decreased the expression of Keap-1 and PV1. In summary, the total glycosides of C. deserticola Y. C. Ma had a protective effect in acute alcoholic liver injury and the mechanism may be related to the activation of the Nrf-2/Keap-1 pathway, improvement of intestinal wall integrity, and inhibition of the transport of harmful substances into the liver.
Keywords: Cistanche deserticola Y. C. Ma; alcoholic liver disease; total glycosides of C. deserticola Y. C.Ma; liver protection; Nrf-2/Keap-1 pathway
Protective effects of the total glycosides of Cistanche deserticola Y. C. Ma in alcoholic liver injury in mice
Contact: ali.ma@wecistanche.com

Alcoholic liver disease (ALD) refers to the liver disease caused by long-term heavy drinking, which usually manifests as alcoholic fatty liver in the initial stage, and then develops into alcoholic hepatitis, liver fibrosis, liver cirrhosis, and even liver failure[1]. In recent years, with the improvement of people's living standards, the proportion of patients with alcoholic liver disease has been increasing, seriously endangering people's health [2]. Studies have found that ALD is mainly the result of the interaction of multiple factors such as inflammatory response, oxidative stress, and gut-derived endotoxins directly or indirectly induced by ethanol and its metabolites, especially gut-derived gut-derived endotoxins caused by impaired intestinal barrier function. Endotoxemia and endotoxin activation of Kupffer cells play an important role in the occurrence and development of ALD [3].
The traditional Chinese medicine Cistanche is the succulent stem of Cistanche deserticola (C. deserticola) Y. C. Ma or Cistanche tubulosa (Schenk) R. Wight of the Lidanaceae plant with dry scaly leaves. Studies have shown that Cistanche can enhance the body's immune function, anti-aging, anti-radiation, anti-oxidation, and anti-lipid peroxidation and protection against alcoholic liver damage [4-7]. Previous studies have mainly focused on Cistanche deserticola, and the hepatoprotective effect of Cistanche deserticola and its active components are not clear. In this paper, based on the acute alcoholic liver injury model, the hepatoprotective effect and mechanism of extracts from different parts of Cistanche deserticola were discussed, and the theoretical basis for the later research and development of Cistanche deserticola in liver protection was provided.

Materials and Methods
Laboratory Animal Healthy Kunming mice, male, weighing (22 ± 5) g, were purchased from Beijing Weitong Lihua Laboratory Animal Technology Co., Ltd., the license number is SCXK (Beijing) 2016-0011, and raised in the Animal Experiment Center of Peking University Medical Department, room temperature 20 ~ 25 ℃, humidity 56% ~ 60%. All operations on experimental animals were performed in strict accordance with the standards of the Animal Ethics Committee of Peking University School of Medicine (LA2019123).
For the preparation and content determination of Cistanche deserticola extract. The preparation method and content determination method of Cistanche deserticola extract previously reported by the research group were referred to [8-10]. 10 kg of Cistanche deserticola was extracted with water and ethanol precipitation to obtain 3.7% polysaccharides, and the eluate in purified water was concentrated to obtain 38% thick paste of total oligosaccharides, and 40% ethanol was eluted and dried to obtain 3.7% total glycosides. These 3 extracts were used for experimental research respectively.
(G1260), alanine aminotransferase (ALT) activity detection kit (batch number BC1555) and aspartate aminotransferase (AST) activity detection kit (batch number BC1560) were purchased from Beijing Soleibo Biotechnology Co., Ltd. Company; Superoxide Dismutase (SOD) Kit (Lot A001-3), Glutathione Peroxidase (GSH-Px) Kit (Lot A005), Malondialde (Lot A005) ‐ hyde, MDA) kit (Lot A003-1), catalase (catalase, CAT) detection kit (A007-1), endotoxin (ET) kit (E039- 1- 1) , diamine oxidase (DAO) kit (A088-2-1) and D-lactic acid (D-LA) kit (H263) were all from Nanjing Jiancheng Institute of Bioengineering; Biphenyl Diester (bifendate, BIF, batch number: S4890) was provided by Shanghai Lanmu Chemical Co., Ltd.; 56% (alcohol content 56%) Hongxing Erguotou was provided by Beijing Hongxing Co., Ltd.; plasma membrane vesicle-associated protein 1 (plasma‐lemma vesicle) -associated protein-1, PV1) antibody (ab32570), nuclear factor E2 related factors (Nrf-2) antibody and kelch-like ECH-asso-ciated protein-1 (Keap) -1) (ab66620) were purchased from Abcam.
Experimental instruments Paraffin microtome (Leica, Germany); Sunrise-Basic microplate reader (TECAN, Switzerland); high-speed refrigerated centrifuge (Beckman, USA); inverted fluorescence microscope (IX73) (Olympus, Japan).
Preparation and grouping of the mouse model of acute alcoholic liver injury Sixty healthy male Kunming mice were weighed, 10 in the normal group (NOR), and the rest of the mice were given Hongxing Erguotou by gavage [9], at a dose of 0.01 mL g - 1 dose by gavage, once a day for 7 consecutive days. After 7 days, they were randomly divided into model group (MOD), bidentate group (BIF, 0.9 mg kg-1) and total glycosides group (TGs, 400 mg kg-1) according to body weight. ), Cistanche deserticola polysaccharide group (PSs, 400 mg·kg-1) and Cistanche deserticola oligosaccharide group (OSs, 400 mg·kg-1), and the dosage was set according to the previous research of the research group[11]. Each group was given normal saline, biphenyl diester, total glucosides of Cistanche deserticola, polysaccharide of Cistanche deserticola, and oligosaccharide of Cistanche deserticola respectively by gavage for 14 days. The medicines were all prepared with normal saline. Due to continuous gavage of alcohol, some mice died, and the last administration Experimental reagents HE staining solution (G1120), Oil red O staining solution, 8 per group.
30 min after the last administration, each organ coefficient of mice was obtained, and each organ was obtained according to this formula: organ coefficient = [organ wet weight (g)/rat body weight (g)] × 100% to calculate each organ Statistical analysis was performed using SPSS 19.0 software.

HE staining After the above-mentioned liver tissues were fixed with tissue fixative, the sections were routinely embedded in paraffin, stained with hematoxylin-eosin (HE), and histopathological changes were observed under a light microscope.
For index detection, the serum of mice in each group was taken, and the contents of ALT, AST, ET, DAO, and D-LA were detected according to the kit instructions. The mice were sacrificed to remove the liver, and liver tissue homogenate was prepared. According to the kit instructions, CAT, SOD, GSH- Px activity, and MDA content.
Oil red O staining took the fixed mouse liver tissue and placed it in a 30% sucrose solution. After the liver tissue settled to the bottom, it was transferred to a 20% sucrose solution. , the slice thickness is 100 μm. Add 1×PBS dropwise to wash for 10 min, then put it into 60% isopropanol for immersion for 20-30 s; then add it to the modified oil red O staining solution, and seal it for 10-15 min; put it in 60% isopropanol solution for a while Washed to remove the dye solution, rinsed with tap water for 10 min, mounted with glycerol gelatin, and observed and photographed under a microscope.
The paraffin sections were dewaxed and rehydrated for immunofluorescence chemical staining, and the antigen was retrieved with sodium citrate. Primary antibodies PV1 (1:200), Nrf-2 (1:100), and Keap-1 (1:200) were added dropwise, and the cells were incubated at 4 ℃ overnight. Rinse with PBS, add TRITC fluorescent secondary antibody (1:100) dropwise, and incubate for 1 h at room temperature in the dark. Nuclei were stained with Hoechst 33258, mounted with anti-fluorescence decay mountant in the dark, and photographed under a fluorescence microscope.
TUNEL-stained paraffin sections were dewaxed and rehydrated, 20 μg·mL-1 of DNase-free proteinase K dilution was added dropwise, the reaction was carried out at 37 °C for 30 min, and then 3% H2O2 solution was added dropwise and incubated at room temperature for 20 min. Wash three times with 0.01 mol·L- 1 PBS buffer solution; add 50 μL biotin labeling solution dropwise, incubate at 37 °C for 60 min to stop the reaction; add 50 μL streptavidin-TRITC working solution dropwise, incubate at room temperature for 30 min in the dark, Nuclei were stained with DAPI, mounted on slides, observed under a microscope, and photographed with a fluorescence microscope.
Statistical analysis was performed using SPSS 19.0 software. The measurement data were expressed as mean ± standard deviation (x-±s), and the measurement data with normal distribution were compared by one-way analysis of variance (ANOVA) between groups. If the variance was homogeneous, the SNK method was used, and if the variance was unequal, Dunnett's method was used. T3 method, count data using the chi-square test (chi-square test), using GraphPad Prism 5 for data mapping, the least significant difference level was set at P < 0.05.
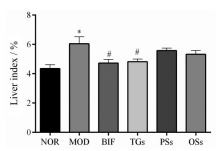
Result
1 TGs can significantly reduce the liver coefficient of alcoholic liver injury mice. As shown in Figure 1, compared with the NOR group, the liver coefficient of the MOD group is significantly
Compared with the MOD group, the liver coefficient was significantly reduced after TGs intervention, while PSs and OSs had no effect on the liver coefficient, and there was no significant difference between TGs and BIF.
2 TGs can significantly improve the pathological morphology of liver tissue in mice with alcoholic liver injury
HE staining results showed that the liver tissue cells in the NOR group had normal morphology, neatly arranged cells, uniform staining, complete cell structure, and dense intercellular spaces; the MOD group had disordered structure, irregular cell arrangement, loose intercellular spaces, and unclear cell boundaries; BIF and TGs After the intervention, the change of cell morphology was significantly better than that of the model group, and the number of cells was more than that of the model group. There was no significant difference between TGs and BIF, and the PSs and OSs groups had no significant improvement on the pathological morphology of the liver, indicating that TGs had hepatoprotective effects. However, PSs and OSs showed no significant improvement, as shown in Figure 2.
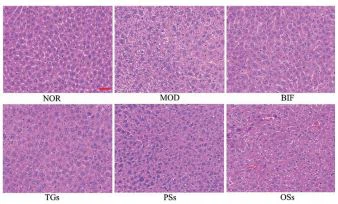
3 TGs can significantly reduce liver lipid deposition in mice with alcoholic liver injury
Oil red O staining showed that there was a small amount of lipid deposition in the NOR group, while the lipid deposition in the MOD group was significantly increased; compared with the MOD group, lipid deposition was significantly reduced after TGs intervention; there was no significant difference between TGs and BIF; PSs and OSs did not significantly alter lipid deposition in the liver. The experimental results showed that TGs could reduce liver lipid deposition in alcoholic liver injury mice, while PSs and OSs did not improve liver lipid deposition in alcoholic liver injury mice, and had no lipid-lowering effect (Figure 3).

4 TGs can significantly reduce liver ALT and AST levels in mice with alcoholic liver injury
The results shown in Figure 4 show that compared with the NOR group, the ALT and AST levels in the MOD group were significantly increased; compared with the MOD group, the ALT and AST levels in the TGs group were significantly decreased; there was no significant difference between TGs and BIF; The levels of ALT and AST did not change significantly, indicating that TGs inhibited liver damage and had hepatoprotective effects.
5 TGs can significantly inhibit the apoptosis of liver cells in mice with alcoholic liver injury
After alcohol damages the hepatocytes, the DNA in the nucleus is broken, and TUNEL staining shows positive expression. As shown in Figure 5, a large number of cells in the liver of the MOD group underwent apoptosis. Compared with the MOD group, the apoptosis of the TGs group was significantly reduced, and there was no significant difference between the TGs and BIF. There was no significant change, indicating that TGs could inhibit apoptosis.
6 TGs can significantly improve the pathological morphology of the small intestine in mice with alcoholic liver injury
HE staining results showed that the small intestinal histology cells in the NOR group had normal morphology, neatly arranged villi, uniform intestinal wall, intact cell structure, and dense intercellular spaces; the MOD group had disordered structure, irregularly arranged villi, fractured, and thin intestinal wall; BIF and TGs After the intervention, the cell morphology was significantly changed, and the intestinal wall was thickened. The PSs and OSs groups did not significantly improve the pathological morphology of the small intestine, indicating that TGs had a protective effect on the integrity of the intestinal wall and villi as shown in Figure 6.


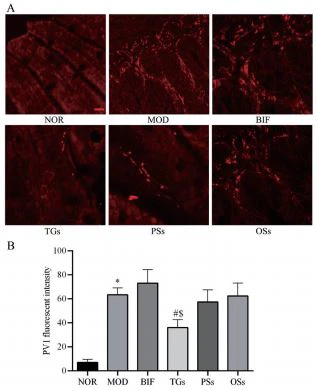
7 TGs significantly reduce PV1 protein expression in the small intestinal wall of mice with alcoholic liver injury
PV1 protein is a type II integral membrane glycoprotein, which is an indicator of intestinal vascular barrier permeability, and its expression was detected by immunofluorescence staining to observe the effect of Cistanche deserticola extract on intestinal wall permeability. As shown in Figure 7, compared with the NOR group, the PV1 protein expression level in the small intestinal wall of the MOD group was significantly increased; compared with the MOD group, the PV1 protein expression level of the TGs group was significantly decreased; TGs had significant differences compared with BIF; while PSs There was no significant change in PV1 protein expression between the two groups and the OSs group, indicating that TGs can promote the repair of the small intestinal wall.

8 TGs can significantly improve serum ET, DAO, and D-LA contents in mice with alcoholic liver injury
The results shown in Figure 8 show that compared with the NOR group, the serum levels of ET, DAO, and D-LA in the MOD group were significantly increased; compared with the MOD group, the levels of ET, DAO, and D-LA in the TGs group were significantly decreased; There was no significant change in the content of these indicators in the group.
9 TGs activate the Nrf-2/Keap-1 signaling pathway in liver tissue of mice with alcoholic liver injury
The results of immunofluorescence staining showed that compared with the NOR group, the Nrf-2 nuclear entry rate in the liver of the MOD group was significantly reduced; the protein expression level of Keap-1 was significantly increased; compared with the MOD group, the Nrf-2 nuclear entry rate of the TGs group was significantly increased; Keap-1 protein expression level was significantly decreased; TGs had no significant difference compared with BIF; PSs and OSs groups had no significant effect on Nrf-2 nuclear entry and Keap-1 expression, as shown in Figure 9.
10 TGs can significantly improve the levels of oxidative stress factors in liver tissue of mice with alcoholic liver injury
This experimental study showed that compared with the NOR group, the activities of SOD, CAT, and GSH-Px in the MOD group were significantly decreased, and the content of MDA was significantly increased; compared with the MOD group, the activities of SOD, CAT, and GSH-Px were significantly increased after BIF and TGs intervention increased, the MDA content decreased significantly, as shown in Figure 10.
Discussion
In this study, a mouse model of acute alcoholic liver injury was used to investigate the effects of extracts from different parts of Cistanche deserticola on the acute alcoholic liver injury when administered for 14 days. , ALT and AST content in serum and MDA content in liver tissue, significantly increased the activities of antioxidant enzymes SOD, CAT, and GSH-Px, improved the morphological structure of liver tissue and intestinal wall permeability, decreased serum ET, DAO, and D -LA content, inhibits the transfer of bacterial secondary metabolites to the liver. At the same time, the total glucosides of Cistanche deserticola can significantly promote the entry of Nrf-2 into the nucleus; inhibit the expression of Keap-1 protein and PV1 protein in the intestinal wall, indicating that the total glucosides of Cistanche deserticola can activate the Nrf-2/Keap-1 signaling pathway and repair the intestinal wall. damage to achieve liver protection.
The traditional Chinese medicine Cistanche is the dry and scaly stalk of Cistanche deserticola or Cistanche Cistanche, and the total glycosides of these two types of Cistanche in liver protection can improve the liver tissue morphology and reduce the degree of fibrosis in fibrotic rats[12]. Luo Huiying's research group[13] conducted some studies on Cistanche deserticola, and the results showed that total glucosides of Cistanche deserticola could enhance the activities of SOD, GSH-Px, and CAT in liver tissue of mice with alcoholic liver injury, which was the same as the results of this study. The chemical constituents of the two are different only in the content of phenylethanoid glycosides, and the most studied is Cistanche piensis. Studies have shown that the liver protective components of Cistanche deserticola are mainly phenylethanoid glycosides, but because the content of phenylethanoid glycosides in Cistanche deserticola is lower than that of Cistanche deserticola, there are not many people who have studied it. If the content is lower than that of Cistanche Cistanche, whether it still has a hepatoprotective effect. A comparative study of other components was carried out to clarify the liver protective components, and also to provide an experimental basis for the effect of Cistanche deserticola in liver protection, and to provide a theoretical basis for later product development.

The development of liver disease is closely related to the Nrf-2/Keap-1 signaling pathway, especially alcoholic liver injury. Studies have found that ethanol can induce the expression of sterol regulatory element-binding protein 1 (SREBP-1) in Nrf-2-/- mice, and can also significantly increase serum triglyceride levels[14]. Long-term alcohol consumption can lead to the activation of Nrf-2 in the liver of mice, thereby reducing ethanol-induced oxidative stress. Oxidative stress is also closely related to the occurrence and development of liver diseases. When the body is in a state of oxidative stress, Nrf-2 binds to nuclear antioxidant response elements (AREs) and then initiates the expression of downstream antioxidant genes. Compared with wild-type mice, Nrf-2-/- mice are more sensitive to liver damage due to their reduced antioxidant capacity and are more susceptible to external stimuli. However, the overexpression of Nrf-2 protein showed no obvious liver injury[15]. It can be seen that the activation of Nrf-2 may be a new potential therapeutic target and pathway for liver injury. In this study, by comparing the effects of three extracts of Cistanche deserticola on acute alcoholic liver injury, it was found that total glucosides of Cistanche deserticola could increase the activities of antioxidant enzymes SOD, CAT, and GSH-Px by activating the Nrf-2/Keap-1 signaling pathway and thus play a role in the activation of Nrf-2/Keap-1 signaling pathway. The role of liver protection.
Alcohol abuse is a major factor in causing alcoholic liver injury, and alcohol can promote the overgrowth of gram-negative bacteria in the gut of patients with chronic liver injury. The portal vein reaches the liver, thereby aggravating liver damage[16]. Studies have found that in ALD patients and animal models, both alcohol and its metabolite acetaldehyde can increase intestinal permeability and allow harmful substances such as lipopolysaccharide (LPS) and D-LA to enter the blood [17]. This study found that the total glucosides of Cistanche deserticola could reduce the permeability of alcohol in the intestinal tract, thereby reducing the content of LPS, DAO, and D-LA in the blood, and preventing harmful substances from entering the liver through the portal vein, thereby playing a role in liver protection.

Bifendate is the first drug for the treatment of hepatitis in my country. It has pharmacological effects such as enzyme-lowering, anti-oxidation, and immune regulation. This study also found that bidentate has a significant effect on lipid deposition, and its effect may be related to its anti-inflammatory effect. Oxidation is related to the improvement of damaged liver tissue, thereby improving lipid metabolism in the liver, but whether it directly regulates lipid metabolism has not been reported. At the same time, it can be seen from the results that the hepatoprotective effect of total glucosides of Cistanche deserticola is not significantly different from that of bidentate, and both have antioxidative effects and reduce the contents of ET, DAO, and D-LA in serum, which means that the total glucosides of Cistanche deserticola have no significant difference. There are differences in the similar liver protection between glycosides and bidentate, which indicates that bidentate has no significant effect on reducing intestinal permeability, and its mechanism of reducing serum ET, DAO, and D-LA remains to be further studied.

To sum up, the total glucosides of Cistanche deserticola is the active site of the traditional Chinese medicine Cistanche deserticola, which can reduce oxidative stress in the liver and inhibit the entry of harmful intestinal substances into the liver, and play a protective role in acute alcoholic liver injury. Its mechanism may be related to It is related to the activation of the Nrf-2/Keap-1 signaling pathway; while Cistanche deserticola polysaccharides and oligosaccharides have no protective effect on acute alcoholic liver injury. The relevant research results provide a useful reference for the further research and development of Cistanche deserticola.

To protect the liver with cistanche para que sirve
References
[1] Zhang YW, Li YJ, Hu BF, et al. Evaluation on three short-term animal models of alcoholic liver disease [J]. Acta Pharm Sin, 2018, 53: 236-243.
[2] Vonghia L, Leggio L, Ferrulli A, et al. Acute alcohol intoxication [J]. Eur J Intern Med, 2008, 19: 561-567.
[3] Hou J, Lu Y, Zhang DK. Association between intestinal dysbacteriosis and liver diseases [J]. J Clin Hepatol, 2018, 34: 1128- 1132.
[4] Liu Y, Wang H, Yang M, et al. Cistanche deserticola polysaccharides protect PC12 cells against OGD/RP-induced injury [J]. Biomed Pharmacother, 2018, 99: 671-680.
[5] Song DZ, Cao Z, Liu ZB, et al. Cistanche deserticola polysaccharide attenuates osteoclastogenesis and bone resorption via inhibiting RANKL signaling and reactive oxygen species production [J]. J Cell Physiol, 2018, 233: 9674-9684.
[6] Zhang AL, Yang XM, Li QX, et al. Immunostimulatory activity of water-extractable polysaccharides from Cistanche deserticola as a plant adjuvant in vitro and in vivo [J]. PLoS One, 2018, 13: e0191356.
[7] Zhang H, Xiang Z, Duan X, et al. Antitumor and anti-inflammatory effects of oligosaccharides from Cistanche deserticola extract on spinal cord injury [J]. Int J Biol Macromol, 2019, 124: 360-367.
[8] Li RY, Zhao MB, Tu PF, et al. Simultaneous determination of five phenylethanoid glycosides in Cistanches Herba using quantitative analysis of multi-components by single marker [J]. J Chin Pharm Sci, 2019, 28: 537-546.
[9] Li SQ, Lu HJ, Wang P, et al. Study on the time of cell apoptosis in alcoholic liver injury in mice [J]. Chin J Clin Pharmacol, 2017, 33: 2154-2157.
[10] Shi ZY, Wu Y, Zhu YM, et al. Quantitative determination of betaine, mannitol, fructose, glucose and sucrose in Cistanches 2019, 21: 1641- 1646.
[11] Gao YJ, Jiang Y, Dai F, et al. Study on laxative constituents in Cistanche deserticola Y. C. Ma [J]. Mod Chin Med, 2015, 17: 307-310.
[12] You SP, Zhao J, Ma L, et al. Effect and mechanism of Cistanche phenylethanoid glycosides on rats with immunological liver fibrosis [J]. Chin J Pharmacol Toxicol, 2016, 30: 504-510.
[13] Luo HY, Liu Y, Xi GZ, et al. Protective effect of Cistanchis glycosides on ethanol-induced liver damage in mice [J]. Chin J Clin Pharmacol Ther, 2009, 14:1225- 1228.
[14] Wu KC, Liu J, Klaassen CD. Role of Nrf2 in preventing ethanol-induced oxidative stress and lipid accumulation [J]. Toxicol Appl Pharmacol, 2012, 262: 321-329.
[15] Qu Q, Liu J, Zhou HH, et al. Nrf2 protects against furosemide-induced hepatotoxicity [J]. Toxicology, 2014, 324: 35-42.
[16] Zhang L, Zu XP, Xie HS, et al. Research progress in mechanism of intestinal microorganisms in human diseases [J]. Acta Pharm Sin, 2016, 51: 843-852.
[17] Parlesak A, Schäfer C, Schütz T, et al. Increased intestinal permeability to macromolecules and endotoxemia in patients with chronic alcohol abuse in different stages of alcohol-induced liver disease [J]. J Hepatol, 2000, 32: 742-747.






