Combining Microbubble Contrast Agent With Pulsed-Laser Irradiation For Transdermal Drug Delivery Part1
Apr 03, 2023
Abstract: The optodynamic process of laser-induced microbubble (MB) cavitation in liquids is utilized in various medical applications. However, how incident laser radiation interacts with MBs as an ultrasound contrast agent is rarely estimated when the liquid already contains stable MBs. The present study investigated the efficiency of the laser-mediated cavitation of albumin-shelled MBs in enhancing transdermal drug delivery. Different types and conditions of laser-mediated inertial cavitation of MBs were first evaluated. A CO2 fractional pulsed laser was selected for combining with MBs in the in vitro and in vivo experiments. The in vitro skin penetration by β-arbutin after 2 h was 2 times greater in the group combining a laser with MBs than in the control group. In small-animal experiments, the whitening effect on the skin of C57BL/6J mice in the group combining a laser with MBs on the skin plus penetrating β-arbutin increased (significantly) by 48.0% at day 11 and 50.0% at day 14, and then tended to stabilize for the remainder of the 20-day experimental period. The present results indicate that combining a CO2 laser with albumin-shelled MBs can increase skin permeability to enhance the delivery of β-arbutin to inhibit melanogenesis in mice without damaging the skin.
According to relevant studies,cistanche is a common herb known as "the miracle herb that prolongs life". Its main component is cistanoside, which has various effects such as antioxidant, anti-inflammatory, and immune function promotion. The mechanism between cistanche and skin whitening lies in the antioxidant effect of cistanche glycosides. Melanin in human skin is produced by the oxidation of tyrosine catalyzed by tyrosinase. The oxidation reaction requires the participation of oxygen, so the oxygen-free radicals in the body become an important factor affecting melanin production. Cistanche contains cistanoside, which is an antioxidant and can reduce the generation of free radicals in the body, thus inhibiting melanin production.

Click on Cistanches Herba For Whitening
for more info:
david.deng@wecistanche.com WhatApp:86 13632399501
1. Introduction
Cavitation refers to the formation of cavities in a liquid and usually occurs when the liquid is subjected to rapid pressure changes. Such pressure changes can be induced using many different methods, with acoustic cavitation being initiated when the amplitude of an applied acoustic pressure exceeds a certain threshold [1]. Acoustic cavitation involves the formation, growth, pulsation, and collapse of microbubbles (MBs) in liquids during sonication by high-intensity ultrasound (US) waves. These phenomena are believed to be responsible for the mixing, fragmentation, erosion, wetting, sonocapillary, and other effects that have various practical industrial applications [1].
US-induced cavitation of MB contrast agents also plays an important role in both diagnostic and therapeutic medical applications. US contrast agents are stabilized and coated MBs that are injected intravascularly to enhance the resolution of diagnostic US imaging [2]. There is evidence from numerous studies that the presence of MB contrast agents in the blood can decrease the threshold for various US-induced biological effects both in vitro and in vivo, such as hemolysis, capillary rupture, and sonoporation [3]. Some studies have indicated that the presence of MB contrast agents in the blood significantly reduces the threshold for US-induced premature cardiac contractions [4,5]. The resonance of MBs (stable cavitation) results in nonlinear harmonic emissions that can be utilized in MB-specific contrast imaging. The inertial cavitation and destruction of MBs can induce strong mechanical stresses that increase the permeabilities of cell membranes and the blood-brain barrier for improving the delivery of therapeutic agents. In our previous studies, we have applied inertial cavitation of MBs induced by the US to enhance transdermal drug delivery (TDD), since inertial cavitation was found to result in far greater permeability enhancement of the stratum corneum compared to stable cavitation [6–8].
Laser irradiation is an alternative approach for enhancing drug permeation and hence facilitating drug delivery into or across the skin. When a laser pulse with an intensity above a certain threshold is focused on a liquid, explosive vaporization of the liquid also can induce MB cavitation [9,10]. The aggressive nature of laser-induced cavitation has resulted in it being utilized in a broad range of applications including cell lysis, cell membrane poration, and ocular surgery [11]. Strategies for safely improving the appearance of postoperative, atrophic, and acne scars have recently been demonstrated using fractional ablative and nonablative lasers [12]. Ablative laser skin resurfacing provides the greatest clinical improvements, but the postoperative recovery takes several weeks [13]. Nonablative laser procedures might be more suitable for patients unable or unwilling to tolerate extended postoperative healing. However, prior herpes simplex infection can reactivate after nonablative laser skin remodeling due to the intense heat produced by the laser or other light source [13].

Some methods have been developed to reduce the heat produced by laser irradiation, such as the use of contact cooling handpieces or dynamic cryogenic devices capable of delivering spurts of a cooling spray of variable durations [14]. However, there is still no consensus about which method of cooling is most effective during treatment. Moreover, unlike the US, the mechanism underlying the effects of laser-induced cavitation with stabilized coated MBs in liquids remains unclear.
2. Materials and Methods
2.1. Production of Albumin-Shelled MBs
Albumin-shelled MBs were prepared according to the procedure used in our previous studies [7,15]. In brief, albumin-shelled MBs were generated by sonication in 10 mL of a solution containing 140 mg of albumin (Octapharma, Vienna, Austria) and perfluorocarbon gas in physiological saline (pH 7.4, 0.9% sodium chloride) using a sonicator (Branson Ultrasonics, Danbury, CT, USA) for 2 min. The number of perflfluorocarbon-fifilled albumin MBs in the solution was measured using the MultiSizer III device (Beckman Coulter, Fullerton, CA, USA) with a 30-µm-aperture probe and measurement boundaries of 0.6–20 µm. The size distribution in the suspension was measured based on dynamic light scattering (Zetasizer Nano, ZS90, Malvern, UK), which revealed that the albumin-shelled MBs had a diameter of 1.02 ± 0.11 µm (mean ± SD) and a concentration of 1.40 × 108 MBs/mL.
2.2. Laser-Induced MB Disruption
Previous studies have suggested that US-mediated MB disruption (i.e., inertial cavitation) is required for effective TDD [8,16,17]. The present study measured the efficiency of MB disruption when using different types of lasers under different conditions. The concentration of MBs was adjusted to 2.8 × 107 MBs/mL (fivefold dilution) and 1.4 × 107 MBs/mL (tenfold dilution) in an Eppendorf tube, and irradiation was provided by four types of laser: Air-cooled argon-ion laser (515 nm, continuous wave), supercontinuum fiber laser (1064 nm, pulsed wave), Nd: YAG laser (532 nm, pulsed wave), and CO2 fractional laser (10,600 nm, pulsed wave). The detailed conditions for the various types of lasers are listed in Table 1.

An example of the optical setup is shown in Figure 1. The change in temperature during laser irradiation was measured using a thermometer (Optris LS, Optris, Berlin, Germany). Then, after exposure to the laser, 100 µL of the MB solution was added to a microscope slide for observation. The numbers of MBs in light-microscopy images before and after laser irradiation were converted into 8-bit grayscale images using MATLAB (The MathWorks, Natick, MA, USA) to facilitate the observations of MB disruption. The destruction rate of MBs was quantified according to the damaged areas using the following equation:
![]()

2.3. Penetration Depth in Pigskin
2.4. In Vitro Skin Penetration by β-arbutin Solution
A 2-mm-thick sample of pigskin was harvested using a Humby knife, carefully cleaned with PBS, and cut into square pieces (2 cm × 2 cm). Circular areas on the skin samples with a radius of 1.5 cm and a height of 5 mm were encircled with gel to prevent leakage when the sample was loaded with 500 µL of MBs. After irradiating the sample seven times (the conditions are listed in Table 1) with a CO2 fractional laser, skin penetration was tested using static Franz diffusion cells over an area of 2.14 cm2 according to the experimental design that we have described previously [7]. The temperature of the diffusion assembly was maintained at 37 ◦C. β-arbutin (30 mg/mL, 500 µL, 4-hydroxyphenyl-β-D-glucopyranoside, molecular mass = 272.25 Da; Sigma-Aldrich, St. Louis, MO, USA) was applied to the epidermal side of the skin and occluded with parafilm (Pechiney Laboratory Safety Products and Apparel, Chicago, IL, USA). The receptor diffusion compartment facing the dermal side was filled with 12 mL of PBS (pH 7.4), which was stirred by a magnetic bar rotating at 600 rpm. Test solutions that did not contain MBs were filtered through a 0.2-µm micropore filter (Nalgene, Rochester, NY, USA) or a 0.22-µm micropore filter (Millex, Darmstadt, Germany). Aliquots (200 µL) of the receptor solution was taken at 0, 1, 2, 3, 4, 6, 8, 10, 12, and 24 h, and replaced by the same volume of fresh receptor solution.

2.5. HPLC Analysis of β-arbutin
An Inspire™ C18 column (250 mm × 4.6 mm, 5 µm particle size; Dikma Technologies, Lake Forest, CA, USA) was used to measure the β-arbutin concentrations. The HPLC system was equipped with a binary pump (PU-2089, Jasco, Tokyo, Japan), and the wavelength of the ultraviolet (UV) detector (UV-2075, Jasco) was set at 280 nm. The mobile phase consisted of methanol: distilled water (pH 5.5, 70:30 v/v) [18] at a fellow rate of 0.6 mL/min. All samples to be analyzed were injected at a volume of 20 µL. The retention time of β-arbutin was about 4.3 min.
2.6. Animal Treatments
The melanin content of organoids was investigated in the C57BL/6J mouse model [19]. Five-week-old mice weighing 20–25 g were obtained from Bio Lasco (Taipei, Taiwan). The experimental protocol was approved by the Institutional Animal Care and Use Committee of the National Defense Medical Center, Taipei, Taiwan. The procedures for animal care complied with institutional guidelines and regulations (approval no. IACUC-17-092). Throughout the experiments, the animals were housed in stainless-steel cages in an air-conditioned room with the temperature maintained at 25–28 ◦C and with alternating light and dark cycles of 12 h each.
The animals were acclimatized for 7 days before the experiment. After their hair had been removed from an area of 2 cm × 2 cm, the skin color was measured using a chroma meter (CR-400, Konica Minolta Sensing, Tokyo, Japan). The animals were then exposed to ultraviolet B (UVB) radiation (G8T5E, Sankyo, Tokyo, Japan) to induce hyperpigmentation (total energy dose per exposure = 1 J/cm2, wavelength = 306 nm, three times per week for 2 weeks), and then the skin color was measured again.
The animals were divided into the following five groups (n = 5 per group, treatment applied once every 3 days for 20 days): (1) no treatment (group C); (2) application of penetrating β-arbutin alone (300 µg/mL, 0.2 mL/cm2 ) (group A); (3) laser irradiating the skin directly with the application of penetrating β-arbutin (300 µg/mL, 0.2 mL/cm2 ) (group L + A); (4) laser irradiating the skin covered by saline and with the application of penetrating β-arbutin (300 µg/mL, 0.2 mL/cm2 ) (group L + S + A); and (5) laser irradiating the skin combined with MBs on the skin and with the application of penetrating β-arbutin (300 µg/mL, 0.2 mL/cm2 ) (group L + MBs + A). The change in skin color induced by each of the treatments was assessed at predetermined times using the chroma meter. The luminosity index, L [20], was calculated on each measurement day before and after treatment.
2.7. Histological Study
Skin tissue samples (approximately 8 mm × 8 mm) were taken from the treatment area immediately after the experiments and stored in a 10% formalin solution. Hematoxylin and eosin (HE; Sigma-Aldrich) staining was applied, and the samples were analyzed by an expert dermatopathologist (H.W.G.). Some other samples were stained with Fontana-Masson silver nitrate (Kojima Chemical, Kashiwabara, Japan) for 30 min at 60 ◦C, and then washed with distilled water and fixed in 5% sodium thiosulfate solution (Duksan, Seoul, Korea) for 2 min, before washing again with distilled water. The samples were then stained with nuclear fast red solution (Fluka, Buchs, Switzerland) for 5 min and washed twice with distilled water. Finally, the samples were dehydrated in 95% followed by 100% ethanol and then washed twice with xylene (Duksan) [21].
2.8. Statistical Analysis
The obtained data were analyzed statistically using Student’s t-test. A probability value of p < 0.05 was considered indicative of a signifificant difference.

3. Results
3.1. Laser-Induced MB Disruption
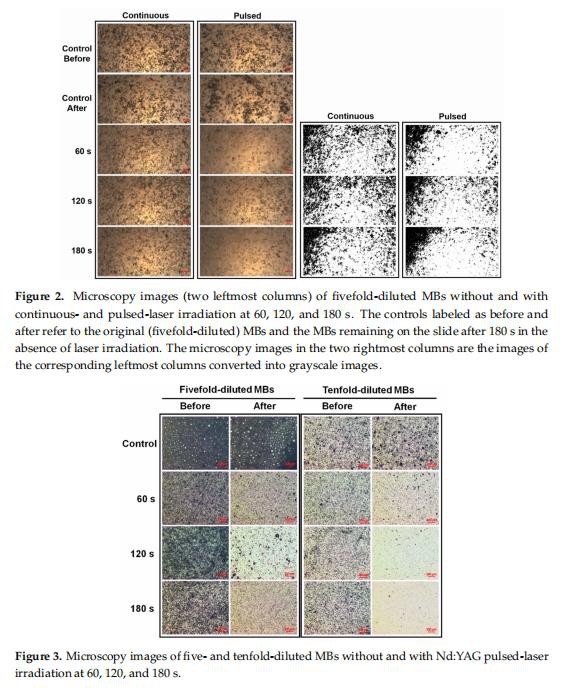
Microscopy images of fivefold-diluted MBs without and with irradiation by a 10.8 mW continuous (air-cooled argon-ion) laser and a 10.8 mW pulsed (supercontinuum fiber) laser for 60, 120, and 180 s are shown in Figure 2. The destruction rates for the pulsed laser increased by 17.66%, 20.52%, and 39.05% compared to the continuous laser at 60, 120, and 180 s, respectively. Figure 3 shows the destruction efficacy of five- and tenfold-diluted MBs when using an Nd: YAG pulsed laser at 60, 120, and 180 s. The destruction rates of five- and tenfold-diluted MBs were 72.46% and 78.59%, respectively, at 60 s, 88.06%, 96.10% at 120 s, 85.22%, and 98.80% at 180 s. Figure 4 shows the destruction efficacy of tenfold-diluted MBs for being irradiated one, three, and seven times by the clinical CO2 fractional pulsed laser. The destruction rate increased with the irradiation time, being close to 100% for seven times irradiation, and so this condition was used in the subsequent in vitro and in vivo experiments.

for more info: david.deng@wecistanche.com WhatApp:86 13632399501






