Type 1 Dendritic Cells (DCs) in Kidney Diseases
Mar 17, 2022
for more information:ali.ma@wecistanche.com
Conventional Type 1 Dendritic Cells (cDC1) in Human Kidney Diseases: Clinico-Pathological Correlations
Titi Chen, Qi Cao & et al.
ABSTRACT
Background: Type 1 dendritic cells (cDC1) is a subset of conventional DCs, whose most recognized function is cross-presentation to CD8+ T cells. We conducted this study to investigate the number and location of Type 1 dendritic cells (cDC1s) in various human kidney diseases as well as their correlation with clinicopathological features and CD8 T cells.
Methods: We analyzed 135 kidney biopsies samples. Kidney diseases included: acute tubular necrosis (ATN), acute interstitial nephritis (AIN), proliferative glomerulonephritis (GN)(IgA nephropathy, lupus nephritis, pauci-immune GN, anti-GBM disease), non-proliferative GN (minimal change disease, membranous nephropathy), and diabetic nephropathy. Indirect immunofluorescence staining was used to quantify cDC1s (Type 1 dendritic cells), CD1ct DCs, and CD8+ T cells.
Results: Type 1 dendritic cells (cDC1s) were rarely present in normal kidneys. Their number increased significantly in ATN and proliferative GN, proportionally much more than CD1ct DCs. cDC1s (Type 1 dendritic cells) were mainly found in the interstitium, except in lupus nephritis,pauci-immune GN, and anti-GBM disease, where they were prominent in glomeruli and peri-glomerular regions. The number of cDC1s (Type 1 dendritic cells) correlated with disease severity in ATN, number of crescents in pauci-immune GN, interstitial fibrosis in lgA nephropathy, and lupus nephritis, as well as prognosis in IgA nephropathy. The number of CD8+ T cells also increased significantly in these conditions and the cDC1 (Type 1 dendritic cells) number correlated with CD8+ Tcell number in lupus nephritis and pauci-immune GN, with many of them closely co-localized. Conclusions: cDC1 (Type 1 dendritic cells) number correlated with various clinicopathological features and prognosis reflecting a possible role in these conditions. Their association with CD8+ T cells suggests a combined mechanism in keeping with the results in animal models.
INTRODUCTION
Dendritic cells (DCs) are the central orchestrators of effective immunity. These cells are heterogeneous and can be divided into distinct subsets based on their phenotype and function. Dendritic cells can be broadly categorized into plasmacytoid DCs (Dendritic Cells) (pDC) and conventional DCs (Dendritic Cells) (cDC).cDC consists of two major subsets: cDC1 (Type 1 dendritic cells)(CD141* DCs (Dendritic Cells) in humans and CD103t or CD8ot DCs in rodents) and cDC2(CD1c DCs in humans and CD1 lbt DCs in rodents) (1).cDC1 (Type 1 dendritic cells) were discovered first in mice and then in humans and are characterized by their superior ability to phagocytose necrotic cells through damage-associated molecular pattern (DAMP) receptor Clec9A and to cross-present to CD8+ T cells. In contrast, cDC2s are effective CD4* T cell activators but inferior at CD8t T cell activation (2).
DCs (Dendritic Cells) have been studied in human kidney disease and their number was found to be increased in glomerulonephritis(GN)(3, 4). After the discovery of cDCl, accumulating animal studies have shown that they play a pivotal role in kidney diseases, such as in adriamycin nephropathy and crescentic GN, through interaction with T cells (5-8). However, such studies are lacking in human kidney disease, with only one study demonstrating increased numbers of cDCs (Conventional Dendritic Cells) in GN (9). We conducted this study to provide analysis of the key cDC subsets, cDC1 (Type 1 dendritic cells), and cDC2, in a wide range of human kidney diseases including non-glomerular diseases [acute tubular necrosis (ATN), acute interstitial nephritis(AIN)], proliferative GN (IgA nephropathy, lupus nephritis, pauci-immune GN,anti-GBM disease), non-proliferative GN (minimal change disease (MCD), membranous nephropathy) and diabetic nephropathy. We found cDC is to be significantly correlated with pathological features including the severity of ATN, crescent formation in pauci-immune GN, and interstitial fibrosis in immune-mediated GN.In addition, consistent with their specialized ability to activate CD8+ T cells in animal models, we also demonstrated their correlation with CD8t T cells in these samples. These findings provide an impetus to explore new therapeutic targets that manipulate these cells for the treatment of kidney diseases.

Click to Cistanche stem for kidney disease
MATERIAL AND METHODS
Patients and Tissue Samples
The study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Human Research Ethics Committee of the Western Sydney Local Health District. Informed patient consent was obtained. We analyzed 176 frozen diagnostic kidney biopsy samples taken from non-pregnant adult patients(>18 years old) between 18 June 2016 to 30* June 2017 in Westmead Hospital, Sydney, Australia. Kidney tissues were snap-frozen in O.C.T.compound (Tissue-Tek Sakura, USA)and stored at-80°C. Forty-one(41)samples had poor tissue quality and were excluded from the study. Baseline patient data including age, gender, estimated glomerular filtration rate (eGFR), and degree of proteinuria were collected at the time of kidney biopsy.eGFR was calculated using the CKD-EPI formula. The diagnosis was made by a renal pathologist, based on light, immunofluorescence(IF), and electron microscopy as well as clinical history.

A variety of diseases were analyzed including non-glomerular diseases(ATN, AIN), proliferative GN(IgA nephropathy, lupus nephritis, pauci-immune GN, anti-GBM disease), non-proliferative GN(MCD, membranous nephropathy), and diabetic nephropathy. For ATN, we only included non-septic cases as septic ATN has different pathophysiology. ATN was further categorized into mild-moderate disease(defined as<50%cortical tubules showing injury)and severe disease (>=50%cortical tubules showing injury). IgA nephropathy was classified according to the Oxford MEST score (M mesangial hypercellularity, E endocapillary hypercellularity, S segmental glomerulosclerosis, T tubular atrophy/interstitial fibrosis). We also analyzed the correlation between cDC1 (Type 1 dendritic cells) number and prognosis (defined a priori as >20% reduction in eGFR on or before 31t December 2019)in IgA nephropathy by dividing patients into 2 groups according to cDC number with a cut-off point at the upper quartile(>=15). Lupus nephritis was further classified into 2 groups according to the level of interstitial fibrosis(<25%,>=25% cortical interstitial involvement). Pauci-immune GN was classified into 2 groups according to the percentage of glomeruli with crescents(<40%,>=40%).

As normal kidney controls, we used 5 normal renal cortices from nephrectomy samples as well as 2 donor kidneys not suitable for transplant. For tumor nephrectomies, samples were taken from the pole opposite to the tumor and at least 5 cm from the tumor margin. These tissues had a normal macroscopic appearance. Microscopically, none of these kidney samples had evidence of significant glomerular or tubulointerstitial inflammation or injury. We used normal adult donor splenic tissue as a positive control for testing antibodies.

Immunofluorescence Staining
Serial cryostat sections were cut at 5μm and placed onto Superfrost Ultra Plus glass slides (Thermo Scientific, USA). Slides were stored at -80°C. Tissue sections were fixed with 100% methanol at -20°C for 10 minutes and then air-dried. Indirect immunofluorescence staining was performed using the following method: tissue sections were washed in DPBS(Lonza, USA)and blocked with 2% Bovine Serum Albumin(Sigma-Aldrich, USA) for 15 minutes; then they were stained with primary antibody at 4°C overnight followed by secondary antibodies for 40 minutes at room temperature. Table 1 is a list of primary and secondary antibodies used and their dilutions. Nuclei were stained with DAPI(1:250,000, ThermoFisher, USA)before samples were mounted on coverslips with fluorescence mounting medium(Dako, USA). Non-specific staining and cross-reactivity between different primary and secondary antibodies were checked and excluded.
TABLE 1 | Primary and secondary antibodies used in the study
cDCls were identified by staining for marker Clec9A. In humans, Clec9A expression is highly restricted to cDCls in blood and tissues(10, 11). To confirm this in the kidneys, we performed double staining of Clec9A and HLA-DRB1 as well as Clec9A and CD1lc in selected normal and diseased conditions. Most, if not all Clec9A overlapped with HLA-DRB1 (Supplementary Figure 1)and CD1lc(Supplementary Figure 2).
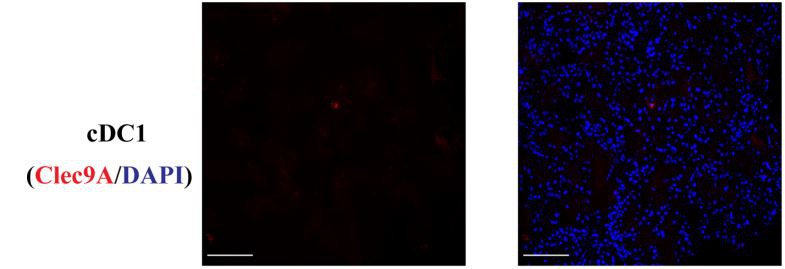
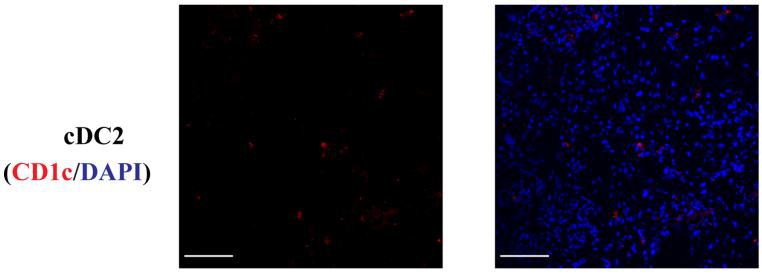
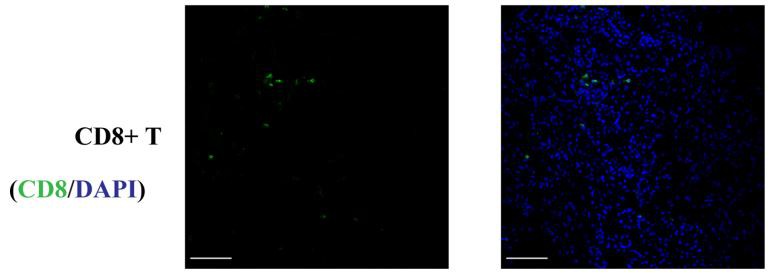
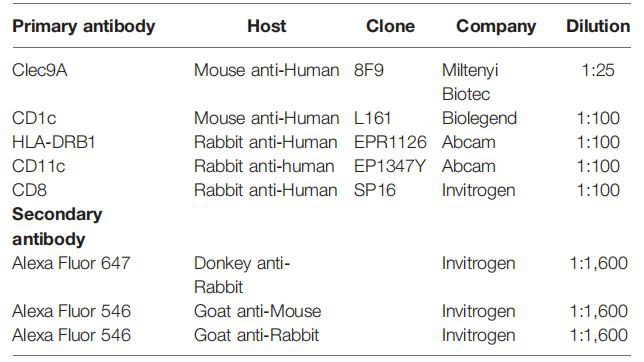
FIGURE 1 | Normal kidney cDC1 (Type 1 dendritic cells), cDC2, and CD8+ T cells.
DCs (Dendritic Cells) were rarely present in normal kidneys and cDC2 numbers were approximately 7 times the number of cDC1 (Type 1 dendritic cells). (Bar = 100 mm).
RESULTS
Patient's Baseline Characteristics. The baseline characteristics of patients in control and disease cohorts are summarized in Table 2. A total of 135 patients were included in the study.A wide range of kidney diseases were analyzed including non-glomerular diseases[ATN (22), AIN (10)], proliferative GN[gA nephropathy (44), lupus nephritis (12), pauci-immune GN(12),anti-GBM disease(4)], non-proliferative GN[MCD(5), membranous nephropathy(5)]and diabetic nephropathy (21). There were more females in the lupus nephritis group and their age tended to be younger compared with other kidney diseases, which is consistent with the literature(12). Patients with pauci-immune GN and anti-GBM disease had the lowest eGFR. The proteinuria level was the highest in MCD and membranous nephropathy.
TABLE 2 | Baseline characteristics.

Number and Location of DCs (Dendritic Cells) in Control and Disease. cDCls were rarely present in normal kidneys(Figure 1) and cDC2 numbers were approximately 7 times the number of cDC1 (Type 1 dendritic cells).The number of cDC1 (Type 1 dendritic cells) increased significantly in ATN and proliferative GN(Figure 2A), while their number remained unchanged compared to control in AIN, membranous nephropathy, MCD, and diabetic nephropathy (Supplementary Figure 3). The number of cDC2 also increased significantly in ATN and proliferative GN(Figure 2B). There was a reduction in the cDC2 (Type 2 dendritic cells)/cDC1 (Type 1 dendritic cells) ratio indicating cDCl (Type 1 dendritic cells) increased proportionally more than cDC2 (Figure 2C).

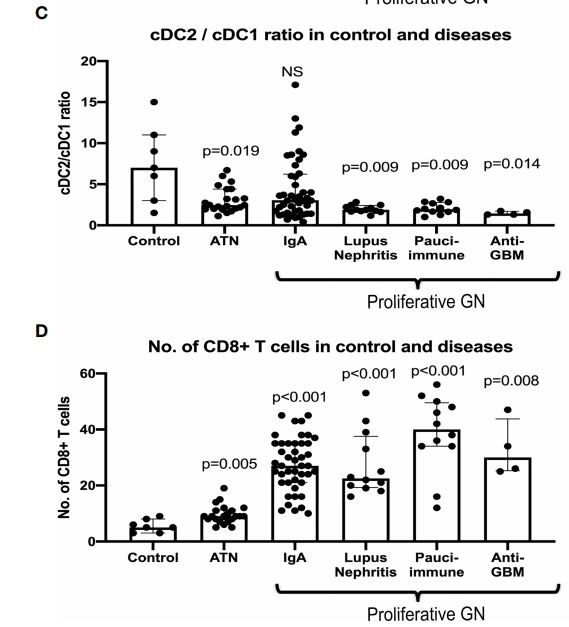
FIGURE 2 | Number of cDC1 (Type 1 dendritic cells) and cDC2, cDC2/cDC1 ratio, and CD8+ T cell in control and selected diseases.
P-value is calculated for each disease versus control. Both cDC1 (Type 1 dendritic cells) and cDC2 increased significantly in ATN, IgA, lupus nephritis, pauci-immune GN and anti-GBM disease (A, B)
with cDC1 (Type 1 dendritic cells) increased proportionally more than cDC2 in ATN, lupus nephritis, pauci-immune GN, and anti-GBM disease (C).
CD8+ T cells were increased significantly in ATN, IgA, lupus nephritis, pauci-immune GN, and anti-GBM disease (D).
Most cDCls were located in the interstitium, except in lupus nephritis, pauci-immune GN, and anti-GBM disease where they were also found in peri-glomerular and intra-glomerular regions (Figure 3A).In addition, we also found a significant number of CD8t T cells in peri-glomerular and intra-glomerular regions (Figure 3A), and many of them co-localized with cDCls(Figure 3B).On the other hand, cDC2s were rarely found in intra-glomerular regions and there was minimal co-localization with CD8+ T cells.
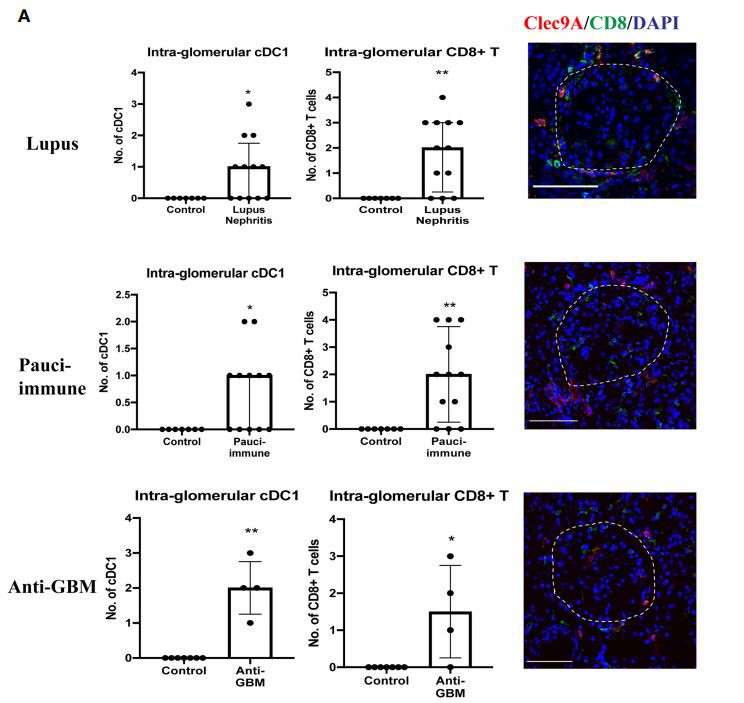

FIGURE 3 | (A) cDC1 (Type 1 dendritic cells) and CD8+ T cells in intra-glomerular regions. (B) cDC1 co-localization with CD8+ T cells. (Bar = 100 mm). * P < 0.05, ** P < 0.01.
Association Between cDC1 (Type 1 dendritic cells) With Clinical-Pathological Features and CD8+ T Cells We analyzed the correlation between cDCl (Type 1 dendritic cells) and clinicopathological features as well as CD8+ T cells. There were 22 cases of ATN(mild-mod disease n=12, severe disease n=10). More severe disease was associated with a higher number of cDC1 (Type 1 dendritic cells)(p=0.032), but not cDC2(Figure 4A).cDC1 (Type 1 dendritic cells) increased proportionally more than cDC2(cDC2/cDC1 ratio 2.5, p=0.019).The number of CD8+ T cells also increased significantly in ATN(p=0.005) (Table3,Figure2D).The number of cDC1 (Type 1 dendritic cells) did not correlate with CD8* T cell number in ATN.
TABLE 3 | CD8+ T cell number and correlation coefficient between cDC1 (Type 1 dendritic cells) and CD8+T cells numbers in control and diseased kidneys.

Forty-four cases of IgA nephropathy were analyzed. Using the Oxford classification MEST score(M mesangial hypercellularity, E endocapillary hypercellularity, S segmental glomerulosclerosis, T tubular atrophy/interstitial fibrosis), a higher number of cDC1 (Type 1 dendritic cells) was associated with a higher T score(p=0.008), but not MES scores(Figure 4B). There were 7 cases with crescents and the number of cCD1 was not associated with the number of crescents. The number of kidney CD8* T cells was significantly higher in IgA nephropathy than in control (p<0.001) (Table 3, Figure 2D). There was no correlation between cDC1 (Type 1 dendritic cells) and CD8+ T cell numbers. Thirty-five(35)patients had follow-up data on or before 31sf December 2019, of whom9experienced>20% reduction in eGFR. Dividing patients into 2 groups according to cDCl (Type 1 dendritic cells) number with a cut-off point at the upper quartile(>=15), the higher cDC1 (Type 1 dendritic cells) number group was associated with worse outcomes (Figure 4E).
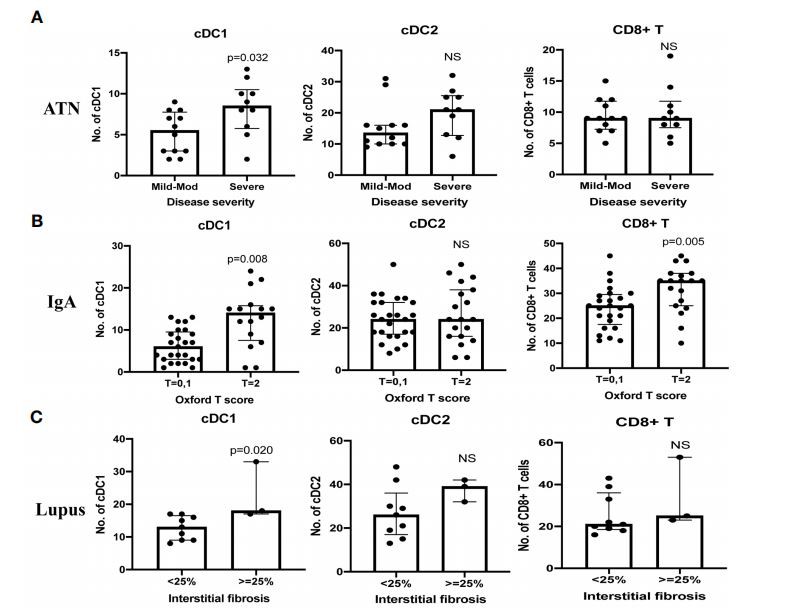

FIGURE 4 | Correlation between cDC1 (Type 1 dendritic cells), cDC2, and CD8+ T cell number and disease severity (A–D).
Kaplan Meier curve of IgA nephropathy survival (E). T score 0 refers to the percentage of the area showing tubular atrophy/interstitial fibrosis < = 25%,
T score 1 refers to the percentage of the area showing tubular atrophy/interstitial fibrosis 26 - 50%, T score 2 refers to the percentage of the area showing tubular atrophy/interstitial fibrosis > 50%.
Survival was defined as > 20% reduction in eGFR on or before 31st December 2019. NS, not significant.
There were 12 cases of lupus nephritis. As in IgA nephropathy, a higher cDCl number was associated with more severe fibrosis (p=0.020)(Figure 4C).In addition, CD8+ T cell number also increased significantly(P<0.001) (Table 3, Figure 2D) and this correlated with the number of cDCl cells (r=0.614, p=0.034) (Table 3).A significant number of cDC1 (Type 1 dendritic cells) and CD8t T cells were found in peri-glomerular as well as intra-glomerular indicating they may play a role in this condition, Second, in interstitial fibrosis associated immune-mediated disease (IgA nephropathy and lupus nephritis), we found that cDC1 (Type 1 dendritic cells) number correlated with the severity of fibrosis, as well as prognosis in IgA nephropathy, while no such correlation was found in non-immune mediated fibrotic disease(diabetic nephropathy). Third, there was a strong correlation between cDCl number and crescent formation in pauci-immune GN, and cDCl were present in large numbers in peri-glomerular and intra-glomerular regions indicating their possible role in a crescent formation. Fourth, the number of cDC1 correlated with CD8+T cell numbers in lupus nephritis and pauci-immune GN, with numerous cDCls co-localized with CD8+ T cells suggesting their possible interaction. This is in keeping with our findings in animal models of kidney disease that show murine homologs of cDC1 (Type 1 dendritic cells) cells preferentially activate CD8t T cells. Taken together, these findings suggest that cDCl plays an important role across a range of kidney diseases including ATN, interstitial fibrosis in immune-mediated disease, and crescent formation and that they may potentially act through activation of CD8t T cells.
cDC1 (Type 1 dendritic cells) Number Correlated With Disease Severity in ATN. In non-septic ATN, for the first time, we showed a significantly increased number of DCs (Dendritic Cells), especially cDCls compared with cDC2s. Importantly, the cDC1 (Type 1 dendritic cells) number correlated with disease severity. In addition, the CD8+ T cell number was also increased. ATN is usually caused by ischemia-reperfusion injury(IRI), nephrotoxins, or sepsis. Traditionally, IRI and toxins cause sterile inflammation, and innate immunity, where DCs and T-cells are less important, was considered to play a dominant role. However, increasing evidence from animal studies has shown both cDCl and CD8+ T cells are important players. Previous studies in animal IRI and cisplatin nephrotoxicity found the total DCand T cell numbers increased(13-17). Our previous studies showed in Adriamycin nephropathy, cDCl numbers increased significantly (5). In IRI, a subset of activated dendritic cells demonstrates increased capacity to the cross-present antigen to CD8t T cells(18). In Adriamycin nephropathy, we also found cDCls elicited a CD8+ T cell response leading to injury (5). The lack of a correlation between cDC1 (Type 1 dendritic cells) and CD8* T cell in human ATN in contrast to the findings in animal models may reflect different non-immunological pathways of injury in human ATN compared to the IRI in mouse models where cDCls play a significant role through CD8* T cells. In addition, it has been demonstrated in rodents that cDCl is recruited into the tissue by chemoattractant XCLl produced by natural killer cells(19). Therefore, it may be worthwhile to study this further in kidney disease.
cDC1 (Type 1 dendritic cells) Number Correlated With Immune-Mediated Interstitial Fibrosis. The second significant finding in this study is that cDCl number correlated with the severity of interstitial fibrosis associated with immune-mediated disease (IgA nephropathy and lupus nephritis), but not in non-immune mediated fibrotic disease (diabetic nephropathy), the Previous study showed increased numbers of cDCls and cDC2s in interstitial fibrosis(9). We, for the first time, showed that this is only true in immune-mediated disease and cDC1 (Type 1 dendritic cells) number increased proportionally more than cDC2s. In addition, we also demonstrated cDC1 (Type 1 dendritic cells) number correlated with prognosis and the number of CD8* T cells also increased significantly in IgA nephropathy. This is supported by animal studies showing DCs (Dendritic Cells) directly contribute to fibrosis. For example, DC-derived amphiregulin promoted fibrosis(20). It is also possible that cDCls contributed to fibrosis through CD8+ T cells, which are known to contribute to fibrosis in other organs(21-23). Since interstitial fibrosis is linked to the progression of chronic kidney disease, it is not surprising that we found the cDC1 (Type 1 dendritic cells) number to be a good prognostic marker in IgA nephropathy. Other studies have shown CD8t T cells correlated with the prognosis of IgA nephropathy (24), which may be the result of cross-presentation from cDCls. In lupus nephritis, we demonstrated a correlation between cDC1 (Type 1 dendritic cells) number and interstitial fibrosis as well as the number of CD8t T cells. Previous studies have also shown an increased kidney cDC1 (Type 1 dendritic cells) number in lupus nephritis(25), especially class III and VI lupus nephritis, with a corresponding reduction in their circulating numbers (26). We extended these findings by showing that they also correlated with chronic changes. It is well established that interstitial inflammation, which is comprised of T cells, B cells, dendritic cells, and macrophages, has a dominant role in the progression of lupus nephritis (27).cDCls may contribute to the progression of lupus nephritis in a variety of ways. First, activation of interferon plays a key role in the pathogenesis of lupus nephritis(28-30) and cDCls are a prominent producer of IFN-入.(31)Second, cDC1 (Type 1 dendritic cells) may contribute to lupus nephritis progression through CD8t T cells(32-37)and our finding of a correlation between cDCls and CD8+ T further supports their possible interaction. The role of CD8+ T cells in lupus nephritis has been previously demonstrated. CD8+ T cells control autoreactive immunity by the release of cytotoxic molecules. CD8+T cells in lupus nephritis were found to have dampened cytotoxic function, which can trigger autoimmunity(38). In addition, these cells can also generate lupus autoantigens(39). There has been abundant evidence that CD8* T cells in both kidney (32,33)and urine(34-36)correlate with disease activity and histological injury in lupus nephritis. In addition, CD8* T-cell exhaustion was shown to predict a favorable prognosis(37).
cDC1 (Type 1 dendritic cells) Number Correlated With the Number of Crescents in Pauci-Immune GN. In pauci-immune GN, we found cDCls aggregated in the peri-glomerular and intra-glomerular regions, and their number correlated with the number of crescents and CD8+ T cells. Previous studies showed that DCs (Dendritic Cells) are rarely present inside the glomerulus(3,4,9) or only in very small numbers(40).On the other hand, T cells were prominent in the interstitium, peri-glomerular, and intra-glomerular regions(41-44). We found that both cDCls and CD8* T cells are prominent in the peri-glomerular and intra-glomerular regions with many of them co-localized. In addition, the cDC1 (Type 1 dendritic cells) number correlated with crescent and CD8+ T cell numbers. All of these findings suggest a role for cDCl in crescent formation through interaction with CD8* T cells. The pathogenic role of CD8t T cells in pauci-immune GN and crescent formation has already been demonstrated in animal models(45,46). Consistent with our findings in humans, in animal crescentic GN, cDC1 (Type 1 dendritic cells) and CD8* T cells were found especially in the periglomerular region(47,48). It has been shown that Bowman's capsule provides a protected immunological niche by preventing access of DCs and cytotoxic CD8+ T cells to Bowman's space and thereby podocytes(45,47). However, when Bowman's capsule was breached, these inflammatory cells gained access and destroyed podocytes resulting in rapidly progressive GN(47).
One limitation of this study is that the IF staining technique allows the use of only a limited number of markers. It would be beneficial to extend our findings using technology such as flow cytometry, which can combine multiple markers to further analyze the phenotype of these DCs (Dendritic Cells) and their relevant cytokine and chemokine profiles. However, location information will be lost. Other techniques such as multiplex immunohistochemistry and Nanostring can also be considered in future studies to further examine these cells in kidney disease. In addition, when staining cDC2s using CD1c, HLA-DRB1, and CD1lc, there may be a small percentage of B cells that express these markers as well, which has not been ruled out.
CONCLUSIONS
Even though cDC1 (Type 1 dendritic cells) comprises a minor subset of DCs (Dendritic Cells) under homeostatic conditions, this study demonstrates a significant correlation between this cell population and clinicopathological features in human kidney disease. This reflects their likely importance in disease processes such as ATN, crescent formation in proliferative GN, and interstitial fibrosis in immune-mediated GN. In addition, their co-localization and correlation with CD8t T cells may provide an explanation for their mechanism of action, corroborating data from animal models. These findings provide an impetus to explore new therapeutic targets that manipulate these cells for the treatment of kidney diseases, as we have done in animal studies(6), and to investigate their use as a prognostic marker. Further studies in both humans and animals are needed to interrogate the role of cDC1s (Type 1 dendritic cells), their mechanism of action, and how best to target them therapeutically.
REFERENCES
1. Pakalniskyte D, Schraml BU. Tissue-Specific Diversity and Functions of Conventional Dendritic Cells. Adv Immunol(2017)134:89-135.doi:10.1016/bs.ai.2017.01.003
2. Merad M, Sathe P, Helft J, Miller J, Mortha A. The Dendritic Cell Lineage: Ontogeny and Function of Dendritic Cells and Their Subsets in the Steady State and the Inflamed Setting. Annu Rev Immunol (2013)31:563-604.doi:10.1146/annual immunol-020711-074950
3. Segerer S, Heller F, Lindenmeyer MT, Schmid H, Cohen CD, Draganovici D, et al. Compartment Specific Expression of Dendritic Cell Markers in Human Glomerulonephritis. Kidney Int (2008)74(1):37-46.doi:10.1038/ki.2008.99
4. Woltman AM,de Fiter JW,Zuidwijk K, Vlug AG,Bajema IM, van der Kooij SW, et al. Quantification of Dendritic Cell Subsets in Human Renal Tissue Under Normal and Pathological Conditions.Kidney Int (2007)71(10):1001-8. doi: 10.1038/sj.ki.5002187
5. Cao Q, Lu J, LiQ, Wang C Wang XM, Lee VW, et al. Cd103+Dendritic Cells Elicit Cd8+ T Cell Responses to Accelerate Kidney Injury in Adriamycin Nephropathy. J Am Soc Nephrol (2016) 27(5):1344-60.doi: 10.1681/ASN.2015030229
6. Wang R, Chen T, Wang C, Zhang Z, Wang XM, Li Q, et al.Flt3 Inhibition Alleviates Chronic Kidney Disease by Suppressing CD103+ Dendritic Cell-Mediated T Cell Activation. Nephrol Dial Transplant(2019)34(11):1853-63. DOI: 10.1093/gfy385
7. Evers BD, Engel DR, Bohner AM, Tittel AP, Krause TA, Heuser C, et al Cd103+ Kidney Dendritic Cells Protect Against Crescentic GN by Maintaining Il-10-Producing Regulatory T Cells. J Am Soc Nephrol (2016)27(11):3368-82. DOI: 10.1681/ASN.2015080873
8. Kitching AR, Ooi JD. Renal Dendritic Cells: The Long and Winding Road. J Am Soc Nephrol (2018)29(1):4-7.doi: 10.1681/ASN.2017101145
9. Kassianos AJ, Wang X, Sampangi S, Muczynski K, Healy H, Wilkinson R. Increased Tubulointerstitial Recruitment of Human CD141(hi)CLEC9A(+)and CD1c(+)Myeloid Dendritic Cell Subsets in Renal Fibrosis and BKidney B. Am J Physiol Renal Physiol (2013)305(10): F1391-401. doi:10.1152/adrenal.00318.2013
10. Guilliams M, Dutertre CA, Scott CL, McGovern N, Sichien D, Chakaro S, et al. Unsupervised High-Dimensional Analysis Aligns Dendritic Cells Across Tissues and Species. Immunity (2016)45(3):669-84.doi: 10.1016/j.immuni.2016.08.015
11. Villani AC, Satija R, Reynolds G, Sarkizova S, Shekhar K, Fletcher J, et al. Single-Cell RNA-seq Reveals New Types of Human Blood Dendritic Cells, Monocytes, and Progenitors. Science(2017)356(6335):4-6. DOI: 10.1126/science.aah4573
12. Almaani S, Meara A, Rovin BH. Update on Lupus Nephritis. Clin I Am Soc Nephrol (2017)12(5):825-35.doi: 10.2215/CJIN.05780616
Note: the above is not a full reference list






