The Promoting Effect Of Cistanche Deserticola On Sexual Function
Feb 28, 2022
Li Gu, Wen Ting Xiong, Yan Lei Zhuang, Jian Shuang Zhang, and Xin Liu*
Food and Health Engineering Research Center of State Education Ministry, Sun Yat-sen University, Guangzhou, China
Abstract: Cistanche deserticola (CD) has been considered a tonic agent on reproductive function for thousands of years. The effects of CD extract on penis erectile response were investigated in the present study. After castration surgery, rats were treated intragastrically with CD extract (0.45, 0.90, and 1.8 g/kg) daily for four weeks. Penis erectile response was measured and the serum hormones were assayed at the end of the experiment. It was evaluated that the erectile latency became longer and the erectile duration shorter significantly in castrated rats compared to sham-operated controls. However, CD extract shortened the erectile latency and prolonged the erectile duration to minimize the negative effects of castration. At the dosage of 0.9g/kg, CD extract regulated the serum luteinizing hormone concentration approach to normal level in castrated rats. These findings indicated that CD facilitated the penis erectile response and modulated the serum hormone level to some extent.
Keywords: Cistanche deserticola; erectile response; castration; hormone.
Contact: joanna.jia@wecistanche.com / WhatsApp: 008618081934791

Cistanche has many effects, click here to know more
INTRODUCTION
Cistanche Hoffmg. Et Link, one of the genera of the Orobanchaceae family, is mainly distributed in the arid lands and deserts in the northern hemisphere, such as Inner Mongolia, Xinjiang, Ningxia autonomous regions of China, Iran, India, Mongolia, etc. The Cistanche species belong to the perennial parasite herbs, which commonly attach onto the roots of sand-fixing plants like Haloxylox ammodendron, H. persicum, Kalidium foliated and Tamarix plants, etc. (Jiang and Tu 2009).
Herba Cistanche, the stem of Cistanche species, has been considered as a superior tonic and earned the honor of “Ginseng of the deserts”. Among Cistanche species, Cistanche deserticola Y. C. MA has been indicated as the primary source material of Cistanche (Stefanova et al. 2011). However, due to over-harvesting in recent years, the natural resources of the wild C. deserticola (CD) is deficient which has been collected as one of the Class II plants needing protection in China, thus other species of this genus, such as C. salsa and C. Sinensis, are also used as substitutes in some areas. From first recorded in Shen Nong’s Chinese Material Medica (an ancient pharmacological compendium), the CD has been thought to tonify the kidney and invigorate the “Yang” in clinical practice under traditional Chinese medicine theory, which is alleged to be effective for reproduction, development and fertility function. Meanwhile, the CD has also been boiled with mutton, potato, or rice to produce a tonic food to treat overstrain-induced impairment for hundreds of years, suggesting that it is safe to take orally (Xiong et al. 2013). However, these anecdotal uses mainly exist in Chinese incunabula, with limited scientific information discussed in terms of modern pharmacological concepts.
Erectile dysfunction (ED) is a condition characterized by either inadequate erection of the penis or erection that does not last sufficiently long in adult males (Cohan and Korenman 2000). In response to modern, sophisticated, chemical agents, there has been a renewed interest to search for effective and safe medication from plants for the treatment of ED (Adimoelja 2000). A previous study (Liu et al. 2009) indicated that CD electuary accelerated the penis erection in normal rats, however, there were no data involved in the effect of CD on erectile dysfunction.
In animals, surgical castration with bilateral orchiectomy has been regarded as the standard of castration for investigating ED (Tombal 2005). Therefore, the present study was undertaken to evaluate the potential effects of CD against ED by investigating the erectile latency and duration in castrated rats.
MATERIALS AND METHODS
Chemicals and materials
Echinacoside (purity >98%) and acteoside (purity >98%) were purchased from the National Institutes for Food and Drug Control (Beijing, China). HPLC grade methanol was purchased from Sigma-Aldrich (St. Louis, MO, USA). Purified water was produced using a Millipore Milli-Q Integral 3 System and Q-POD Milli-Q System (Bedford, MA, USA). The stems of C. deserticola were purchased from Shenzhen GURU Biology Co., Ltd (Guangdong, China) and ground into a fine powder with a pulverizer passed through a 60mesh sieve.
HPLC analysis and plant material preparation Echinacoside and acteoside contents of CD powder were determined following our previous method (Zhao et al. 2011). Then, every 100 g dried powder was boiled in 1.5L water for 1.5 h. Cooling down at room temperature, the mixed liquor was centrifuged at 2000 rpm for 10 min (Eppendorf 5810R, Germany) and the precipitate was re-extracted twice under the same condition. The combined supernatants were concentrated by a vacuum rotary evaporator (Heidolph laborota 4001-efficient, Germany) at 60°C, and then lyophilized and yielded about 42% dried extract powder. When conducting the subsequent animal experiment, the dried extract was dissolved in purified water at room temperature.
Experimental animals
Adult male Sprague-Dawley rats (200-220g) were obtained from Medicine Experimental Animal Center of Guangdong Province (Guangzhou, PR China), maintained in temperature-controlled quarters with 12-hr light/dark cycles at room temperature (22-24°C) and constant humidity (40-70%) in SPF (specific pathogen-free) laboratory and fed ad libitum. The experimental procedures were in compliance with the National Institutes of Health Guide for Care and Use of Laboratory Animals.
Experimental procedure
One week before administration, fifty male SD rats were anesthetized with sodium pentobarbital intraperitoneally (40 mg/kg) (Jung et al. 1999) and randomly allocated to five groups: except rats in sham-operated group (Sham), the rest were bilaterally castrated via the scrotal approach (Anderes 2003, Schlatt et al. 2002). Meanwhile, the CD-treated groups were administered intragastrically with 0.45g/kg (low dose of CD extract, LCD +Cas), 0.9g/kg (median dose of CD extract, MCD +Cas), and 1.8g/kg (high dose of CD extract, HCD +Cas) CD extract, respectively, while the sham and castrated groups were administered with the corresponding amount of water only. Bodyweight measurement was taken at the beginning of the procedure and then every week. All rats underwent laparotomy immediately after death for removal and posterior weighing of the following organs: preputial glands, seminal vesicle, prostate gland, and levator ani muscle.
Penis erectile response measurement
The erectile function of rats in each group was assessed following the previous methods (Ji et al. 2009, Luo et al. 2006). Briefly, a stainless-steel bipolar electrode (BL- 420F Data Acquisition & Analysis System, Time Technology Co., Ltd, Chengdu, P. R. China) was carefully positioned on the penis until it was erected with the pulse of the electric stimulation (5 V) 0.2 s. Time from stimulation to erection was recorded as the penis erectile latency. When the penis erectile response was observed, the electrode could be withdrawn from the penis, and the time length that penis kept erection was recorded as the erectile duration.
Blood sampling and serum hormone analysis.
Rats were anesthetized with sodium pentobarbital intraperitoneally (40mg/kg) (Jung et al. 1999). Blood samples were drawn and clotted at 4°C for 4h, from which the serum was collected by centrifugation at 4000 rpm for 5 min at 4°C (Eppendorf 5810 R, Germany) and immediately stored at -20°C freezer until further analysis. Total LH and FSH levels were measured using commercial radioimmunoassay (RIA) kits (Beijing North Institute of Biological Technology, Beijing, China) referring to the manufacturer’s instructions (Matsumoto et al. 1986). Based on the competitive radioactive immune technique, I125-labeled LH (or FSH) was used in the assay. Briefly, 100µl I125-labeled LH (or FSH) agent was added into 100µl serum sample and incubated with 100µl purified rabbit antibody at 4°C (or 37°C) overnight. Then, donkey-anti-rabbit immune separating agent was added into antigen-antibody complex and centrifuged (3500 rpm, 15 min) to separate free and antibody-bound 125I- LH (or FSH). Afterward, the radioactivity of the precipitate was measured at radio immune assay γ counter (DMF-96, Zhongcheng electrical technology development Co., Ltd., Hefei, China). The sensitivity limits of the LH and FSH assay were both 1.0mIU/ml.
STATISTICAL ANALYSIS
Data analysis was performed using SPSS version 16.0 software. Values were expressed as mean ± standard deviation (SD). Parametric data were analyzed using analyses one-way analysis of variance (ANOVA) followed by post hoc LSD tests. Nonparametric data were analyzed by the Kruskal-Wallis test. Differences were considered statistically significant if the probability for deviation was less than 0.05.

RESULTS
Quantitative determination of echinacoside and acteoside contents of CD powder
The contents of echinacoside and acteoside in CD powder were 1.27±0.01% and 0.52±0.003%, which conformed to the requirement of Chinese Pharmacopoeia. The HPLC chromatogram of CD powder was shown in fig. 1.

Fig. 1 The HPLC chromatographic profile of Cistanche deserticola powder. Peak 1, echinacoside; Peak 2, acteoside. X axis, retention time; Y axis, absorbance unit. HPLC conditions: Waters HPLC system (Binary HPLC Pump1525, Refractive Index Detector 2414, Photodiode Array Detector 2996); stationary phase: Symmetry C18 (4.6mm×250 mm, 5 µm, Waters, Milford, MA, USA); mobile phase: 0.5% (V/V) acetic acid aqueous solution (A) and methanol (B) ingradient (0min, A: B=25:75; 10- 30 min, A: B=40: 60; 30 min, A: B=0: 100); flow rate: 1ml/min; detective wavelength: 330 nm; temperature: 30 C.

Body weights and relative accessory organs weights Shown in table 1, all rats were of similar initial body weights with an increase from day 1 to day 28. However, mean values in the castrated group increased from 216.8±9.2 g to 251.4±12.7, 283.3±15.1, 306.8±15.4, and 322.2±18.4g, gaining fewer weights compared to the sham-operated counterparts (p<0.05). There was no statistical difference between the sham group and CD treated group.

Further, the mean relative weights of the preputial gland, prostate gland & seminal vesicle, and levator ani muscle in castrated rats were significantly lower (p<0.05) than those of normal controls, respectively. Compared with the rats castrated only, the relative values of these organs' weight tended to be higher under the treatment of CD extract.
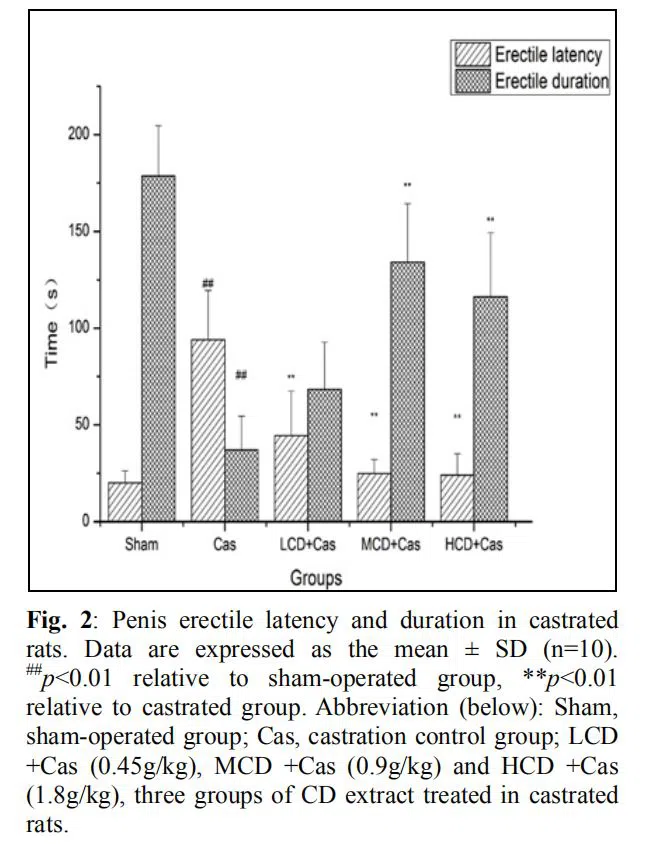
Erectile response assessment
In contrast to the sham group, castration made the rats spend more time gaining penis erection under electric stimulation (p<0.01). However, the erectile latency was shortened by CD extract, exhibiting a significant decrease of 52.7% in the LCD +Cas group (p<0.01), 73.5% in MCD
+Cas group (p<0.01) and 74.5% in HCD +Cas group (p<0.01), respectively. Furthermore, castration shortened the erectile duration with the mean value of 37.2±17.3 s compared to the sham-operated group (p<0.01). However, the erectile duration in rats was significantly prolonged approximately by 2.6-fold in the MCD +Cas group (p<0.01) and 2.1-fold in the HCD +Cas group (p<0.01). It showed no strict dose-related correlation. (fig. 2)

Serum hormone levels
Analyzed by RIA assay, the average serum concentration of LH in castrated rats was 2.50±0.34mIU/ml, showing an increase compared to sham-operated control (p<0.05). However, the value descended about 49.2% in MCD +Cas group relative to that in castrated control (p<0.05) (fig. 3). As shown in fig. 4, no statistical differences were observed among all groups on FSH concentration.
DISCUSSION
Our results demonstrated that castration significantly prolonged the penis erectile latency and shortened the erectile duration, with remarkable accessory organs atrophy. However, CD extract facilitated the penis erectile response and modulated the serum LH level to some extent in castrated rats.
The spontaneous erection is mainly stimulated and maintained by androgen via cellular, molecular, and physiologic mechanisms, involving the relaxation of cavernosal smooth muscle (Traish et al. 2007, Talha et al. 2007). Insufficient testosterone levels lead to a decrease in its relaxation (McClure 1988), which accordingly results in impotence in males (Bivalacqua et al. 2000). Especially in rats, it is maintained by a wide range of systemic testosterone concentrations as 10-12%, below which the erectile response is attenuated in a dose-dependent manner (Armagan et al. 2006). In the present study, the testosterone concentration in circulation was too low to meet the sensitivity limit (0.02ng/ml) in all castrated rats (the data were not shown in the results). However, CD extract significantly shortened the penis erectile latency at three dosages and prolonged the erectile duration at 0.90 g/kg and 1.8 g/kg. It was probably the consequence of the androgen-like effect of CD mentioned in some previous studies (He et al. 1996a, He et al. 1996b).
Some accessory organs, such as the prostate and seminal vesicle, are androgen-dependent, thus the weight changes may reflect the alterations in endocrine status (Trisomboon et al. 2007, Jang et al. 2011). It existed a threshold level of testosterone, below which the metabolism of these organs was inhibited (Trachtenberg 1985). It was confirmed in our study that castration induced the accessory reproductive organs weight decreased due to the testosterone deficiency.
Testosterone is primarily secreted by the testis Leydig cell and regulated by hypophysial hormones, such as luteinizing hormone (LH) and follicle-stimulating hormone (FSH), which play an important role in maintaining the penis function of the castrated animals (Robert et al. 2004). As the hypothalamus-pituitary-gonad (HPG) axis is a closed-loop feedback control mechanism, low gonadal hormone levels can regulate the secretion of LH and FSH in turn (Morales et al. 2004). Our study has investigated that the serum LH level was significantly increased in castrated rats and then descended to the normal levels by CD extract at 0.90 g/kg, which was agreed with the previous conclusion that increased LH level was associated with a higher risk of erectile dysfunction (Kupelian et al. 2006). In testis, LH stimulates synthesis and release of testosterone, the feedback control of which is relatively consistent, while FSH directly acts on the Sertoli cells for nourishment with the feedback control complex (Hellqvist et al. 2008). These findings offered more support for the surmise that CD expressed an androgen-like effect, which might be mainly responsible for the reduction of serum LH more than FSH.
In the current observation, the influence of castration on body weight development in male rats was similar to the findings in the previous study (Christoffersen et al. 2006), while contrary to the results in female rats (Pantaleão et al. 2010). CD extract was unable to significantly improve the bodyweight, which was probably due to the insufficient treatment duration or the complex impacts on overall health status.
In conclusion, castration prolonged the penis erectile latency and shortened the duration significantly, with serum LH increase and accessory sex organs atrophy. However, CD extract facilitated the penis erectile response by shortening the erectile latency and prolonging the erectile duration in castrated rats. These effects were probably due to the androgen-like effect, which was supported by the observation that CD extract normalized the serum LH concentrations, especially at the dosage of 0.90 g/kg. However, additional evidence is needed to demonstrate the effect of CD extract against ED at hypothalamic and limbic levels and more attention could be gained to this valuable traditional medicine in the modern world.

ACKNOWLEDGEMENTS
This work was supported by grants from the Science and Technology Support by Project in the Xinjiang Uygur Autonomous Region (P. R. China) (200840102-15).
REFERENCES
Adimoelja A (2000). Phytochemicals and the breakthrough of traditional herbs in the management of sexual dysfunctions. Int. J. Androl., 23: 82-84.
Anderes KL (2003). Biological characterization of a novel, orally active small molecule gonadotropin-releasing hormone (GnRH) antagonist using castrated and intact rats. J. Pharmacol. Exp. Ther., 305: 688-695. Armagan A, Kim NN, Goldstein I and Traish AM (2006). Dose-response relationship between testosterone and erectile function: Evidence for the existence of a
critical threshold. J. Androl., 27: 517-526.
Bivalacqua TJ, Champion HC, Hellstrom WJ, and Kadowitz PJ (2000). Pharmacotherapy for erectile dysfunction. Trends. Pharmacol. Sci., 7: 524-540.
Christoffersen B, Raun K, Svendsen O, Fledelius C and Golozoubova V (2006). Evaluation of the castrated male Sprague-Dawley rat as a model of the metabolic syndrome and type 2-diabetes. Int. J. Obes., 30: 1288- 1297.
Cohan P and Korenman SG (2000). Progress in erectile dysfunction and hormone function. J. Anti-aging Med., 3: 169-175.
Feldman HA, Goldstein I, Hatzichristou DG, Krane RJ and McKinlay JB (1994). Impotence and its medical and psychosocial correlates: Results of the Massachusetts male aging study. J. Urol., 151: 54-61.
He W, Shu XF, Zong GZ, Shi ML, Xiong YL and Chen MH (1996b). Studies on kidney reinforcing and Yang supporting the action of Cistanche deserticola Y.C. Ma before and after preparation. Chin. J. Chin. Mater. Med., 9: 534-537.
He W, Zong GZ, Wu GL and Chen MH (1996a). Exploration for active ingredients of Cistanche on androgen-like effect. Chin. J. Chin. Mater. Med., 9: 564-565.
Hellqvist A, Schmitz M and Borg B (2008). Effects of castration and androgen-treatment on the expression of FSH-β and LH-β in the three-spine stickleback, Gasterosteus aculeatus Feedback differences mediating the photoperiodic maturation response? Gen. Comp. Endocr., 158: 178-182.
Jang M, Min JW, In JG and Yang DC (2011). Effects of red ginseng extract on the epididymal sperm motility of mice exposed to ethanol. Int. J. Toxicol., 30: 435-442.
Ji DB, Ye J, Li CL, Wang YH, and Zhao J (2009). Antiaging effect of Cordyceps Sinensis extract. Phytother. Res., 23: 116-122.
Jiang Y and Tu PF (2009). Analysis of chemical constituents in Cistanche species. J. Chromatogr. A., 1216: 1970-1979.
Jung GW, Kwak JY, Kim IH, Koo MY, Park JI, Yoon S, Jung DG, Jung SI, Kwon HY and Yoon JH (1999). The role of growth factor on regeneration of nitric oxide synthase (NOS)- containing nerves after cavernous neurotomy in the rats. Int. J. Impot. Res., 11: 227-235.
Kupelian V, Shabsigh R, Travison TG, Page ST, Araujo AB and McKinlay JB (2006). Is there a relationship between sex hormones and erectile dysfunction? Results from the Massachusetts male aging study. J. Urology, 176: 2584-2588.
Liu ZR, Zhang D, Wu HJ, Li YL, Chen SP and Yang YM (2009). The effect of Cistanche electuary on nitric oxide synthase and kidney Yang nourishing. J. Med. & Pharm. Chin. Min., 5: 33-34.
Luo Q, Li Z, Huang X, Yan J, Zhang S, and Cai Y-Z (2006). Lycium barbarum polysaccharides: Protective effects against heat-induced damage of rat testes and H2O2-induced DNA damage in mouse testicular cells and beneficial effect on sexual behavior and reproductive function of Hemi castrated rats. Life Sci., 79: 613-621.
Matsumoto AM, Karpas AE and Bremner WJ (1986). Chronic human chorionic gonadotropin administration in normal men: evidence that follicle-stimulating hormone is necessary for the maintenance of quantitatively normal spermatogenesis in man. J. Clin. Endocrinol. Metab., 62: 1184-1192.
McClure RD (1988). Endocrine evaluation and therapy of erectile dysfunction. Urol. Clin. N. Am., 15: 53-64.
Morales A, Buvat J, Gooren LJ, Guay AT, Kaufman JM, Tan HM and Torres LO (2004). Endocrine aspects of sexual dysfunction in men. J. Sex Med., 1: 69-81.
Pantaleão TU, Mousovich F, Rosenthal D, Padrón ÁS, Carvalho DP and Costa VMCd (2010). Effect of serum estradiol and leptin levels on thyroid function, food intake, and body weight gain in female Wistar rats. Steroids, 75: 638-642.
Robert WH and Robert EB (2004). Hormonal regulation of spermatogenesis. Int. J. Androl., 27: 335-342.
Schlatt S, Kim SS and Gosden R (2002). Spermatogenesis and steroidogenesis in mouse, hamster, and monkey testicular tissue after cryopreservation and heterotopic grafting to castrated hosts. Reproduction, 124: 339-346. Stefanova NA, Fursova AZ, Sarsenbaev KN and Kolosova NG (2011). Effects of Cistanche deserticola on behavior and signs of cataract and retinopathy in senescence-accelerated OXYS rats. J. Ethnopharmacol., 138: 624-632.
Talha M, Bilal G, Gökhan T, Arı Z and Coşkun B (2007). A relationship of sex hormone levels and erectile dysfunction: Which tests should be done routinely? Yonsei. Med. J., 48: 1015.
Tombal B (2005). Appropriate castration with luteinizing hormone-releasing hormone (LHRH) agonists: What is the optimal level of testosterone? Eur. Urol. Suppl., 4: 14-19.
Trachtenberg J (1985). Optimal testosterone concentration for the treatment of prostatic cancer. J. Urology, 133: 888-890.
Traish AM, Goldstein I and Kim NN (2007). Testosterone and erectile function: from basic research to a new clinical paradigm for managing men with androgen insufficiency and erectile dysfunction. Eur. Urol., 52: 54-70.
Trisomboon H, Watanabe G, Wetchasit P and Taya K (2007). Effect of daily treatment with Thai herb, Kaempferia parviflora, in Hershberger assay using castrated immature rats. J. Reprod. Develop, 53: 351- 356.
Xiong WT, Gu L, Wang C, Sun HX and Liu X (2013). Anti-hyperglycemic and hypolipidemic effects of Cistanche tubulosa in type 2 diabetic db/db mice. J. Ethnopharmacol., 150: 935-945.
Zhao SY, He XY, Jia BG, Peng QY and Liu X (2011). Production of acteoside from Cistanche tubulosa by beta-glucosidase. Pak. J. Pharm. Sci., 24: 135-141.






