Farnesyltransferase Inhibitor LNK-754 Attenuates Axonal Dystrophy And Reduces Amyloid Pathology in Mice Part 4
Sep 26, 2023
LNK‑754 and lonafarnib enhance axonal endolysosomal trafficking in cultured neurons
To investigate the mechanism of LNK-754 to decrease the accumulation of LAMP1, BACE1, and LT in dystrophic neurites in 5XFAD mice, we performed in vitro studies using wild-type mouse primary cultured neurons. We first evaluated the distribution pattern of endolysosomal organelles in neurons treated with vehicle, LNK-754, or lonafarnib by confocal immunofluorescence microscopy. In vehicle-treated neurons, LAMP1 fluorescent vesicles were concentrated mostly in cell bodies and proximal neurites, whereas in LNK-754 and lonafarnib-treated neurons, LAMP1 vesicles were increased in proximal and distal neurite areas compared to vehicle-treated neurons (Fig. 6A-F).
Similar to our in vivo results and in agreement with previous reports [9], increased total LAMP1 was also present in LNK-754 and lonafarnib-treated cultures (Fig. 6F-H). Lonafarnib treatment significantly increased total LAMP1 and LAMP1 localizing to neurites, whereas a trend was observed for LNK-754 treatment (Fig.6D, F). LAMP1 non-specifically marks various endosomal and autophagic vesicles, so we next analyzed the localization and acidification of late endosomes and lysosomes in live neuron cultures treated with LysoSensor Green, a fluorescent dye that exhibits a pH-dependent increase in fluorescence intensity in acidifed compartments.
Neurite is one of the important functional structures in nerve cells, which can transmit nerve signals and communicate with other cells. In neurites, LAMP1 is an important membrane protein that participates in the transport process of the intracellular membrane system and participates in autophagy and vesicle formation. In addition, research shows that LAMP1 is also closely related to immunity.
Some studies have found that LAMP1 plays an important role in diseases such as tumors, infections, and inflammation. The expression of LAMP1 is closely related to tumor invasion, metastasis, and prognosis. At the same time, LAMP1 can also participate in the body's immune response. Researchers found that LAMP1 can promote the activation of T cells and natural killer cells and enhance their killing ability. In addition, LAMP1 can also promote the body's clearance of pathogens and improve immune response.
In addition, LAMP1 is also related to the occurrence of some autoimmune diseases. For example, the expression level of LAMP1 is significantly increased in autoimmune diseases such as systemic lupus erythematosus. In addition, researchers also found that some patients with autoimmune diseases also have higher levels of anti-LAMP1 antibodies, indicating that LAMP1 may be related to the occurrence of autoimmune reactions.
In summary, the role of LAMP1 in neurites is not limited to the function of the nervous system but also includes the body's immune response. Although research related to LAMP1 is still in its infancy, it may become a new target for preventing and treating some diseases in the future. Further research on the relationship between LAMP1 and immunity will provide new ideas and opportunities for the development of new drugs. It can be seen that we need to improve our memory. Cistanche deserticola can significantly improve memory, because Cistanche deserticola can also regulate the balance of neurotransmitters, such as increasing the levels of acetylcholine and growth factors. These substances are very important for memory and learning. In addition, meat can also improve blood flow and promote oxygen delivery, which can ensure that the brain receives sufficient nutrients and energy, thus improving brain vitality and endurance.

Click Know to improve short-term memory
Total LysoSensor Green fluorescence intensity was unchanged in neurons treated with vehicle, LNK-754, or lonafarnib (Fig. 6I, J). Consistent with our findings for LAMP1, the localization of LysoSensor vesicles increased in distal neurites of LNK-754 or lonafarnib-treated cultures (Fig. 6I, arrows).
A considerable percentage of LAMP1-positive vesicles were localized to proximal and distal neurites in FTI-treated neurons, suggesting that FTase inhibition affects the motility of endolysosomal organelles. We assessed the effects of FTI treatment on the dynamics of late endosomes and lysosomes by time-lapse imaging of live wild-type primary neuronal cultures stained with LT (Fig. 7A).
Kymograph analysis of axonal LT vesicles revealed that, under all treatment conditions, most axonal LT vesicles were stationary, or traveled in a low velocity, retrograde direction (Fig. 7B). However, there was a strong shift in the percentage of stationary, retrograde, and anterograde axonal LT vesicles in LNK-754 and lonafarnib treated neurons. The percentage of retrograde LT vesicles in LNK-754 (36%) or lonafarnib (36%) treated neurons was significantly higher than in vehicle-treated neurons (19%) while the percentage of stationary LT vesicles in LNK-754 (62%) or lonafarnib (60%) treated neurons was lower compared to vehicle-treated neurons (74%) (Fig. 7C).
A significant increase in the number of LT vesicles per axon length was observed in lonafarnib-treated neurons and a trend was observed for LNK-754 treatment (Fig. 7D), as in the distribution of LAMP1 (Fig. 6A-D) and LysoSensor (Fig. 6I, J). The distance traveled by LT vesicles was significantly increased in LNK-754 and lonafarnib-treated neurons (Fig. 7E, G) and the average velocity of LT vesicles was significantly faster in LNK-754 (0.17μm/s) and lonafarnib (0.18μm/s) treated neurons than in vehicle (0.11μm/s) treated neurons (Fig. 7F, H; movie S1, S2, S3). Together with our results from FTI-treated 5XFAD mice, these data support the role of FTase in regulating both the generation and axonal motility of late endosomes and lysosomes.
Discussion
Many neurodegenerative diseases are characterized by impaired lysosomal systems and the buildup of disease-specific protein aggregates. AD brains in particular show extensive lysosome dysfunction and can be distinguished from other neurodegenerative diseases by the accumulation of autophagic and endolysosomal organelles in enlarged dystrophic neurites surrounding amyloid plaques [54].
FTIs are a promising therapeutic candidate for repurposing towards treating neurodegenerative diseases that share the pathologies of lysosome dysfunction and protein accumulation. By increasing lysosomal function, FTIs could have significant therapeutic effects on dystrophic neurites in the AD brain. However, until now, the impact of FTIs on Aβ pathology and axonal dystrophy has yet to be tested in vivo.

We determined the effect of brain penetrant FTIs LNK754 and lonafarnib on AD-relevant pathology in the 5XFAD mouse model and present new data showing that chronic treatment of 5XFAD mice with LNK-754 reduced amyloid plaque burden and attenuated dystrophic neurite formation. While not a direct focus of this study, LNK-754 and lonafarnib treatment also decreased tau hyperphosphorylation in the brains of 5XFAD mice. In contrast to our findings for LNK-754, lonafarnib-treated 5XFAD mice showed only slight improvements in ADrelevant pathology, and consistently, markers of FTase inhibition were mostly unchanged in the brains of lonafarnib mice.
LNK-754 and lonafarnib were both effective in vitro; the number and velocity of axonal endolysosomal organelles trafficking in a retrograde direction were dramatically increased in live cultured primary neurons. LNK-754 and lonafarnib also increased levels of endolysosomal marker LAMP1 in primary neuron cultures and the brains of 5XFAD mice. Tus, FTI treatment stimulated endolysosomal biogenesis and retrograde axonal trafficking of late endosomes and lysosomes in vitro, which may have slowed axon degeneration and inhibited dystrophic neurite formation and plaque deposition in 5XFAD mice. Notably, LNK-754 reduced BACE1 accumulation in dystrophic neurites and blocked the feed-forward pathway of axonal dystrophy, Aβ42 generation, and plaque deposition. Our results agree with previous reports pointing to FTase as a therapeutic target for treating proteinopathies and suggest that FTIs target dystrophic neurites by enhancing lysosome function and promoting endolysosome trafficking.
We compared brain-penetrant FTIs LNK-754 and lonafarnib because both are known to activate lysosomes and autophagy in mouse models of proteinopathies and have been investigated clinically for cancer treatment [8, 9, 41, 55–57]. Lonafarnib was of particular interest because it markedly reduces tau inclusions [9], is FDA-approved for the treatment of progeria, and has a known safety and adverse event profile in humans [57]. Nonetheless, we found that LNK-754 was considerably more efficacious than lonafarnib in improving the AD pathology of the 5XFAD mouse.
Evidence that both LNK-754 and lonafarnib cross the blood-brain barrier has come from human clinical cancer trials [41, 57] but a comparison of LNK-754 and lonafarnib blood-brain permeability in 5XFAD mice has yet to be performed, so it is conceivable that lonafarnib did not cross the blood-brain barrier as efficiently as LNK-754. The discrepancy between LNK-754 and lonafarnib could also be due to differences in potency of LNK-754 and lonafarnib, as the IC50 for H-Ras inhibition is 0.57nM for LNK-754 [41] and 1.9nM for lonafarnib [51]. We readily detected changes in known markers of FTase inhibition in the brains of LNK754-treated 5XFAD mice, but not in lonafarnib mice, supporting the idea that a higher dosage of lonafarnib may be required to achieve a therapeutic effect.
High doses of FTIs are necessary to maximally suppress farnesylation of Ras as well as other canonical CAAX FTase substrate proteins. A low-dose (1mg/kg/day) treatment regimen was followed in this study because evidence indicates that low doses of FTIs can activate non-canonical protein substrates that are involved in protein clearance pathways [8, 28]. Furthermore, low-dosage treatment in patients can avoid side effects typically seen with high levels of Ras inhibition. LNK-754 treatment (0.9mg/kg/day) reduced α-syn in the brains of PD transgenic mice and elevated lysosomal activity above baseline levels in wild-type mice [8]. Conversely, intermittent treatment of lonafarnib by oral gavage at a dose that was substantially higher (80mg/kg/day) was necessary to inhibit tau aggregation [9].
A higher dose of lonafarnib may be required to enhance both endolysosomal function and axonal trafficking since we found elevated levels of LAMP1 in the brains of lonafarnib-treated mice but minimal effects on dystrophies and plaque burden. We tested only a single dosage of FTIs, and further studies will be required to determine the dosage and route of administration required to achieve optimal reduction of Aβ and dystrophic neurite pathologies. The in vitro effects of LNK-754 and lonafarnib were consistent, suggesting that lonafarnib would generate similar phenotypes in 5XFAD mice if comparable inhibitor activity on FTase was achieved.
It is possible that drug exposure in cell cultures was more similar for the two compounds, as compared to the in vivo model.
If comparable FTase inhibition occurred in vivo, another possibility is that LNK-754 and lonafarnib altered the farnesylated proteome in the 5XFAD brain in distinct ways. This has been observed in leukemia cell lines, where clear differences in farnesylated proteins were reported following treatment with the FTIs BMS241,662 and L-778,123 [24]. Additionally, this study demonstrated unique patterns of farnesylation in different cell lines, indicating that a specific farnesylated proteome could exist in each cell type, which could exacerbate small differences in protein farnesylation inhibition between LNK-754 and lonafarnib. Therefore, it is possible that farnesylated proteins specifically inhibited by LNK754, but not lonafarnib, are responsible for mediating plaque and dystrophy reduction.

An alternative possibility, although unlikely [58], is that of-target inhibition of geranyl-geranyl transferase (GGTase) by LNK-754 or lonafarnib could have resulted in differential inhibition of substrates regulated by GGTase. Finally, prenylation pathways for substrates involved in plaque reduction could be activated in lonafarnib-treated mice but not LNK-754 mice. It is likely that the simultaneous inhibition of multiple substrates by LNK-754 led to plaque reduction in 5XFAD mice and some or all of these substrates were not inhibited in lonafarnib mice. To identify mechanisms of farnesylated substrates in regulating plaque growth and seeding, future experiments should be performed to identify which substrates are most reduced in hippocampal and cortical brain regions, and correlate with decreased plaque burden and dystrophies in the brains of LNK-754 and lonafarnib treated 5XFAD mice.

A trend was observed throughout our chronic study where significant differences were found between vehicle and LNK-754 in the cortex and between lonafarnib and LNK-754 in the hippocampus. We observed significant decreases in plaques and dystrophy cortical brain regions between vehicle and LNK-754 treated mice, while significant differences between LNK-754 and lonafarnib were found in the hippocampus. Amyloid pathology begins in layer 5 cortical neurons and is most severe in this brain region [36] so we suspect this was due to the low number of plaques in the hippocampus and variability in plaque burden in 5XFAD mice. We expect that our results would reach statistical significance between LNK754 and vehicle for all outcomes by increasing treatment length or assessing the mice at an advanced age when more plaques would be found in the hippocampus. However, distinct mechanisms may occur between the hippocampus and cortex in LNK-754-treated mice. For example, differences in physiological levels of FTase in cells concentrated in the hippocampus compared to the cortex, or differential expression of FTase substrates may exist between the hippocampus and cortex.
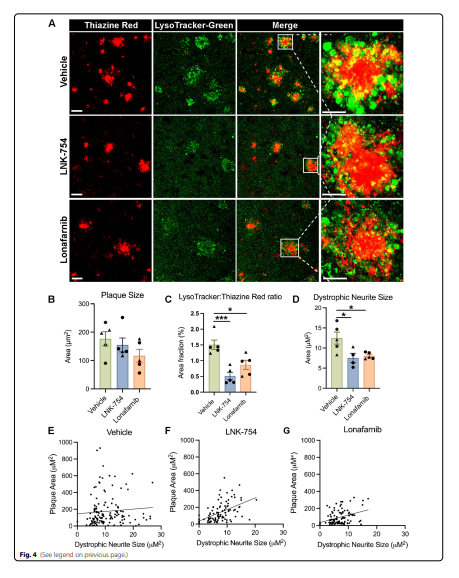
To investigate the mechanism of LNK-754 to reduce amyloid pathology in 5XFAD mice, we focused our study specifically on dystrophic neurites. Dystrophic neurites contain high levels of LAMP1 and BACE1 and significantly contribute to extracellular plaque deposition and axonal dystrophy, resulting in synaptic loss, neurodegeneration, and cognitive deficits [14, 16, 18]. By assessing multiple markers of dystrophic neurites, including LAMP1, BACE1, and LT, we found that equally sized plaques have fewer dystrophies in LNK-754 mice compared to vehicle or lonafarnib-treated mice. A moderate reduction in the ratio of LAMP1:Aβ42 per plaque was observed compared to the greater decreases of other dystrophic neurite markers in LNK-754 treated mice, possibly because a portion of plaque-associated LAMP1 is present in microglia [59].
LT accumulated in dystrophies around plaques in living mouse brain tissues (Fig. 4A), showing that highly acidifed late endosomes and lysosomes are present in dystrophic neurites. Furthermore, we showed that LNK-754 treatment had a dramatic effect on decreasing the ratio of LT: TR, compared to the ratio of LAMP1:Aβ42. Based on our findings with LT staining, we hypothesize that the subset of LAMP1-positive vesicles that were reduced in LNK-754 chronically treated mice were acidifed late endosomes and lysosomes.
Most importantly, LNK-754 prevented the abnormal accumulation of BACE1 in dystrophies. Inhibiting BACE1 activity in dystrophic neurites in the early stages of Aβ deposition suppresses the formation of new plaques, rather than the growth of the original plaque [18], so a similar mechanism could occur in LNK-754 treated mice. In the APP/PS1 mouse model, dystrophic neurites plateau after plaques reach a radius of 10μm, which may be explained by complete axon loss balanced with dystrophic neurite generation [18]. The positive correlations between LAMP1 and plaque burden after chronic LNK-754 treatment, and LT and plaque size after acute LNK-754 treatment, could be caused by a shift in the equilibrium of dystrophy loss and generation, an increase in the number or size of viable axons and neurons surrounding plaques, or a decrease in the abundance of large plaques.
A key finding of our study was that LNK-754 and lonafarnib dramatically enhanced retrograde axonal trafficking of endolysosomal compartments. It has been reported that FTIs, including lonafarnib, increase MT acetylation and stability in cancer cell lines [31, 32, 60]. Therefore the enhanced trafficking of of endolysosomal compartments in our study was potentially mediated through substrates responsible for the MT-stabilizing effect of FTIs. Interestingly, a synergistic effect between traditional MT stabilizing agents and FTIs has been well-documented in cancer settings [32, 33, 61], which raises the intriguing possibility that a similar treatment could prevent dystrophic neurite formation in AD brains. The additive effects of enhancing the degradative capacity of lysosomes and trafficking of these compartments likely promoted the targeting of BACE1 to mature lysosomes for degradation.
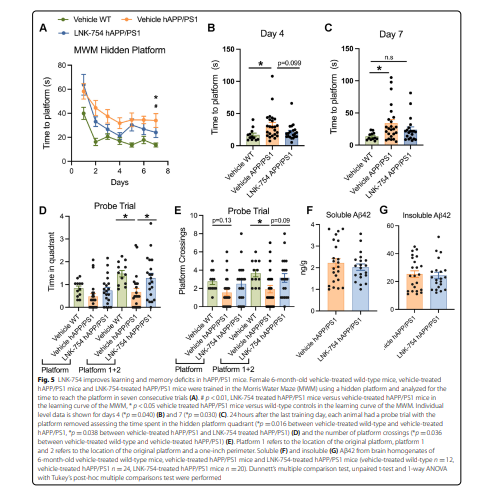
It is noteworthy that acute treatment with LNK-754 attenuated dystrophic neurites in aged 5XFAD mice and improved spatial learning and memory in hAPP/ PS1 mice. This supports the dual role of FTIs in the treatment of AD, by inhibiting Aβ generation and protecting against dystrophic neurites and cognitive decline after advanced plaque deposition. In line with this, haplodefciency of FTase specifically in APP/PS1 mice rescued cognitive impairment, while deficiency of FTase or GGTase both reduced Aβ deposition [62]. Further, the level of farnesylation has been shown to regulate synaptic plasticity and spine density in hippocampal slices in wildtype mice [63], so farnesylated proteins specifically may alter synaptic plasticity and microtubule stability, and these substrates could function independently of other prenylated substrates that affect plaque deposition.
While we hypothesize that farnesylated substrates regulating microtubule stability and endolyosome function improve dystrophic neurites and rescue behavior in the APP/ PS1 mouse, other mechanisms are possible. For example, Rhes is a small GTPase that undergoes farnesylation and is known to induce autophagy [64]. Rhes mediated behavior improvement in tau transgenic mice by lowering phospho-tau levels [9], and similarly Rhes could be responsible for improving memory in the LNK754 treated hAPP/PS1 mouse by reducing tau phosphorylation (e.g., p-tau ser404), as observed in LNK-754 treated 5XFAD mice, independently of dystrophic neurite improvement.
Farneslyation is critical for the proper functioning of multiple physiologically relevant proteins that could be responsible for mediating the beneficial effects observed in our study. Over 32 farnesylated substrates were recently identified through metabolic labeling in a human endothelial cell line [65], and in another recent study, 11 farnesylated proteins were enriched in forebrain-neuron-specific FTase knockout mice [63]. A major family of proteins affected in forebrain neurons by farnesylation is Ras GTPases [63], which mediate many cellular functions including endosomal trafficking. Ras proteins are implicated in AD pathogenesis; in postmortem brain tissues of patients with AD and mild cognitive impairment, increased activation of Ras signaling correlates with amyloid pathology and elevated levels of its downstream signaling molecule ERK [30].
Furthermore, the Ras family member Rhes was identified as a target of lonafarnib to activate lysosomes and reduce mutant tau aggregates [9]. Transcriptomics of neuronal FTase knockout in APP/ PS1 mice identified mTORC1 signaling and promotion of non-amyloidogenic processing as the top contributor to the beneficial effects on cognition and amyloid pathology, possibly mediated through a reduction in Ras or Rheb signaling [30]. Changes in Rhes or Rheb activity could reduce Aβ burden in 5XFAD mice, although it is unlikely that Ras was completely inactivated at the LNK-754 and lonafarnib concentrations used in this study. Several noncanonical non-Ras FTase substrates could instead mediate the observed effects. For example, the SNARE protein ykt6 enhances the trafficking of lysosomal hydrolases and could promote lysosomal function [8]. A direct FTase effect could also be responsible, since FTase can directly bind microtubules and affect the activity of the histone deacetylase HDAC6, thereby preventing tubulin deacetylation [33].
Conclusions
Upregulation of FTase signaling and protein farnesylation appears to be an early initiating event in AD pathogenesis [30]. In this study, we take a pharmacological approach to inhibit FTase in vivo in the 5XFAD mouse model. In mouse models of synucleinopathies and tauopathies, toxic protein aggregates were cleared directly by enhanced lysosomal degradation induced by FTI treatment [8, 9]. In contrast, we hypothesize that Aβ generation was inhibited through an indirect mechanism where enhancing axonal trafficking inhibited dystrophic neurite formation and BACE1 elevation. After the onset of plaque pathology, short-term LNK-754 treatment reduced dystrophic neurites in 5XFAD mice, and improved memory and spatial learning behaviors in hAPP/PS1 mice.

Our results provide a proof-of-concept study towards the use of FTIs to prevent Aβ accumulation and add to the substantial body of work that has demonstrated FTase genetic knockdown improves amyloid pathology and rescues cognitive function in AD mouse models [30, 62, 66]. The specific FTase substrates and downstream pathways responsible for the phenotypes observed in our study should be further elucidated in future studies. Lowering BACE1 buildup in dystrophic neurites with FTI treatment may slow the deposition of Aβ42 while offering a therapeutic strategy that avoids the of-target side effects typically seen when using BACE1 inhibitors [67, 68]. Furthermore, reducing the amyloid plaque burden in the brain will likely predict the clinical benefit of AD patients [67], and as such, FTIs could have important therapeutic implications for AD.


Acknowledgments
We thank D. Kirchenbuechler, P. Dluhy, and C. Arvanitis of the Center for Advanced Microscopy at Northwestern University for expert advice on the quantification of immunofluorescence images and helpful discussions, and funding support from NIH R01 AG030142. Finally, we thank S. Kemal and J. Popovic of the Vassar lab for their expertise and assistance with live imaging experiments and mouse perfusions, respectively.
Author Details
The Ken and Ruth Davee Department of Neurology, Northwestern University Feinberg School of Medicine, Chicago, IL 60611, USA. 2 BioEnergetics, Boston, MA 02115, USA. 3 Bial Biotech, Cambridge, MA 02139, USA. 4 Department of Neurology, Harvard Medical School, Cambridge, MA 02139, USA. 5 Mesulam Center for Cognitive Neurology and Alzheimer’s Disease, Northwestern University Feinberg School of Medicine, Chicago, IL 60611, USA.
References
1. Hardy J, Allsop D. Amyloid deposition as the central event in the etiology of Alzheimer’s disease. Trends Pharmacol Sci. 1991;12(10):383–8.
2. Hardy J, Selkoe DJ. The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science. 2002;297(5580):353–6.
3. Selkoe DJ. The molecular pathology of Alzheimer’s disease. Neuron. 1991;6(4):487–98.
4. Liu PP, Xie Y, Meng XY, Kang JS. History and progress of hypotheses and clinical trials for Alzheimer’s disease. Signal Transduct Target Ther. 2019;4:29.
5. Sevigny J, Chiao P, Bussière T, Weinreb PH, Williams L, Maier M, et al. The antibody aducanumab reduces Aβ plaques in Alzheimer’s disease. Nature. 2016;537(7618):50–6.
6. Cuddy LK, Prokopenko D, Cunningham EP, Brimberry R, Song P, Kirchner R, et al. Aβ-accelerated neurodegeneration caused by Alzheimer ’s-associated. Sci Transl Med. 2020;12(563):eaaz2541.
7. Cummings J, Lee G, Ritter A, Sabbagh M, Zhong K. Alzheimer’s disease drug development pipeline: 2020. Alzheimers Dement (N Y). 2020;6(1):e12050.
8. Cuddy LK, Wani WY, Morella ML, Pitcairn C, Tsutsumi K, Fredriksen K, et al. Stress-induced cellular clearance is mediated by the SNARE protein ykt6 and disrupted by α-Synuclein. Neuron. 2019;104(5):869–84.e11.
9. Hernandez I, Luna G, Rauch JN, Reis SA, Giroux M, Karch CM, et al. A farnesyltransferase inhibitor activates lysosomes and reduces tau pathology in mice with tauopathy. Sci Transl Med. 2019;11(485):eaat3005.
10. Mazzulli JR, Zunke F, Tsunemi T, Toker NJ, Jeon S, Burbulla LF, et al. Activation of β-Glucocerebrosidase reduces pathological α-Synuclein and restores lysosomal function in Parkinson’s patient midbrain neurons. J Neurosci. 2016;36(29):7693–706.
11. Nixon RA. The role of autophagy in neurodegenerative disease. Nat Med. 2013;19(8):983–97. 12. Bonam SR, Wang F, Muller S. Lysosomes as a therapeutic target. Nat Rev Drug Discov. 2019;18(12):923–48.
For more information:1950477648nn@gmail.com






