Rewiring Of Glucose And Lipid Metabolism Induced By G Protein-Coupled Receptor 17 Silencing Enables The Transition Of Oligodendrocyte Progenitors To Myelinating Cells Ⅱ
Jul 13, 2023
3. Results
3.1. GPR17 Silencing in Differentiating OPCs Signifificantly Altered the Expression of Genes Involved in Lipid and Glucose Metabolism
To highlight the biological processes signifificantly affected by GPR17 expression during OL differentiation, we interfered with its expression by transfecting rat OPCs with SMART-pool siRNAs specifific for rat GPR17. After having validated successful GPR17 knockdown by qRT-PCR (Figure S1), we performed a microarray analysis of 30,584 rat transcripts in the GPR17-silenced versus control OPCs and analyzed the differentially expressed genes (DEGs) by different bioinformatic tools. First, the 640 DEGs have been studied by Meta-Core, to perform a pathway enrichment analysis and identify the processes predicted to be signifificantly affected. This analysis suggested alteration of the mTOR signaling, which has been already shown to be linked to GPR17 function [37], and of other processes known to be relevant for OPC maturation, such as “cytoskeleton remodeling” and “regulation of lipid metabolism” (Table 1).
 +
+
Click Here To Know Cistanche For Bone Growth
Table 1. The software MetaCore has been used to perform a pathway enrichment analysis on the DEGs after GPR17 silencing. The table shows the most signifificant pathways resulting from the analysis, the number of associated genes, and of common genes included in the dataset
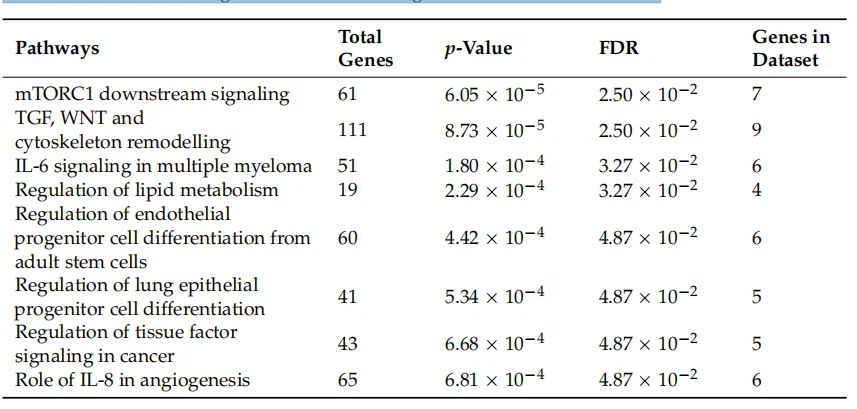
Then, the Ingenuity pathway analysis (IPA) tool was used to perform an analysis match, which automatically identifies curated IPA datasets with signifificant similarities and differences compared to a query dataset. This analysis strengthens the predicted link between GPR17 expression and lipid metabolism, showing that the expression changes of 38 genes in our dataset (Table S1) are associated with altered fatty acid synthesis (Figure 1). Among these, GPR17 silencing induced upregulation of LXRα (liver X receptor alpha, NR1H3 in Figure 2 and Table S1, a nuclear receptor responding to oxysterols) and SREBP1 (sterol regulatory element binding protein 1, SREBF1 in Figure 1 and Table S1), two key players in fatty acid and cholesterol synthesis. In our dataset, we also observed that several altered genes, including PDH (pyruvate dehydrogenase), Ldha (lactate dehydrogenase alpha), and Pdk1 (pyruvate dehydrogenase kinase 1), are related to glucose metabolism and Krebs’ cycle, suggesting the potential involvement of the GPR17 receptor in the regulation of these metabolic processes. According to this, a gene ontology-based enrichment analysis (ToppGene suite) on the DEGs revealed alteration of the “monocarboxylic acid metabolic process” (GO:0032787, p-value: 0.02), beyond other processes related to CNS development and cell metabolism, such as “regulation of nervous system development” (GO:0051960, p-value: 0.033) and “sphingolipid metabolic processes” (GO:0006665, p-value: 0.043) (Figure 2; full list in Table S2).
Globally, these changes suggest that GPR17 silencing may act as a trigger to address cells to myelination, by tuning several genes involved in the transcriptional regulation of lipid and glucose homeostasis, as well as in utilizing cholesterol and lipids for myelin production.
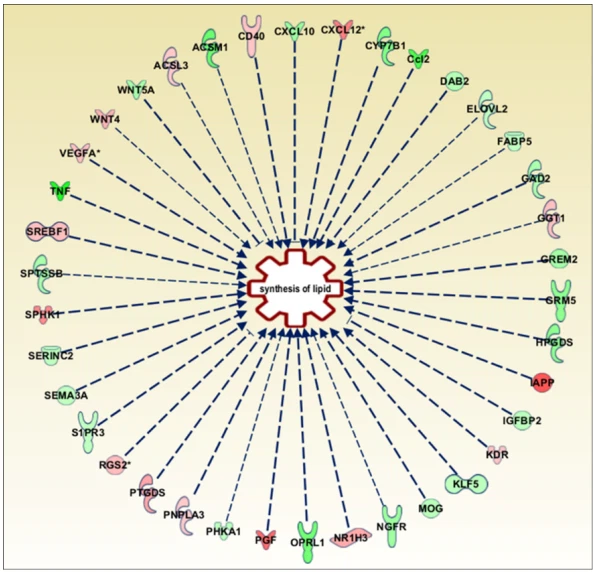
Figure 1. Ingenuity pathway analysis tool (IPA, Qiagen) was used to analyze the dataset and explore the diseases and biological processes which are predicted to be increasing or decreasing based on the pattern of differentially expressed genes in the dataset. From this analysis, we extrapolated the scheme in the Figure that includes the genes related to lipid synthesis differentially expressed in our dataset (p-value = 9.67 × 10−4 ). The full names of these genes, together with the relative fold change (log2 ratio) are reported in Table S1. In red, upregulated genes; in green, downregulated genes

Figure 2. The Toppgene suite has been used for GO-based enrichment analysis on the DEGs. The picture shows some of the signifificant biological processes related to the central nervous system and cell metabolism potentially altered by GPR17 silencing. Blue bars indicate the total genes associated to each term; red bars indicate the genes in common with our dataset for each term. The complete list of biological processes resulting from this analysis and the relative p-values are reported in Table S2.

3.2. Metabolomic Analysis during OPC Differentiation In Vitro
To assess the correlation between GPR17 expression and the activation of specifific metabolic pathways, we performed a metabolomic analysis of cultured OPCs during their physiological maturation. OPCs were maintained in a differentiation medium and then lysed at four different time points (after 0, 1, 3, and 5 days in differentiation, DID), that correspond to varying stages of OL maturation. In parallel, to assess that in all the experiments maturation occurred with the expected timing, OPCs were cultured and stained for GPR17 and MBP. The results showed that the number of GPR17-expressing cells reached its maximum at 3 DID and then returned towards basal levels at 5 DID, whereas the number of MBP-positive cells progressively increased during maturation (Figure 3a), consistently with previous results [37]. OPC metabolomic evaluation was performed by liquid chromatography-tandem mass spectrometry (LC-MS/MS) as previously described [38], focusing on energetic metabolites involved in glycolysis, the pentose phosphate pathway, the Krebs cycle, fatty acid β-oxidation, their cofactors NADH, NAD+, NADPH, NADP+ , ATP, ADP, AMP, amino acids and some of their derivatives. The metabolomic profiles of the four-time points analyzed are reported in Figure 3b. At day 0, the levels of several metabolites belonging and/or linked to the Krebs cycle (e.g., succinate, malate, alpha-Ketoglutarate, glutamate, aspartate, alanine, and proline) were higher, as expected from cells coming from a highly proliferating state (Figure 3c). At intermediate stages (from DID 1 to 3), OPCs showed a marked upregulation of molecules involved in fatty acid and cholesterol synthesis. Accordingly, we observed increased levels of acetyl-CoA (Figure 3c), which represents the precursor of cholesterol and fatty acids and of malonyl-CoA, a specifific intermediate of fatty acid synthesis. At DID 5, we observed increased levels of several acyl-carnitines and amino acids in parallel to a signifificant increase of free intracellular glucose (Figure 3c). We also observed decreased levels of AMP and ADP from day 0 to DID 5 and a transient increase of ATP between DID 1 and DID 3. These data indicate that, from day 0 to DID 3, corresponding to the temporal window during which GPR17 reaches its maximum expression, OPCs mainly used glucose and amino acids to sustain cholesterol and fatty acid biosynthesis. At DID 5, as highlighted by increased acyl-carnitine levels, OPCs likely rely on fatty acid utilization. Together, these data indicate that during physiological differentiation OPCs progressively rewire their energetic metabolism to become mature myelinating OLs.

3.3. Metabolomic Analysis after GPR17 Silencing during OPC Maturation In Vitro
To unveil the contribution of the GPR17 receptor in driving the energy metabolic changes occurring during cell differentiation, we performed metabolomics on GPR17- silenced OPCs, in comparison to control OPCs receiving a scrambled RNA. We first examined the impact of GPR17 silencing on OPC maturation by immunofluorescence staining for GPR17 and MBP. As expected, after receptor silencing, we found a reduction in GPR17 staining, clearly visible at 3 DID. We also observed a marked increase in the number of mature MBP-positive OLs at both 3 DID (not shown) and 5 DID (Figure S2). In parallel, detailed qPCR analysis was performed at five-time points (0, 1, 2, 3 and 5 days in differentiation, DID), revealing, in control OPCs, a strong increase in GPR17 expression after 1 DID, that reaches signifificant levels starting after 2 DID, a progressive increase in MBP expression up to 5 DID and a signifificant decrease in the early marker NG2 (Figure 4a). Conversely, after GPR17 silencing (Figure 4b), GPR17 mRNA reached lower levels at DID1 and then remained stably low, MBP expression further increased and NG2 further decreased. A direct comparison of NG2 and MBP expression in GPR17-silenced versus control OPCs at each time point is shown in Figure 4c. Taken together, these results suggest that, under these experimental conditions, GPR17 silencing in OPCs accelerated their differentiation process. Then, OPC metabolomics was performed by LC-MS/MS at the five selected time points. Comparison of metabolomic data obtained from GPR17-silenced and control OPCs at each time point allowed us to identify the metabolites that are signifificantly affected by GPR17 silencing
Figure 3. (a) The graph shows changes in the percentage of GPR17+ and MBP+ cells during differentiation. n = 3 per group; one-way ANOVA with Tukey’s test. * p < 0.05, ** p < 0.01) (b) The heatmap shows metabolite abundance in each single sample stopped at 0, 1, 3 or 5 DID. The abundance of the analyzed metabolites is represented by a chromatic scale ranging from dark blue (very low abundance) to dark brown (very high abundance). n = 4–5 per group. (c) Metabolites with an abundance signifificantly different among the conditions (0, 1, 3 and 5 days in differentiation, DID; one-way ANOVA with Fisher’s LSD test).
At DID 1 (Figure 5a), we did not find significant changes. At DID 2 (Figure 5b), we found a significant increase in some metabolites of the Krebs cycle (malate, fumarate citrate), a modest increase in free carnitine (CO), acetyl-carnitine(C2), propionyl-carnitine(C3:0), butyryl-carnitine (C4:0), valeryl-carnitine (C5:0), hexadecanoyl-carnitine (C16:0and a significant reduction in glucose, lactate, creatine, and ornithine. The reduction oilactate and creatine and the increase of propionyl-carnitine (C3) was also observed at DID3 (Figure 5c). At DID 5 (Figure 5d), we still found an increase in free carnitine (CO), acetylcarnitine (C2), propionyl-carnitine (C3:0), butyryl-carnitine (C4:0), valeryl-carnitine (C5:0)tetradecanoyl-carnitine (C14:0), hexadecanoyl-carnitine (C16:0), octadecanoyl-carnitineC18:0), octadecenoyl-carnitine (C18:1), similarly to what originally observed in more differentiated cells (see Figure 3), an increase in dihydroxyacetone-3-phosphate/ glyceraldehyde3-phosphate (DHAP/GAP), fumarate and citrate, and a decrease in acetyl-CoA, Met-SOcreatine, taurine and carnosine.
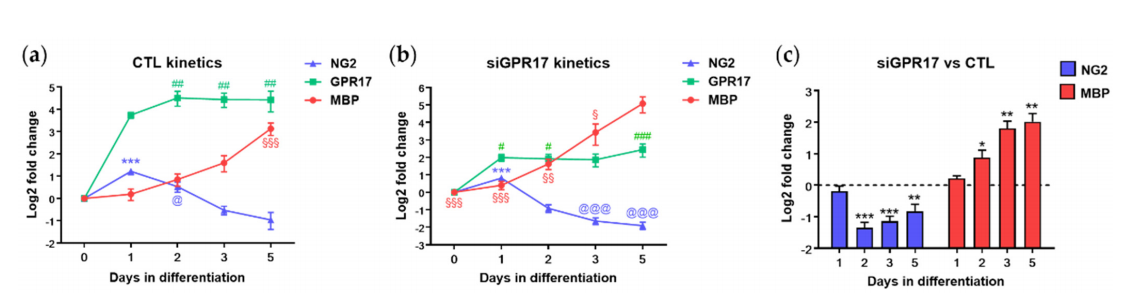
Figure 4. Effect of GPR17 silencing on OPC maturation. The expression levels of GPR17 (green), NG2blue) and MBP (red) were evaluated by real-time PCR during differentiation in control conditions (cells transfected with scramble RNA and after GPR17 silencing (b), and normalized versus expression at T0 (set to 0). n = 6 per each time point. One-way ANOVA with Tukey's multiple comparisons tests (a): ** p < 0.001 VS. DID 0-2-3-5, @ p < 0.05 Vs. DID 3-5; ## p < 0.01 vs. day 0; SSS p < 0.001 VS. DID0-1-2-3; (b): *** p < 0.001 Vs. DID 0-2-3-5, @@@ p < 0.001 vs. day 0, # p < 0.05, ### p < 0.001 vs. day 0.S p < 0.05, SS p < 0.01, SSS p < 0.001 V. DID 5. (C) NG2 and MBP expression in GPR17-silenced OPCsvs. control OPCs (set to 0) at each time point. Fold change (FC) has been reported as LOG,(FC). One sample t-test, * p < 0.05, * p < 0.01; *** p < 0.001 vs. relative DID in control OPCs.
Figure 5. Metabolomic changes induced by GPR17 silencing in OPCs. (a–d) The abundance of each metabolite after GPR17 silencing has been normalized to that in the relative control sample. The resulting fold changes were used to generate a volcano plot for each time point (1, 2, 3, 5 days in differentiation). The points over the dotted horizontal line represent the metabolites that have shown a statistically signifificant change (Fisher’s LSD corrected by FDR; p.adj < 0.1). In green, metabolites with lower expression, in red, those with higher expression, after GPR17 silencing. n = 7–9 per each group. DID: days in differentiation.
3.4. GPR17 Modulates Lactate Release during OPC Maturation
Based on the metabolomics results described above, GPR17 silencing was found to reduce the intracellular levels of lactate. To assess if this reduction reflects a general decrease in lactate metabolism or an increased release of this metabolite, we measured its levels in cell lysates and in the corresponding media at different time points. As shown in Figure 6, in control conditions, intracellular lactate reached the highest abundance at DID 2 (Figure 6a) (when GPR17 receptor expression is also maximal), and then it reduced when the cells became mature. Instead, after GPR17 silencing, the rise of intracellular lactate was abolished, and its levels further decreased toward differentiation. Indeed, a comparison of lactate levels in GPR17-silenced OPCs versus control OPCs at each time point highlighted a reduction in its levels at DID 2 and 3 (Figure 6b). The extracellular lactate followed different kinetics, showing a signifificant reduction compared to DID 1 at DID 2-3-5 in control conditions, whereas in GPR17-silenced OPCs it remained stably higher and then signifificantly decreased at DID 5 (Figure 6c). However, when comparing lactate levels at the different time points between the two experimental conditions, GPR17 silencing hampered the physiological reduction, increasing lactate presence in the medium at DID 2 and 3 (Figure 6d), suggesting an increased release.
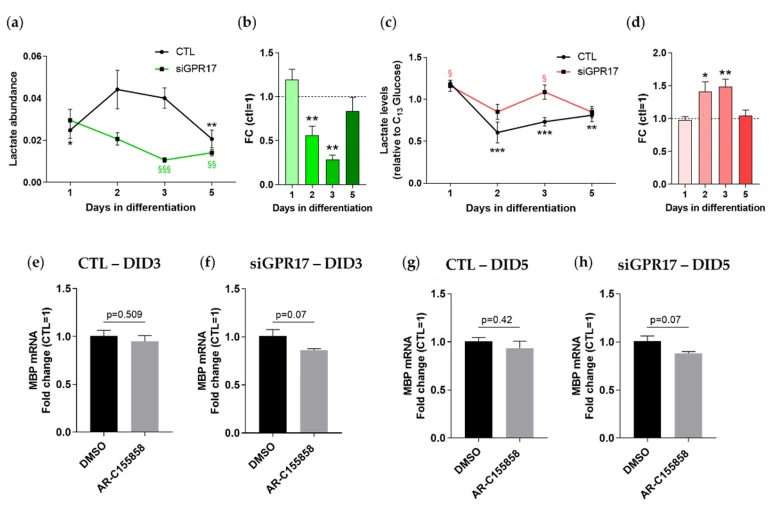
Figure 6. GPR17 silencing in OPCs alters lactate metabolism. (a) Lactate abundance during OPC maturation in control conditions (green line) and after GPR17 silencing (orange line). n = 7–9 per group. One-way ANOVA with Tukey’s multiple comparisons test. * p < 0.05, ** p < 0.01 vs. DID2 CTL; §§ p < 0.01, §§§ p < 0.001 vs. DID1 siGPR17 (b) Lactate levels after GPR17 silencing were normalized versus control samples (set to 1) at each time point (n = 7–9). ** p < 0.01; one sample t-test. Conditioned media from GPR17-silenced and control OPCs were collected from the same culture used for metabolomic analysis. Lactate levels in the media were evaluated by LC-MS/MS. (c) The abundance of lactate in the extracellular space during OPC maturation in control conditions (green line) and after GPR17 silencing (orange line). One-way ANOVA with Tukey’s multiple comparisons test. ** p < 0.01, *** p < 0.001 vs. DID1 CTL; § p < 0.05 vs. DID5 siGPR17. (d) Extracellular lactate levels after GPR17 silencing were normalized versus control samples (set to 1) at each time point. * p < 0.05; ** p < 0.01; one sample t-test. The effect of MCT1 inhibition in control (e,g) and GPR17-silenced (f,h) OPCs has been evaluated by analyzing the expression of MBP, in inhibitor-treated samples vs. relative controls (vehicle-treated), at at 3 and 5 days of differentiation. n = 6 per group.

One of the mechanisms by which OPCs can release lactate is through the monocarboxylate transporter MCT1. Moreover, recent studies reported that OPCs themselves can acquire extracellular lactate through MCT receptors promoting their cell cycling and differentiation [39]. Thus, we wondered whether the increased release of lactate induced by GPR17 silencing could be mediated by MCT1 and whether this may be partially responsible for the effect observed on OPC maturation in the same conditions (Figure 4). To this aim, we treated OPCs with AR-C155858, a selective inhibitor of MCT1, during normal differentiation and after GPR17 silencing. As expected, lactate release was successfully inhibited in both conditions (Figure S3). The effect of MCT1 inhibition on OPC maturation in the different experimental conditions was evaluated by analyzing the expression levels of MBP. A modest reduction in the gene expression of MBP, which is close to statistical significance (p = 0.07), has been found only in samples transfected with specifific siRNA for GPR17 (Figure 6e–h); however, no signifificant variation emerged in the samples transfected with control RNA (siNEG). These results suggest that the increase in extracellular lactate mediated by GPR17 downregulation fosters the maturation of OPCs; however, the small extent of MBP reduction indicates that this metabolite is not the only factor involved in the process.
3.5. Lipidomic Analysis after GPR17 Silencing during OPC Maturation
The results of the transcriptomic analysis after GPR17 silencing also highlighted a potential link between its function and the synthetic pathways of lipids and cholesterol, which are the main components of myelin. To further investigate the role of GPR17 in lipid metabolism, we applied LC-MS/MS to the lipidomic analysis of GPR17-silenced OPCs after 2, 3 and 5 days in differentiation, compared to day 0. The list of the analyzed lipids, relative fold changes and p-values are reported in Table S4. The two most abundant lipid classes in the experimental groups were fatty acids and the diacyl-phosphatidylcholine (PCaa) family. During the differentiation of control OPCs, we did not find any signifificant changes in the abundance of any lipid class. Instead, GPR17-silenced OPCs showed a trend toward an altered abundance of free fatty acids, ceramides, acyl-alkyl-phosphatidylcholine (PCae), and phosphatidylinositol (PI) compared to control OPCs, especially at DID 3 and 5, but only diacyl-phosphatidylethanolamine (PEaa) and sphingomyelin (SM) showed a statistically signifificant change (Figure 7).

Figure 7. Changes in lipidic classes abundance induced by GPR17 silencing during OPC differentiation. The abundance of each lipid class in control conditions at different time points is reported (a)The kinetics of each lipid class after GPR17 silencing were analyzed: (b) fatty acids, (C) ceramides(d) LysoPC (lysophosphatidyl-cholines), (e) LysoPE (lysophosphatidylethanolamines), (f) PCaa(diacyl-phosphatidyl-cholines), (g) PCae (acyl-alkyl-phosphatidyl-cholines), (h) PEaa (di-acylphosphatidyl-ethanolamines), (i) PEae (acyl-alkyl-phosphatidyl-ethanolamines), (i) PI (phosphatidylinositols), (k) PS (phosphatidyl-serines), (D SM (sphingomyelins). n = 5 for each group. Kinetics: Two-way ANOVA followed by Tukey's multiple comparison test. siGPR17: Two-way ANOVA followed by Sidak's multiple comparison test. * p.adi < 0.05, ** p.adi < 0.01.

Comparison of lipidomic data obtained from GPR17-silenced and controls at each time point allowed us to identify the lipids that were significantly affected by GPR17 silencing. After 2 DID, no significant changes were found (data not shown). After 3 DID, diacylphosphatidylcholine (PCaa) C40:4 was less abundant and diacyl-phosphatidylethanolaminePEaa) C34:3 was more abundant in GPR17-silenced OPCs (Figure 8a). After 5 days of differentiation, GPR17 silencing led to several changes in lipid abundance: reduction of several diacyl-PCs and diacyl-PE and a concomitant increase in several plasmalogens (acyl alkyl-PC) along with sphingomyelin (SM) C18:1 and SM(OH) C16:1 (Figure 8b). These data indicate that the downregulation of GPR17 alters the lipid profile of the main component of myelin as PC and PE.
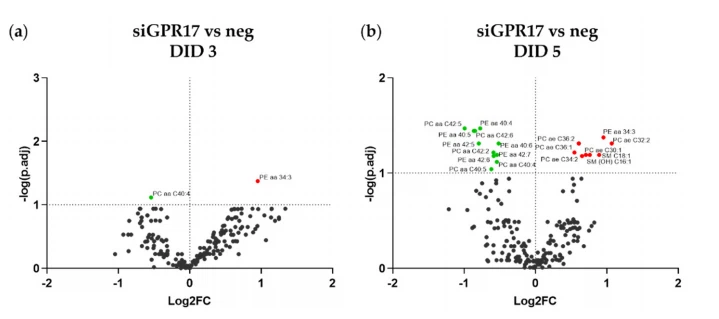
Figure 8. Lipidomic changes induced by GPR17 silencing in OPCs. (a,b) The abundance of each lipid after GPR17 silencing has been normalized to that in the relative control sample. The resulting fold changes were used to generate a volcano plot for each time point (3, 5 days in differentiation). The points over the dotted horizontal line represent the metabolites that have shown a statistically significant change (Fisher’s LSD corrected by FDR; p.adj < 0.1). In green, lipids with lower expression, in red, those with higher expression, after GPR17 silencing. n = 5 per group.
Ask for more:
Email:wallence.suen@wecistanche.com
Whatsapp/Tel:+86 15292862950
SHOP:
https://www.xjcistanche.com/cistanche-shop






