Taurine: A Source And Application For The Relief Of Visual Fatigue Part 1
Sep 26, 2023
Abstract: According to reports, supplementation with appropriate doses of taurine may help to reduce visual fatigue. Presently, some progress has been made in research related to taurine in eye health, but the lack of systematic summaries has led to the neglect of its application in the relief of visual fatigue. This paper, therefore, provides a systematic review of the sources of taurine, including the endogenous metabolic and exogenous dietary pathways, as well as a detailed review of the distribution and production of exogenous taurine. The physiological mechanisms underlying the production of visual fatigue are summarized and the research progress of taurine in relieving visual fatigue is reviewed, including the safety of consumption and the mechanism of action in relieving visual fatigue, to provide some reference basis and inspiration for the development and application of taurine in functional foods for relieving visual fatigue.
Cistanche can act as an anti-fatigue and stamina enhancer, and experimental studies have shown that the decoction of Cistanche tubulosa could effectively protect the liver hepatocytes and endothelial cells damaged in weight-bearing swimming mice, upregulate the expression of NOS3, and promote hepatic glycogen synthesis, thus exerting anti-fatigue efficacy. Phenylethanoid glycoside-rich Cistanche tubulosa extract could significantly reduce the serum creatine kinase, lactate dehydrogenase, and lactate levels, and increase the hemoglobin (HB) and glucose levels in ICR mice, and this could play an anti-fatigue role by decreasing the muscle damage and delaying the lactic acid enrichment for energy storage in mice. Compound Cistanche Tubulosa Tablets significantly prolonged the weight-bearing swimming time, increased the hepatic glycogen reserve, and decreased the serum urea level after exercise in mice, showing its anti-fatigue effect. The decoction of Cistanchis can improve endurance and accelerate the elimination of fatigue in exercising mice, and can also reduce the elevation of serum creatine kinase after load exercise and keep the ultrastructure of skeletal muscle of mice normal after exercise, which indicates that it has the effects of enhancing physical strength and anti-fatigue. Cistanchis also significantly prolonged the survival time of nitrite-poisoned mice and enhanced the tolerance against hypoxia and fatigue.

Click on muscle fatigue
【For more info:george.deng@wecistanche.com / WhatsApp:8613632399501】
Keywords: taurine; visual fatigue; source; safety; relief of visual fatigue
1. Introduction
When the eye is overused, or when the environment is too dry or disturbed by other physiological (and possibly pathological) factors, it is easy for the eye to adjust the refraction for a long time to form normal visual feedback, thus triggering visual fatigue, which is mainly manifested by frequent blurred vision, eye soreness, dryness, tearing and other eye discomfort [1]. A study in 2021 showed that the prevalence of visual fatigue among university students around the world is 46~71% [2]. Visual fatigue not only affects work and study status but can also lead to video terminal syndrome and dry eye syndrome if one remains in a chronic state of visual fatigue [3]. At the same time, patients with eye diseases, such as strabismus and dry eye, are more likely to suffer from visual fatigue. It has been reported that 71.3% of people with dry eye suffer from varying degrees of visual fatigue [4]. At present, the incidence of visual fatigue is high and the number of people affected is increasing every year [5]. These people are of all ages and are, especially, students, teachers, and those who work with electronic devices such as computers [6–8]. This shows that visual fatigue occurs in all age groups and that the number of people who need relief from visual fatigue is very large. Studies have shown that the physiological mechanisms of visual fatigue are mainly due to various stress injuries to the retina or macula and damage to nerve cells.
1.1. Retinal Stress Damage: Oxidation, Inflammation, Apoptosis
The retina, located in the fundus of the eye, is a fragile neuronal tissue that perceives and receives external light to form visual signals that are transmitted to the brain to obtain external information [9]. The retina is composed of cells such as the pigment epithelium (RPE), optic cells, bipolar cells, and ganglion cells, and studies have shown that reduced retinal function is one of the major causes of the development of visual fatigue [10]. In particular, impairment of the structural integrity and function of the RPE will directly affect retinal function [11]. RPE, as one of the most metabolically active tissues within the body, has a high oxygen demand and is, therefore, susceptible to the production of reactive oxygen species (ROS) and their attack. When the eye is exposed to prolonged work, intense or dense light, and high oxygen pressure, the metabolic rate of the extraocular and ciliary muscles increases, contributing to the accumulation of large amounts of metabolites such as peroxides and ROS. When there is an imbalance between the production of free radicals and endogenous antioxidant defense mechanisms, this results in oxidative stress damage to the eye [12], leading to the development of visual fatigue. At the same time, there are large amounts of polyunsaturated fatty acids (PUFA) in the retina, particularly docosahexaenoic acid, which is essential for the integrity of photoreceptor cells and is involved in the phototransduction cascade [13]. ROS tend to oxidize these PUFA, producing large amounts of lipid peroxides and further inducing the production of large amounts of reactive aldehydes, such as 4-hydroxynonenoic acid and Malondialdehyde (MDA), which, when bound to intracellular proteins and DNA, can cause cellular inflammation and apoptosis [14], thereby inducing visual fatigue.
The aging of RPE will lead to the aging of the eyes, the reduction of macular pigment optical density (MPOD), and the impairment of macular function, affecting normal visual function. Furthermore, excessive accumulation of N-retinylidene-N-retinylethanolamine (A2E) on the RPE predisposes cells to oxidative stress or cell death, a condition that can also contribute to the development of visual fatigue [15]. Li et al. [16] found that the intervention of a 28 g wolfberry diet 5 times per week for 90 days in healthy people can significantly increase the retinal eccentrics of MPOD at 0.25 and 1.75, indicating that regular intake of wolfberry in healthy middle-aged people can increase MPOD, which is helpful to prevent or delay macular injury. This effectively protects the retina from the effects of aging. From another perspective, it also suggests that dietary interventions exerting retinal protection strategies are feasible.
1.2. Damage to Nerve Cells
The retina is rich in various nerves and retinal glial cells including microglia, astrocytes, and retinal Müller cells. Among them, the retinal macroglia (Müller cells and astrocytes) and microglia are very important in maintaining retinal homeostasis and protecting the retina from damage [17,18]. Studies have shown that retinal macroglia is the basis for maintaining retinal neuronal homeostasis [19]. Müller cells, which have the highest content—up to 90%—are the main structural and nutritional support for retinal cells [20]. Astrocytes, on the other hand, play a role in protecting the retina from damage, mainly through the reactive glial proliferation pathway [21]. However, increased levels of some proteins expressed by reactive astrocytes, such as Matrix metalloproteinase-9 (MMP-9) and Urokinase-type Plasminogen Activator (uPA), can cause retinal damage [22], so the role of astrocytes in the retina is dual. In contrast, retinal microglia play a deleterious role in cell death [23]. When microglia are activated, this leads to an accelerated release of pro-inflammatory and pro-phagocytic factors, resulting in cell death and more microglia activation, which in turn contributes to inflammatory damage to the retina and photoreceptor damage [24]. At the same time, retinal glial cells are also involved in glutamate buffering in neurons. Glutamate is thought to be the main chemical signal in photoreceptors, bipolar cells, and ganglion cells in the retina [25,26]. However, when glutamate concentrations are too high, it can cause neuronal excitotoxicity, with the result that neurons are damaged or even die [27]. When retinal Müller cells are damaged, the excitotoxicity of glutamate may be further exacerbated. It has been reported that the inflammatory damage condition of the retina, as well as the damaged photoreceptor condition, can be effectively protected by inhibiting microglia activity [28]. Thus, the regulation of glutamate homeostasis by retinal glial cells may exert an important protective mechanism against neuronal damage [29].

Taurine, chemically known as 2-aminoethanesulfonic acid, is a specific non-protein amino acid that is present in high concentrations in the brain, retina, and muscle in its free state [30]. Data show that the levels of taurine in the human brain, retina, and plasma are 1~20 µmol/g, 30~40 µmol/g, and 50~100 µmol/L, respectively [31]. Taurine is mainly involved in physiological activities, including bile acid binding, osmoregulation, neuronal excitability, inflammatory response, and glucose metabolism [32]. For eye health, taurine plays an important role in promoting retinal differentiation and development [33], while lack of taurine in diet can lead to impaired retinal function and even blindness [34]. It has been reported in the literature that taurine depletion and exposure to light causes shortening of the outer segments of photoreceptor cells, activation of microglia, oxidative stress in the outer and inner nuclear layers and ganglion cell layer, loss of synapses [35] and loss of optic cone cells [36]. This suggests that taurine depletion can cause damage to cells such as retinal pigment epithelial cells, photoreceptors, and retinal ganglion cells in the retina, contributing to increased glial cell responses and oxidative stress. It is also suggested that taurine is essential for ocular health and that dietary supplementation of taurine can effectively improve ocular stress damage and exert good neuroprotective effects, thereby repairing damaged retinas and reducing visual fatigue [37]. At present, some progress has been made in research related to taurine in eye health, but the lack of systematic summary has led to the neglect of its application in the alleviation of visual fatigue. This paper, therefore, provides a systematic review of the sources of taurine, including the endogenous metabolic and exogenous dietary pathways, as well as a detailed review of the distribution and production of exogenous taurine. The physiological mechanisms underlying the production of visual fatigue are summarized and the research progress of taurine in relieving visual fatigue is reviewed, including the safety of consumption and the mechanism of action in relieving visual fatigue, to provide some reference basis and inspiration for the development and application of taurine in functional foods for relieving visual fatigue.
2. Sources of Taurine
Currently, taurine is obtained from two sources: the endogenous metabolic synthetic pathway of the body and the exogenous dietary route.
2.1. Endogenous Metabolic Synthesis of Taurine
Taurine is mainly found in the liver, central nervous system, retina, heart, skeletal muscle, and other excitable tissues [38,39]. In mammals, taurine is synthesized from methionine or cysteine in the presence of adequate levels of vitamin B6 [40]. The pathway of taurine synthesis in the liver is detailed in Figure 1. In the endogenous synthesis of taurine, the decarboxylation reaction is the rate-limiting step, and cysteine sulphinic acid decarboxylase (CSAD) and cysteine dioxygenase (CDO) are rate-limiting enzymes. Studies have shown that CSAD levels vary considerably between organisms, with higher CSAD activity in rodents and lower in humans, which seems to explain why cats prefer to prey on rats. Therefore, the normal requirements of the body cannot be met by the body's synthetic taurine content alone and a dietary supplement of exogenous taurine is required [41].
2.2. Exogenous Distribution and Mode of Production of Taurine
2.2.1. Sources and Distribution
It is reported that the main dietary sources of taurine are animal foods, especially marine organisms, and mammals, which contain high concentrations of taurine in their organs and tissues [42]. In addition, seaweeds such as kelp and spirulina and a few plants such as Ganoderma lucidum spores, wolfberries, and cordyceps also contain some taurine [43–46]. The main dietary sources of taurine and the proportion of each food are detailed in Figure 2.

2.2.2. Mode of Production of Exogenous Taurine
Currently, exogenous taurine is produced by natural extraction, chemical synthesis, and bio-fermentation and each production method has certain characteristics, as detailed in Table 1.
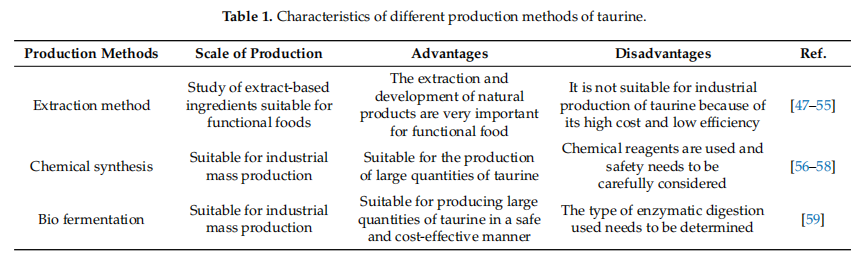
Extraction Methods
Taurine is soluble in water, but insoluble in ethanol, ether, and acetone. Boiling is a simple and inexpensive method of extraction. However, the long extraction time and low extraction efficiency of the boiling method have been observed in the early years of extraction research [47]. Currently, the boiling method is mostly supplemented by other extraction methods to compensate for the shortcomings of a single extraction, e.g., ultrasound-assisted boiling extraction [48]. Ultrasound-assisted extraction is a new nonthermal physical processing technology that uses ultrasound cavitation to disrupt cell membranes (walls), to accelerate and enhance the dissolution of active ingredients in natural products and, therefore, induce a certain solubilizing effect [49]. Compared to conventional extraction methods, ultrasonic extraction has the advantages of suitability for all kinds of solvents, fast extraction speed, simple method, high yield, and reduced solvent consumption, and it is particularly suitable for the extraction of heat-unstable components [50,51]. However, ultrasonic extraction techniques are difficult and costly to prepare for industrial implementation. Therefore, it is mainly used for laboratory studies.
Solvent extraction is also an extraction method for taurine from natural products. A solution of 60–75% ethanol is mostly used as a solvent for extraction in existing studies, but the extraction rate of taurine is not greatly improved compared to boiling [52–54] and ethanol as an extraction solvent for industrial production is not only costly but also poses certain safety risks.

Enzymatic digestion can also be used for the extraction of taurine and can be combined with boiling. Enzymatic digestion has the advantages of safe operation, high extraction efficiency, and low cost, but the experimental conditions are strict and the appropriate enzyme type needs to be determined according to the target. Enzymes have a high degree of specificity, the source of raw materials is different and the corresponding enzyme hydrolysis types need to be investigated. Taurine is mostly sourced from animal foods and the use of direct hydrolysis by biological enzymes can result in the dissolution of free hydrogenated acids, peptides, and trace elements from animal raw materials in the enzymatic digest, leading to increased difficulty in separation [55]. In addition to boiling, solvent extraction, enzymatic digestion, and ultrasound-assisted methods, there are also ultra-high pressure treatment methods [60] or a combination of extraction methods to extract taurine. In general, the extraction efficiency and effectiveness of taurine from natural products are poor if a single extraction method is used. Therefore, a combination of extraction methods has been used to improve the extraction efficiency and yield. Additionally, the cost of obtaining taurine from natural extraction is high and most of these methods are not suitable for industrial production.
The yield of taurine prepared by different extraction methods is detailed in Table 2.

Chemical Synthesis and Bio-Fermentation Methods
More than 20 methods have been developed for the chemical synthetic production of taurine. Of these, most are produced commercially by ethylene oxide or monoethanolamine [66]. However, ethylene oxide is more toxic, has higher reagent recovery costs, and is also volatile and explosive. The production of taurine by this method is therefore very demanding and costly [56]. The industrial production of taurine from monoethanolamine has better safety and environmental benefits than the ethylene oxide process; it is a two-step process that involves the synthesis of 2-aminoethyl sulfate from monoethanolamine and sulphuric acid by heat treatment and the production of taurine from 2-aminoethyl sulfate by heat treatment with an acidic solution of sodium sulfite [57]. However, the production of taurine from monoethanolamine takes too long and consumes too much energy. Moreover, some intermediate products are susceptible to thermal hydrolysis, which can lead to an increase in by-products and affect the yield of taurine [58]. In conclusion, each method has its advantages and disadvantages and each step of the reaction must be strictly controlled during the industrial production process.
In addition, bio-fermentation can also be used to produce taurine. At present, the following microorganisms have been reported for the production of taurine by bio-fermentation: Yarrowia lipolytica, Chlamydomonas reinhardtii, Saccharomyces cerevisiae, Corynebacterium glutamicum, etc. [59]. Among them, Corynebacterium glutamicum is considered to be a safe and suitable strain for taurine production. By constructing a recombinant strain of Corynebacterium glutamicum, 62.0 ± 1.5 mg/g (cell dry weight) of taurine can be obtained with high yield and low production cost [59], thus having a high industrial production value.
3. Research Progress on Taurine in the Relief of Visual Fatigue
3.1. Food Safety
The first reports on the safety of taurine for consumption began in 1999 when the Scientific Committee on Food (SCF) first evaluated the safety of 'energy' drinks containing taurine and concluded that taurine was not genotoxic, teratogenic nor carcinogenic [67]. More recently, Serrano et al. [68] have refined the data on the genotoxicity of taurine and their final results show that taurine did not show mutagenicity or genotoxicity in the Ames test, the micronucleus test nor the standard and enzyme-modified comet tests. The results of these toxicity studies on taurine confirm that it is not genotoxic, carcinogenic, or teratogenic [69–72]. Also, studies addressing taurine tolerance limits have been reported evaluating taurine with no observable adverse effect levels (NOAEL). There was also a risk assessment study in human clinical trials that reported that no adverse effects were observed in subjects given a maximum daily dose of 3 g/d of taurine [73]. Furthermore, the European Food Safety Authority (EFSA) opinion concluded that taurine added to energy drinks at a dose of 1000 mg/kg/day was the NOAEL [69].

In addition, toxicological evaluation tests with taurine compounding also reflect the good safety of taurine for consumption. Wang Xue et al. [74] evaluated the toxicological safety of taurine as the main ingredient of the compound powder (daily consumption of each ingredient: taurine 0.91 g/d, inositol 32 mg/d, vitamin E 100 mg/d) and showed that SPF-grade Kunming breed mice undergoing an acute toxicity test were judged to exhibit maximum tolerated dose (MTD) > 20.0 g/kg·bw. In the 30-d feeding test in SD rats, no abnormal changes were observed in the overall health status, physiological and biochemical functions, and organ histomorphology of the rats; the results of the mouse bone marrow multi-stained erythrocyte micronucleus test, sperm malformation test, and rat Ames test were uniformly negative, indicating that the taurine powder was safe, non-toxic, non-mutagenic and sub-chronic toxic. Similarly, the toxicological evaluation of a beverage made from taurine with L-carnitine, D-hydroglucose hydrochloride, caffeine, inositol, and B-vitamins was carried out and the final result was found to be non-toxic [75]. In addition, the toxicological evaluation of taurine in combination with selenium-enriched yeast or with lacto-mineral salts showed that the substance was safe and non-toxic [76,77].
These results show that taurine, a naturally occurring amino acid, has low toxicity.
3.2. Progress in Research on the Mechanism of Action of Taurine in Relieving Visual Fatigue
Normal retinal function is essential for maintaining proper visual function and eye health. Therefore, ways to protect and repair damaged retinal function can help alleviate the onset of visual fatigue. The main mechanisms by which taurine protects the retina to reduce visual fatigue are detailed in Figure 3.

【For more info:george.deng@wecistanche.com / WhatsApp:8613632399501】






