Acteoside in Chinese Natural Plant-Cistanche To Ameliorate Kidney Disease--Part II
Mar 02, 2022
Contact: emily.li@wecistanche.com
Click here for information about Part I (introduction, materials, and methods )of this article.
Qinwen Wang | Xinxin Dai et.al
This study was designed to investigate the protective effects and mechanisms of acteoside on DKD in diabetes male db/db mice and high glucose-induced HK-2 cells. The diabetes db/db mice were divided randomly into the model group, metformin group, irbesartan group, and acteoside group. We observed the natural product of acteoside exhibiting a significant effect in renal protection through analyzing biochemical indicators and endogenous metabolites, histopathological observations, and western blotting. HK-2 cells subjected to high glucose were used in in-vitro experiments. The molecular mechanisms were investigated by RT-PCR and western blot. Acteoside prevents high glucose-induced HK-2 cells and diabetes db/db mice by inhibiting the NADPH/oxidase-TGF-β/Smad signaling pathway. Acteoside regulated the disturbed metabolic pathway of lipid metabolism, glyoxylate and dicarboxylate metabolism, and arachidonic acid metabolism. We discovered the natural product of acteoside exhibits a significant effect on renal protection. This study paved the way for further exploration of pathogenesis, early diagnosis, and the development of a new therapeutic agent for DKD.
KEYWORDS acteoside, diabetic kidney disease, metabolic profiling, NADPH/oxidase-TGF-β/Smad signaling pathway, ROS
Part II
Acteoside treat Kidney Disease
3 | RESULTS
3.1 | Acteoside inhibited the NADPH/oxidaseTGF-β/Smad signaling pathway in diabetic db/db mice
3.1.1 | Acteoside improved the biochemical indicators
The analytical results of biochemical indicators are presented in Figure 1B. Compared with the control group, the biochemical indicators of the model group were significantly increased, such as BUN (p < .05), FBG (p < .001), GOT (p < .01), GPT (p < .01), INS (p < .05), T-CHO (p < .001), TG (p < .001), Scr (p < .05), and mALB (p < .05). After treatment of MRHTG, the levels of BUN (p < .05), FBG (p < .001), GOT (p < .01), GPT (p < .01), INS (p < .05), T-CHO (p < .05), TG (p < .01), Scr (p < .05) in serum, and mALB (p < .05) in urine were nearly restored to normal. In MRHTG, the levels of FBG, GOT, GPT, and INS were lower than those in the other groups after treatment.
Compared with the model group, the expression levels of BUN, GOT, INS, Scr in serum (p < .05) were nearly restored to normal, and the expression levels of FBG, GPT, T-CHO, TG (p < .05) in serum, and mALB (p < .05) in urine were decreased, after the treatment of EJSG (250 mg kg-1 d-1). Compared with model group, the expression levels of BUN (p < .05), GOT (p < .05), INS (p < .05), Scr (p < .05) in serum, and mALB in urine were nearly restored to normal, and the expression levels of FBG, GPT (p < .05), T-CHO, TG (p < .05) in serum were decreased, after the treatment of EBST (50 mg kg-1 d-1). In general, MRHTG had a better effect than positive drugs EJSG and EBST. Compared with the positive drug group, MRHTG has a better effect on the same biochemical indicators such as FBG (p < .001) and INS (p < .5). At the level of TG, GPT, GOT, BUN, and Scr, the result of MRHTG is the same as EBST and EJSG.

Treat Kidney disease--Cistanche acteoside
3.1.2 | Acteoside alleviated pathological changes
The results of the histological examination were consistent with the biochemical analysis (see Figure 1C). The results of PAS showed that compared with the control group, the glomerular area increased, the basement membrane thickened, and the average optical density value increased significantly in the model group (p < .05); HE staining showed that the structure of renal interstitium, glomerulus, and tubule was basically normal in the control group (Cao et al., 2019). However, the model group revealed glomerular basement membrane thickened and mesangial hyperplasia a small area of the cortex showed a decrease in the eosinophilic cytoplasm of the renal tubular epithelial cells (Xiang et al., 2020).
3.1.3 | The expression of α-SMA, TGF-β1, Smad2, Smad3, P-Smad2/3, Smad4, NOX1, NOX2, and NOX4 protein in kidney tissue of db/db mice
Western blotting analysis (Figure 1D) showed that the expression levels of NOX1, NOX2, NOX4, α-SMA, TGF-β1, Smad2, Smad3, PSmad2/3, Smad4, NOX1, NOX2, and NOX4 in renal tissue of db/db mice were significantly increased from that in db/m mice (p < .05). In general, the positive drug EBST had a better effect than EJSG and MRHTG. But MRHTG had the advantage of regulating the level of P-Smad2/3 protein. After the treatment of MRHTG, the expression levels of Smad2, Smad4, and NOX4 were significantly decreased compared with the model group. The protein expression levels of α-SMA, Smad2, NOX1, and NOX2 were significantly lower in EJSG than in the model group. EBST had a significant regulatory effect on the level of TGF-β1, Smad3, Smad4, NOX4 protein compared with the model group. Compared with the model group, the expression levels of α-SMA, TGF-β1, Smad2, Smad3, P-Smad2/3, Smad4, NOX1, NOX2, and NOX4 protein were restored to normal, after the treatment of EJSG (250 mg kg-1 d-1), EBST (50 mg kg1 d1), and MRHTG (70 mg kg-1 d-1). The protein expression levels of NOX1, NOX2, and NOX4 were not significantly different among the groups.


3.2 | Metabolomics insights
3.2.1 | QC samples analytic results
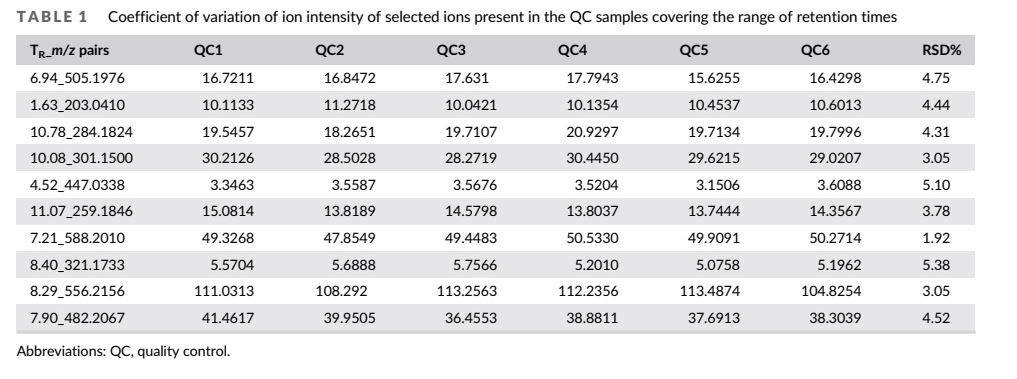
The relative clustering of QC samples (Figure 2A) and relative standard deviations (RSD%) of ion intensity (Table 1) expound and prove the quality of QC data. The trend plot shows the variation of the type's overall observations (Figure 2B). The extracted ion chromatographic peaks of 10 ions were selected for method validation. The repeatability of the method was evaluated by using six replicates of the QC sample. This type of result demonstrated that the method had excellent repeatability and stability.
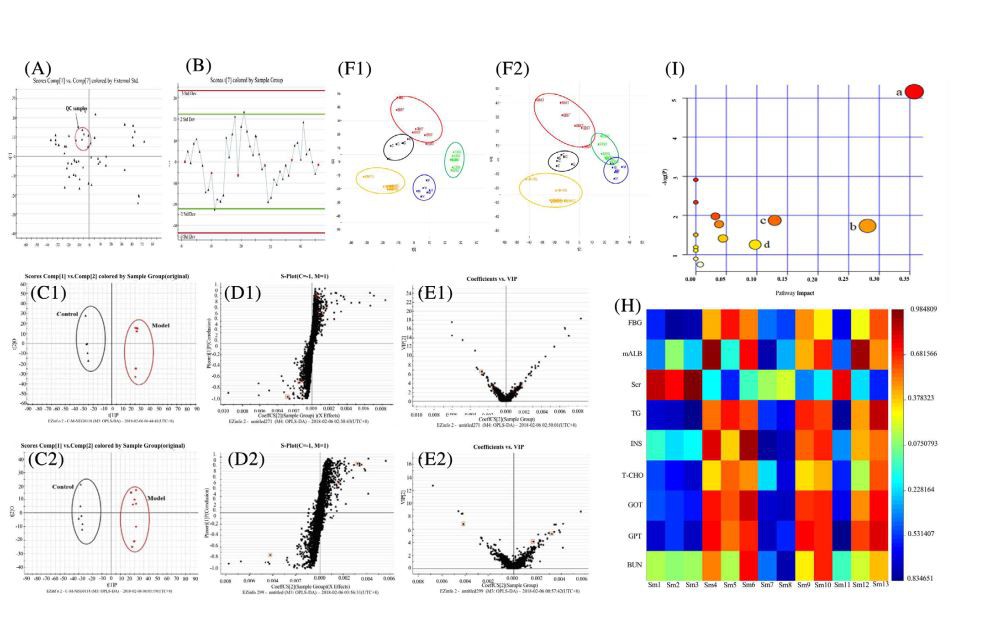
3.2.2 | Thirteen endogenous metabolites were ultimately identified by multivariate data analysis
All the data containing the retention time, peak intensity, and exact mass were imported into the Masslynx™ software for multiple statistical analyses. The supervised OPLS-DA model, a pattern recognition approach, was established to separate serum samples into two blocks between the model group and the control group. The supervised OPLS-DA with 100% sensitivity and no less than 95% specificity using a leave one out algorithm showed better discrimination between the two groups (Figure 2C1, C2), which demonstrated that the DKD model was built successfully. Based on these results, the OPLS-DA score plot (Figure 2D1, D2) and VIP plot (Figure 2E1, E2) was used to look for potential markers associated with DKD progress. The R2Y and Q2 of the PLS-DA model in positive and negative modes for serum samples were suggested that the PLS-DA model was good for fitness and prediction.

The UPLC-QTOF/MS analysis platform provided the retention time and precise molecular mass within measurement errors (<5 ppm) as well as the fragments of the corresponding production for the structural identification of metabolites. According to the precise molecular mass, the predicted elemental composition was predicted and the potential molecular formula could be searched in Human Metabolome Database (http://www.hmdb.ca/). Thirteen endogenous metabolites were ultimately identified by comparing with authentic standards or based on the protocol detailed above method. The information about the detected endogenous metabolites is summarized in Table 2.

3.2.3 | Acteoside regulated the abnormal endogenous metabolites
In order to study the efficacy and action mechanism of MRHTG for treating DKD disease, a PLS-DA model analysis was built to obtain the changes during the control group, model group, and administration group mice (Figure 2F1, F2). The variations of metabolic profiling in serum for administration group mice had the tendency to restore back to the levels of the control group (Table 3). Furthermore, the relative quantities of 13 endogenous metabolites were ultimately identified by comparing with authentic standards or based on the protocol detailed above method. Thirteen endogenous metabolites in serum significantly affected by MRHTG were restored back to the control group. Among them, the adjustment of three treatment groups for cisaconitic acid, corteclone, ceramide (d18:1/12:0), PGH3, was more apparent than the other endogenous metabolites. For the MRHTG group, the cut effect of cis-aconitic acid, ceramide (d18:1/12:0), morphine- 3-glucuronide, N1-(alpha-D-ribosyl)-5, 6-dimethyl-benzimidazole, and PGH3 role was relatively higher, and the most obvious raising effect was on aconitic acid. It also showed that the content of metabolites picked up and the disorder of the metabolic pathway was also improved in the administration group of MRHTG. The detailed information is shown in Figure 2G.

3.2.4 | Correlation analysis
It can be seen from the heat map of correlation analysis in Figure 2H, FBG was negatively correlated with Sm2 and Sm3 (r<-.65); blood lipid levels (T-CHO, TG) and GPT were negatively correlated with Sm3 and Sm11 (r<-.65); Scr was positively correlated with Sm1 and Sm3 (r>- .65); mALB was positively correlated with Sm4 and Sm12 (r >-.65); INS was positively correlated with Sm6 (r >-.65), negatively correlated with Sm7 (r<.65); BUN and Sm8 were negative relevant (r<-.65).
Cistanche treat kidney disease
3.2.5 | Acteoside regulated the disturbed metabolic pathway of lipid metabolism, glyoxylate, and dicarboxylate metabolism, and arachidonic acid metabolism
The metabolic pathway was established by importing the potential metabolites into the web-based database MetPA. The pathway impact value calculated from the pathway to topology analysis with MetPA above 0.1 was screened out as the potential target pathway. Here in Figure 2I, for the four pathways, ether lipid metabolism with the impact-value 0.36; sphingolipid metabolism with the impact-value 0.28; glyoxylate and dicarboxylate metabolism with the impact-value 0.13, and arachidonic acid metabolism with the impact-value 0.1 were filtered out as the most important metabolic pathways.
3.3 | Acteoside inhibited the NADPH/oxidaseTGF-β/Smad signaling pathway on HK-2 cells
3.3.1 | Cell morphology
The HK-2 cells in the control group, model group, osmotic pressure control group, and the 50 μmol/L administration group were cultured for 48 hr, and the cell morphology is shown in Figure 3A. The cells in the control group showed paving stones and the HK-2 cells with high glucose-induced in the model group appeared notable morphological differences compared to the control cells, with an observable loss of the normal tight cell-cell adhesion and cells exhibiting a more elongated phenotype suggestive of a fibroblast-like phenotype. After administration of 50 μmol/L MRHTG, the morphology of the cells was similar to that of the control group.
3.3.2 | Effects of acteoside on the protein expression of TGF-β1, Smad2, Smad3, P-Smad2/3, Smad7, Smad4, α-SMA, NOX1, NOX2, NOX4, NF-κB, and E-cadherin in cells
As shown in Figure 3B, the protein expression levels of NOX1, NOX2, NOX4, α-SMA, NF-κB p65, TGF-β1, Smad2, Smad3, P-Smad2/3, and Smad4 were increased and E-cadherin and Smad7 were decreased significantly in high glucose-induced HK-2 cells (p < .05). The effect of MRHTG is significant, especially in the protein expression of Smad3, Smad7, P-Smad2/3, and positive drug, EBST had a better effect on Smad2, Smad4, and α-SMA. Compared with the model group, the expression levels of NOX1, NOX2, NOX4, α-SMA, NF-κB p65, TGF-β1, Smad2, Smad3, Smad4, Smad7, P-Smad2/3, and E-cadherin were nearly restored to normal, after the treatment of EBST and MRHTG. NOX4, α-SMA, NF-κB p65, TGF-β1, Smad2, Smad3, Smad4, Smad7, P-Smad2/3, and E-cadherin were similar to control group.
3.3.4 | Effects of acteoside on the expression of MCP-1, IL-1β, TNF-α, and IL-6 in cells
Moreover, as shown in Figure 3D, compared with the control group, the expression of MCP-1, IL-1β, TNF-α, and IL-6 were significantly increased (p < .05) in the supernatant of HK-2 cell which was induced by high glucose. The regulation trend of positive drug EBST and MRHTG was consistent. And the MRHTG can significantly regulate the expression of MCP-1, IL-1β, TNF-α, and IL-6.
Cistanche for kidney
4 | DISCUSSION
The db/db diabetic mouse model is an internationally recognized animal model of diabetes and the pathogenesis of DKD in db/db mice is similar to that in humans (Sharma, McCue, & Dunn, 2003). After the administration of MRHTG, the kidney injury index (Scr, BUN, and mALB) levels in db/db mice were significantly improved; blood lipid levels (T-CHO and TG) and blood glucose levels (FBG and INS) were also had varying degrees of improvement in the administration group. In conclusion, acteoside can effectively alleviate the degree of renal interstitial fibrosis and improve abnormal glycolipid metabolism. The regulation of acteoside on INS level may be through inhibiting oxidative stress and inflammatory response of β-cells, regulating the activation of folding protein response, promoting mitochondrial dynamics, and thereby increasing β-cell activity and insulin content.
TGF-β/Smad signaling pathway plays an important role in regulating renal fibrosis in CKD conditions (Trionfini & Benigni, 2017; Wang et al., 2016). Studies have shown that TGF-β1-induced renal fibrosis is mainly achieved through TGF-β1 and Smad-related proteins (Smad2, Smad3, P-Smad2/3, Smad4, and Smad7) (Meng, Nikolic-Paterson, & Lan, 2016). Smad2 and Smad3, two important downstream signaling mediators, are phosphorylated and activated by the TGF-β receptor after its binding to TGF-β. Phosphorylated Smad2/3 bind to Smad4 proteins, form heterooligomeric complexes, and translocate into the nuclei, inducing the transcription of fibrotic genes. The activation of Smad2 and Smad3 is observed in obstructed mouse kidneys (Wang et al., 2013; Wang et al., 2014), type 1 diabetes patients, and diabetic
mice (Wang, Lin, Ren, Liu, & Yang, 2015) Wei et al., 2010; (Wolf & Ziyadeh, 1999), as illustrated by the increase in phosphorylated Smad2 (p-Smad2) and phosphorylated Smad3 (p-Smad3) expression. Matrix metalloproteinase-mediated E-cadherin disruption directly leads to renal tubular epithelial cell EMT through Slug (Zheng et al., 2009). The loss of E-cadherin expression is a hallmark of EMT. As a downstream signal protein of the TGF-β1 family, SMAD has an important role in the process of α-SMA mediated renal interstitial fibrosis. The results of western blotting showed the expression of TGF-β1 and its signal transduction proteins α-SMA, Smad2, Smad3, Smad4, and P-Smad2/3 protein in early kidney tissues decreased significantly, especially after the administration of MRHTG. Similar results were obtained in the HK-2 cell experiment. Therefore, MRHTG may play a role in improving early kidney injury via inhibiting of TGF-β/Smad signaling pathway.
It has been reported that elevated levels of reactive oxygen species (ROS) in cells are an important factor leading to tubulointerstitial fibrosis and renal fibrosis. High glucose-induced ROS are mainly derived from renal epithelial cells NADPH oxidase (Chen, Chen, & Harris, 2012). The prototype NADPH oxidase identified in phagocytic cells consists of two membrane-bound subunits, NOX1, NOX2, and NOX4 are detected in kidney cells, with NOX4 being the most abundant (Sedeek et al., 2010). In addition, increased ROS is associated with renal fibrosis; promotes the production of collagen, fibronectin, and α-SMA (Kim, Seok, Jung, & Park, 2009); and plays a key role in inflammation through the NF-κB pathway. The in vitro study by HK-2 cells found that a high glucose-induced increase in the expression of NOX1, NOX2, NOX4, α-SMA, NF-κB p65 protein, and mRNA, and inflammatory factor MCP-1, IL-1β, TNF-α, and IL-6 were significantly increased (p < .05). The results showed the expression of NOX1, NOX2, NOX4 protein in high glucose-induced HK-2 cells, especially after the administration of MRHTG. And the MRHTG can significantly regulate the inflammatory factor expression of MCP-1, IL-1β, TNF-α, and IL-6. Similar results were obtained in the db/db mice experiment. However, there was no significant difference in NOX1 and NOX2 in the db/db mice compared with the normal group, and the NOX4 difference was significant. High glucose increased the expression of Nox4, but not other Noxes. Some findings, which were similarly demonstrated in isolated proximal tubules from db/db mice, suggest that upregulation of Nox4-based NADPH oxidase may be counter-regulated by downregulation of Nox2/NADPH oxidase (Sedeek et al., 2010). Therefore, MRHTG may play a role in improving DKD via inhibiting of NADPH/oxidase signaling pathway.
Notably, there are several distinct metabolic pathways in various stages of the progress of DKD.
Ceramide (d18:1/12:0) has been found and used to explain sphingolipid metabolisms, and ceramide plays an important role in vascular endothelial function. Ceramide may cause NAPDH oxidase subtype increased expression of NOX4, and then reduce intracellular NO activity, which in turn causes endothelial dysfunction. It is suggested that the NADPH/oxidase signal pathway is related to ceramide. Increased ceramide synthase activity is responsible for the increased ceramide generation, leading to an apoptotic change of the renal epithelial cells (Itoh et al., 2006; Yi, Zhang, Janscha, Li, & Zou, 2004) In addition, studies of insulin-sensitive cells have shown that ceramide and its derivatives (eg, ganglioside GM3 and sphingosine, etc.) can antagonize insulin signaling pathways, induce oxidative stress, inhibit glucose uptake and storage, and may trigger many cause insulin resistance (Summers & Nelson, 2005), but the relationship between ceramide and insulin resistance in vivo has been controversial (Gorska, Dobrzy n, Zendzian-Piotrowska, & Gorski; Holland et al., 2007; Straczkowski et al., 2004). In DKD, the content of ceramide increased significantly compared to normal mice and recovered to normal after administration of acteoside. PC (18:1[9Z] e/2:0) and LysoPC (O-18:0) have been found and used to explain the ether lipid metabolism. PC (18:1[9Z] e/2:0) is an intermediate link in the metabolism of ether lipids and is an ether lipid with a platelet-activating factor function. Ether lipid metabolism and sphingolipid metabolism are lipid metabolism pathways, which is consistent with the results of biochemical markers of lipid metabolism. In addition, cis-aconitic acid can be used to explain glyoxylate and dicarboxylate metabolism, which is related to the citric acid metabolic pathway (Serrano & Bonete, 2001). The chronic inflammatory response plays a key role in diabetic nephropathy (Navarro-Gonzalez, Mora-Fern andez, De Fuentes, & GarcíaPérez, 2011). ROS and inflammatory cytokines promote the activation of phospholipases, and phospholipase A2 (PLA2) stimulates the release of arachidonic acid (Gijon, Spencer, Siddiqi, Bonventre, & Leslie, 2000; Shin & Kim, 2009), arachidonic acid can be further metabolized into prostaglandins, so the up-regulation of prostaglandin H3 (PGH3) can be used explain the role of arachidonic acid metabolism in the inflammatory response of diabetic nephropathy. TGF-β/Smad signaling pathway was involved in fibrosis development, leading to inflammation response, together with arachidonic acid metabolism to advance diabetic nephropathy. Results suggested that these target pathways showed marked perturbations over the time-course of the treatment and could contribute to the development of DKD.
In conclusion, acteoside can improve db/db mice's biochemical indicators, such as BUN, FBG, GOT, GPT, INS, T-CHO, TG, Scr, and mALB. Metabolomics results showed that diabetic nephropathy may act on these four metabolic pathways: ether lipid metabolism, sphingolipid metabolism, glyoxylic acid, dicarboxylic acid metabolism, and arachidonic acid metabolism. And acteoside may regulate glycolipid level of db/db mice as well as NADPH/oxidase-TGF-β/Smad signaling pathway regulation to improve early kidney damage caused by diabetes. This study paved the way for further exploration of pathogenesis, early diagnosis, and the development of a new therapeutic agent for DKD.
Cistanche for improving kidney function
ACKNOWLEDGEMENT
This work was supported by the National Natural Science Foundation of China (No. 81373889, 81373889; 81673533), and Priority Academic Program Development of Jiangsu Higher Education Institutions (ysxk-2014).
CONFLICTS OF INTEREST
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
AUTHOR CONTRIBUTIONS
Qinwen Wang: Conceptualization, Writing—Original Draft. Xinxin Dai: Data Curation. Xiang Xiang, Project Administration. Zhuo Xu: Investigation. Shulan Su: Supervision, Methodology. Dandan Wei: Resources. Tianyao Zheng: Software, Vali dation. Er-Xin Shang: Visualization. Dawei Qian: Visualization. Jin-ao Duan: Methodology, Supervision. All authors read and approved the manuscript.
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Excerpted from: ' A natural product of acteoside ameliorate kidney injury in diabetes db/db mice and HK-2 cells via regulating NADPH/ oxidase-TGF-β/Smad signaling pathway' -----by Qinwen Wang | Xinxin Dai et.al
----Phytotherapy Research. 2021;35:5227–5240. wileyonlinelibrary.com/journal/ptr © 2021 John Wiley & Sons Ltd.