How Important Is The Value Of Monitor Unit (MU) in The Quality Of Kidney Stereotactic Ablative Body Radiotherapy(SABR) Plan
Mar 18, 2022
Contact: joanna.jia@wecistanche.com / WhatsApp: 008618081934791
On the reduction of aperture complexity in kidney SABR
Mathieu Gaudreault, Keith Offer, Tomas Kron, Shankar Siva Nicholas , Hardcastle
Abstract
Background: Stereotactic ablative body radiotherapy (SABR) of primary kidney cancers is confounded by motion. There is a risk of interplay effect if the dose is delivered using volumetric modulated arc therapy (VMAT) and flattening filter-free (FFF)dose rates due to target and linac motion. This study aims to provide an efficient way to generate plans with minimal aperture complexity.
Methods: In this retrospective study,62 patients who received kidney SABR(Stereotactic ablative body radiotherapy) were reviewed. For each patient, two plans were created using internal target volume-based motion management, on the average intensity projection of a four-dimensional CT. In the first plan, optimization was performed using a knowledge-based planning model based on delivered clinical plans in our institution. In the second plan, the optimization was repeated, with a maximum monitor unit (MU) objective applied in the optimization. Dose-volume, conformity, and complexity metric (with the field edge metric and the modulation complexity score) were compared between the two plans. Results are shown in terms of the median (first quartile—third quartile). Results: Similar dosimetry was obtained with and without the utilization of an objective on the MU(maximum monitor unit). However, complexity was reduced by using the objective on the MUs(monitor units) (modulation complexity score=0.55(0.50-0.61)/ 0.33(0.29-0.36), P-value<10-10, with/without the MU(monitor units) objective). Reduction of complexity was driven by a larger aperture area (area aperture variability = 0.68 (0.64-0.73)/ 0.42 (0.37-0.45), P-value<10-10,with/without the MU(monitor units) objective). Using the objective on the MUs(monitor units) resulted in a more spherical dose distribution (sphericity 50% isodose =0.73 (0.69-0.75)/ 0.64 (0.60-0.68), P-value<10-8, with/without the MU(monitor unit) objective) reducing the dose to organs at risk given respiratory motion.
Conclusions: Aperture complexity is reduced in kidney SABR(Stereotactic ablative body radiotherapy) by using an objective on the MU(monitor unit) delivery with VMAT and FFF dose rate.
KEY WORDs: FFF, interplay, kidney, SABR(Stereotactic ablative body radiotherapy), VMAT

cistanche can treat kidney disease improve renal function
INTRODUCTION
Stereotactic ablative body radiotherapy(SABR) is a novel treatment to treat patients with renal cell carcinoma (RCC) for whom surgery is not an option. It results in excellent local control and low toxicity rates in primary RCC.12 This technique is non-invasive and delivered in an outpatient procedure. Moreover, kidney SABR treatment is not limited to tumor size or kidney position as is the case with other alternatives to surgery such as radiofrequency ablation or cryoablation.3
As the tumor moves during treatment with respiratory motion, the interplay between the moving multileaf collimator(MLC) and tumor motion may result in discrepancies between planned and delivered doses. Several studies have demonstrated the limited impact of the interplay effect on conventionally fractionated treatments, as any discrepancies are averaged out during the course of treatment.4-10 As opposed to conventionally fractionated treatments, the interplay effect may impact SABR(Stereotactic ablative body radiotherapy) treatments because the number of fractions is typically 1-5. 1011 Strategies to minimize interplay effect include reducing the aperture complexity, 112 increasing the number of beams and fractions,10.12 reducing dose rate, 1213 decreasing tumor amplitude 13.14 and breathing cycle 10-1215, and treating with higher dose per fraction. 1012,16
Contemporary kidney SABR(Stereotactic ablative body radiotherapy) treatments are often treated with volumetric modulated arc therapy (VMAT), often due to the challenging geometric relationship between the target and adjacent organs at risk(OAR). Furthermore, flattening filter-free(FFF) delivery is highly attractive to reduce the treatment time via increased dose rates. Due to substantial reductions in beam-on time with high dose rates, and the use of ultra-hypofractionated regimens, the interplay effect may impact the fidelity of the planned treatment dose. Furthermore, reduction of aperture complexity and reduction of required monitor units (MUs) are of interest to reduce the treatment delivery time, which may be particularly important in the context of respiratory gating or breath-hold treatment delivery.
We have recently shown that reduction in aperture complexity can be achieved in lung SABR(Stereotactic ablative body radiotherapy) by using an optimization objective on the total number of MUs(monitor units), referred to as the"MU(monitor units) objective" in the Eclipse treatment planning system.17 The MU(monitor unit) objective has been available in the Eclipse treatment planning system for RapidArc optimization since version 8.5. Previous work established use of the MU(monitor units) objective results in a reduction in the total MUs(monitor units), and therefore in the beam-on time, while preserving adequate dosimetry by using the MU(monitor units) objective in the prostate,18.19head, and neck,18.20 gynecological,18 and lung SABR.21 However, the impact on the aperture complexity by using the MU(monitor units) objective was not addressed in these previous studies.
The purpose of this study is to determine the dosimetric impact of reducing aperture complexity via the inclusion of a penalty on total MUs(monitor unit) in the optimization of kidney SABR(Stereotactic ablative body radiotherapy) VMAT treatment plans. It is hypothesized that substantial reductions in aperture complexity can be achieved with minimal impact on dosimetric quality in kidney SABR.

cistanche bodybuilding
MATERIALS AND METHODS
We included consecutive 62 patients with primary RCC treated with SABR(Stereotactic ablative body radiotherapy) between 2012 and 2018 at our institution. Fractionation was 26 Gy in a single fraction for lesion size smaller or equal to 4 cm and 42 Gy in three fractions for lesion size larger than 4 cm.22 Out of the 62 patients, 23 patients received 26 Gy in a single fraction and 38 patients had 42 Gy in three fractions. One patient was treated at 18 Gy in one fraction but was replanned in this study with 26 Gy in one fraction. These patients were treated with 3D conformal radiation therapy, intensity-modulated radiation therapy, or VMAT. Ethics approval for this study was provided by Peter MacCallum Cancer Centre.
Each patient was simulated using a four-dimensional CT scan (4DCT). The tumor was segmented into all respiratory phases. Gross tumor volume (GTV) was accumulated on the average intensity projection (AlP)of the 4DCT to generate an internal target volume (ITV). A planning target volume (PTV)was created using an isotropic 5 mm expansion of the ITV. The AlP of the 4DCT was used for contouring, planning, optimizing, and calculating the dose distribution. OARs were segmented on the AIP depending on the extent of respiratory motion. OAR contours in some cases overlap with the ITV contour in cases where the OAR is proximal to the tumor.
All patients were replanned for the purpose of this study. Plans were generated by using the Eclipse treatment planning system (Varian Medical Systems, Palo Alto, CA, USA) with AcurosXB Algorithm (v15.6.06) reporting dose to medium for dose calculation and Photon Optimization Algorithm (v15.6.06) for optimization.
Two coplanar ipsilateral arcs of 210°were used for each plan with the arc rotating from the posterior-anterior direction around to 30°past midline. The isocenter was placed at the centroid of the PTV. Collimator angles were set at 5°/355°. A clinical 10 MV-FFF beam model with a maximum dose rate of 2400 MU(monitor units)/min was used with the HD 120 MLC.
Dose objectives to target and constraints to normal tissue used for optimization were applied by using a knowledge-based planning (KBP) model (RapidPlan v15.5.11 Varian Medical Systems, Palo Alto, CA, USA). The model was constructed from 53 clinical kidney SABR(Stereotactic ablative body radiotherapy) plans delivered at our institution. An upper point does objective was added to certain OARs to control the high dose region. Parameters used in the model are shown in Table 1. The normal tissue objective (NTO) was set to "Automatic NTO" with a priority of 150.
The primary method of reducing aperture complexity was an objective on the upper value of MU(monitor units). Once selected, this parameter defines a range of targeted MU(monitor units) for the plan. The optimizer penalizes any MU(monitor units) outside the desired interval. The penalty is weighted by a strength assigned to the MU(monitor units) objective. This parameter takes values between 0 and 100. In addition to the MU(monitor units) objective, the aperture shape controller was set to "Very High", the convergence mode to "On", and the multiresolution(MR) level at the restart to "MR3" for all plans.
Optimization and calculation was a two-step process to determine the upper value of the MU(monitor units) objective. The original MU(monitor units) was first obtained by optimizing and calculating the dose without the MU(monitor units) objective in one single process without user interaction. The plan was then normalized so that 100% of the prescription dose covers 95% of the target volume and is referred to as the "NMUO" plan in this study.
The NMUO plan was copied and an objective on the MU(monitor units) was added. The upper value of the MU(monitor units) objective was set to 50% of the original total MUs(monitor units) with a strength of 70, and the plan was then optimized from scratch through one process without user interaction. The plan was also normalized so that 95% of the target volume is covered with 100% of the prescription, referred to as the "MUO" plan.
Any plan for which a dose constraint was not respected was replanned by adjusting the objectives to respect dose limits. These replays are referred to as"modified KBP" as opposed to the "original KBP" plan. These were all for cases in which an OAR was close to or overlapping with the target, and was identified as those with challenging plan geometry. If a structure was overlapping with the PTV or the ITV, the PTV and ITV structures were cropped to generate an optimization structure. In two patients, the two partial arcs of 210°had to be modified to two full rotation arcs of 358°. The two calculation steps method was repeated by modifying the upper dose limit to the organ up to the point where all dose limits were respected. Where possible while meeting OAR constraints, the plans were normalized to 95% of the target volume was covered by the prescription dose. Where this was not possible, loss to target cover-age was accepted to ensure OAR constraints were respected.
Dose metrics for the target and OARs, shown in Table 2, were evaluated. Dose limits were based on QUANTEC recommendations.23-25 Plan generation, optimization, calculation, and metrics extraction were done by using the Eclipse Scripting Application Programming Interface (ESAPI. Plan conformity was determined using the RTOG conformity index, defined as the reference isodose volume divided by the target volume.26 The 95% isodose was used as reference isodose to calculate the conformity index (CI95). A value of Cl95=1 indicates ideal conformation. The target was partially irradiated if Cl95<1, while the irradiated volume was greater than the target volume if Cl95>1.27Moreover, acceptable Cl95 values were defined as values smaller than 1.2 while minor deviations were defined for values greater than 1.2 but smaller than 1.5.28 Conformity of the low dose region, was assessed with the Cl50, or equivalently the R50, defined as the 50% isodose volume divided by the target volume. CI50 conformity deviation was assessed through the ALARA principle with a planning goal of Cl50 <5 for all PTV volumes.
Due to the proximity of bowel structures to the target, and their variation in position between treatment planning and each treatment session, it is desirable to minimize higher isodose lines extending between bowel loops which may arise as a consequence of using the bowel structures for optimization. This was assessed by calculating the sphericity of the isodoses lines. The 100% and the 50% iso-dose lines were converted to contours and exported. Pyradiomics v3.02? was used to calculate the sphericity of the contour. The resulting value ranged between O and 1, where 1 indicated a perfect sphere.
The robustness of the plans was measured by calculating the edge metric (EM) and the modulation complexity score(MCS). EM was calculated according to C1=0 and C2=1.30-32 In this representation, EM reports they-leaf sides normalized by the area aperture weighted per control point. Plan complexity decreases as EM decreases to 0. MCS was interpreted according to McNiven et al.3 and used by others.3134 MCS is a score based on adjacent leaf sides, measured by leaf segment variability (LSV), and on the area of the aperture, measured by the area aperture variability (AAV). For a given control point, the LSV is proportional to the average position difference between adjacent leaves' overall leaves contributing to the open field normalized by the difference between maximal position and the minimal position overall leaves within a bank. The AAV is the aperture area normalized by the maximum aperture area overall control points. LSV and AAV were also weighted per control point and defined as LSVw and AAVw. The three quantities take values between 0 and 1. Plan complexity decreases as MCS, LSVw, and AAVw increase to 1. The weights per control point used were the same for the calculation of EM and MCS.30 Both plan complexity metrics were calculated from an in-house script. Details of the calculation are shown in the appendices. To provide a lower bound of complexity reference point, the beams in the MUO(monitor units) plan were converted to dynamic conformal arc therapy (DCAT) fields by fitting the aperture to the PTV in each control point. The complexity metrics EM and MCS, including LSVw and AAVw, were computed for the DCAT plans.
The average difference of each metric between plans with and without the MU(monitor units) objective was calculated and a Wilcoxon signed-rank test was performed to determine the statistical significance of the median difference by using Scipy v1.5.2. The null hypothesis was rejected if the P-value was less than 0.05 (5% significance level). Statistical quantities are reported in terms of the median(first quartile-third quartile).
| T ABLE 1 Knowledge-based planning model optimization objectives | T ABLE 2 Normal dose tissues constraints were used |
 |  |
RESULTS
3. A |Dosimetry
An example of typical dose distribution in the axial plane is shown in Fig.1 for an original KBP plan (top row) and a modified KBP plan (bottom row) obtained with the MU(monitor units) objective (a) and (c) and without the MU(monitor units) objective(b) and (d). PTV volumes for the whole cohort ranged from 25 ccs to 390 ccs with a median of 131 ccs.
Resulting dose metrics for the single fraction cohort are shown in Fig. 2(a). ITV coverage by the 100% isodose and the ITV hotspot were slightly higher in plans with the MU(monitor units) objective(ITV D100%=27.5(27.4-27.7)Gy/ 27.1 (27.0-27.3) Gy, P-value <10-5 and ITV 2%= 32.2(31.8-32.9) Gy/31.7 (31.5-32.2)Gy,P-value<10-5, MUO/NMUO).
The addition of the MU(monitor units) objective did not significantly change most OAR dose metrics. Near maximum dose to the ipsilateral kid-ney was slightly higher when the MU(monitor units) objective was used(ipsilateral kidney D1.5cc=28.6 (28.3-28.8) Gy/28.2(28.1-28.5)Gy, P-value <10-, MUO/NMUO). There were no statistically significant differences in dose metrics for the remaining OARs (P-value =[0.2,0.65]).
In one patient the dose limit to the large bowel was not respected due to overlap with the ITV. Coverage to the PTV and ITV was slightly higher with the MU(monitor units) objective for this modified KBP plan (PTV D95%=23.4 Gy / 22.9 Gy, MUO/NMUO and ITV D100%=16.4 Gy /16.3 Gy, MUO/NMUO). For this patient, the dose to ipsilateral kidney, large bowel, and skin increased with MUO while dose metrics for the small bowel, stomach, and liver were decreased with MUO.
Dose metrics for the whole multi fraction cohort are shown in Fig.2(b).Coverage to the PTV and ITV was similar irrespective of use of the MU(monitor units) objective (PTV D95%=42.0(41.1-42.0) Gy/ 42.0 (41.6-42.0) Gy, P-value<10-² and ITV D100%=43.4(41.2-43.9)Gy/43.3(41.2-43.7)Gy,P-value<10-3,MUO/NMUO).Near maximum dose to the ITV was also similar with and without the MU(monitor units) objective(ITV D2%=53.7 (52.9-54.3)/53.1(52.1-53.6)Gy,P-value <10-3, MUO/NMUO).
Interestingly, near-maximum dose to the small bowel was reduced in some cases by using the MU(monitor units) objective but the differences were not statistically significant (small bowel D0.03cc=19.2 (15.4-27.1)Gy/23.7(18.1-27.2)Gy, P-value=0.08, MUO/NMUO). The near maximum spinal cord dose increased with the application of the MU(monitor units) objective but remained below the dose limit (spinal cord D0.03cc=10.3(6.9-11.6)Gy /9.9(6.4-11.8) Gy, P-value=0.01, MUO/NMUO). Near maximum dose to the ipsilateral kidney was similar between the two plans (ipsilateral kidney D1.5cc= 46.2 (45.9-47.1)Gy / 46.0(45.6-46.7)Gy, P-value<10-2, MUO/NMUO). Application of the MU(monitor units) objective did not make a statistically significant difference for other OARs(P-value=[0.17,0.75]).
KBP plans required modification for 15/38 patients in the multi-fraction group to meet dose constraints. Refinement of the objectives was required to meet spinal cord constraints in one patient. The dose limit to the skin was not respected in two patients. These were two left-sided tumors exophytic close to the patient skin. The small bowel was overlapping with the target in seven patients, the large bowel was overlapping the ITV in three patients, and both the small and the large bowel were overlapping the ITV in two patients.
Optimization to make modified KBP plans clinically deliverable resulted in a loss of coverage to the target (PTV D95%= 40.9 (40.0-41.6)Gy/ 41.4 (40.7-41.8)Gy,MUO/NMUO,P-value<10-2)as opposed with original KBP plans (PTV D95%=42 Gy for all plans MUO/NMUO). Use of the MU(monitor units) objective increased the ITV D100%and ITV D2% in the original KBP group (ITV D100%=43.8(43.6-44.1)Gy/ 43.5 (43.3-43.7)Gy,P-value<10-2and ITV 2%=53.0 (52.6-53.7)Gy/ 52.4(51.7-53.3)Gy,P-value <10-2,MUO/NMUO). Differences in the ITV D100% and ITV D2% medians were not significant in the modified KBP group (P-value=0.18 and 0.06, respectively).
Near maximum dose to the small bowel was close to the dose limit in modified KBP plans with 60%(9/15) of plans having cover-age reduced to meet this limit (small bowel D0.03cc=28.8 (23.8-29.6) Gy/ 28.5 (25.4-28.9) Gy, P-value=0.42, MUO/NMUO). However, the near-maximum small bowel dose was reduced for original KBP plan with the MU(monitor units) objective (small bowel D0.03cc =17.4 (13.5-19.2) Gy / 21.7(17.0-23.9)Gy,P-value=0.01,MUO/NMUO).
The only significant statistical difference in dose metric medians of modified KBP plans was for PTV D95%(other P-value =[0.06,0.93]). Dose metric median values were systematically higher in all OARs except small bowel in original KBP plans with the MU(monitor units) objective, but still far from any dose limit. Near maximum spinal cord dose was larger with the MU objective (spinal cord D0.03cc=8.6 (6.5-11.6) Gy /7.1 (5.9-11.3)Gy, P-value=0.01, MUO/NMUO)while differences to the stomach were similar with and without the MU(monitor units) objective (stomach D5cc= 2.62(0.4-6.9) Gy/2.60 (0.4-6.8)Gy, P-value =0.04, MUO/NMUO). Other differences in original KPB were not statistically significant (P-value = [0.09,0.72]).
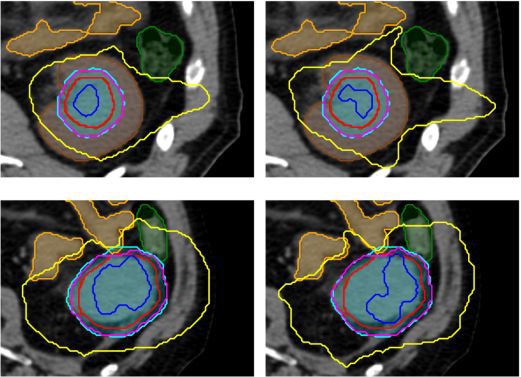
F I G . 1. Axial view of typical dosimetry obtained for one patient with original KBP (top row) and one patient with modified KBP (bottom row). Isodoses 50% (yellow), 100% (magenta), and 120% (blue) are shown for plan with the MU objective (a) and (c) and without the MU objective (b) and (d). The structures ITV (red), PTV (cyan), nontumor ipsilateral kidney (brown), small bowel within 5 cm of the ITV (orange), and large bowel (green) are shown.
3.B Conformity Index and sphericity
CI95 is shown in Fig.3(a). The Cl95 median and interquartile range were similar with and without the MU(monitor units) objective(Cl95=1.10 (1.08-1.11)/ 1.11(1.09-1.13), P-value<10~5, MUO/NMUO). Ci95 was smaller in modified KBP plans when using the MU(monitor units) objective (CI95=1.03(1.01-1.08)/1.08(1.06-1.10), P-value<10-3,MUO/NMUO)while it was similar in original KBP plans(Cl95=1.11 (1.09-1.12)/ 1.11(1.09-1.13), P-value<10-2, MUO/NMUO). One plan had CI95<1 without the MU(monitor units) objective and three plans had Cl95<1 with the MU(monitor units) objective, all for modified KBP plans in the multi fraction cohort. All values of CI95 satisfied the planning objective (CI95<1.2) with and without the MU objective.
Cl50 was reduced when using the MU(monitor units) objective(Cl50=3.4 (3.2-3.7)/3.6(3.3-3.8),P-value<10-', MUO/NMUO), as shown in Fig 3(b).The same conclusion was obtained with modifed KBP plans (CI50=3.3(3.1-3.5)/ 3.5(3.3-3.7), P-value<10-2,MUO/NMUO)and original KBP plans(Cl50=3.5 (3.3-3.7)/3.6(3.4-3.9),P-value<1073, MUO/NMUO). All values of Cl50 were smaller than the plan-ning objective (CI50<5) with and without the MU(monitor units) objective.
Isodoses were more spherical when using the MU(monitor units) objective. An example of the 100% and 50% isodoses for the patient with the worst isodose 50% sphericity in NMUO plan is shown in Fig.4(a) with and in Fig.4(b) without the MU(monitor units) objective. A signify-cant increase in the isodose 50% sphericity was observed when using the MU(monitor units) objective (sphericity 50% isodose =0.73(0.69-0.75)/ 0.64(0.60-0.68),P-value <10-8, MUO/NMUO) as shown in Fig.4(c). This effect was also observed with the 100% isodose,as shown in Fig.4(d), but to a lower extent (sphericity of 100% iso-dose=0.82 (0.80-0.83)/ 0.80 (0.79-0.82), P-value<10-8, MUO/NMUO). The same conclusions hold for both modified and original KBP plans.
3. C Aperture complexity
Using the objective on the MU(monitor units) yielded a reduction in the total MU(monitor units) for both the single fraction group(total MU=5617 (5164-5910)/10439 (9560-11237), P-value<10-6, MUO/NMUO)and the multi fraction group (total MU=3022 (2619-3238)/5818 (5064-6428), P-value<10-7, MUO/NMUO).

F I G . 2. Comparison of dose metrics with (MUO) and without (NMUO) the MU objective for patients with fractionation (a) 26 Gy/1 Fx (24 patients) and (b) 42 Gy/3 Fx (38 patients). Red dotted lines indicate a dose limit.
Using the objective on the total MUs also reduced the aperture complexity, as measured by EM and MCS showed in Fig. 5(a). Results are compared with the aperture complexity of their DCAT counterpart in the figure. The median and interquartile range of the distribution and statistical significance are reported in Table 3. Multifractionation subset plans were less complex than the single fraction subset plans. The type of fractionation was based on lesion size with the PTV larger in the multi fractionation subset. This leads to a larger aperture area to cover the target and to a smaller EM and larger MCS. Using the MU(monitor units) objective decreased aperture complexity (relative difference of the medians with respect to NMUO plan was-61% and 64% in EM and MCS, respectively, both P-value <10-8). The two regimes are shown in Fig. 5(b). Even if some modulation is reduced in the MUO plan, they are still more complex than their DCAT counterpart(relative difference of the medians in the complexity of CF with respect to MUO plan was -61% and 50% in EM and MCS, respectively, both P-value <10-10).
The impact of using an objective to the MU(monitor units) is explained by AAVw and LSVw. These two quantities are shown in Fig. 5(b)while the median and interquartile range of the distribution and statistical significance are detailed in Table 3. Reduction in complexity was driven by a larger area aperture with the MU(monitor units) objective while leaf traveling changed minimally. This was expected as the parameter controlling the leaf travel, the so-called aperture shape control, was the same in both subsets. Using an objective on the MU(monitor units) reduces the degree of freedom available to the optimizer as the MUs(monitor units) were constrained. This loss was compensated by a larger aperture area. Therefore complexity was reduced.

cistanche bodybuilding
DISCUSSION
Results suggest that aperture complexity is minimized in kidney SABR(Stereotactic ablative body radiotherapy) treatment by using VMAT with FFF dose rate and an objective on the MU(monitor units). Using an objective on the upper-value MU forced the optimizer into using a larger aperture area. Since leaf travel was kept constant, aperture area increase resulted in a reduction in plan complexity. Importantly, there was a minimal dosimetric compromise when using the MU(monitor units) objective. Using the objective on the MU was even beneficial to the small bowel in the multi fraction group for the original KPB plan. Furthermore, constraining the MUs(monitor units) resulted in more spherical isodoses which reduced the conformity indices, and dose splay between bowel loops was reduced. This is particularly important with respect to positional variation of small and large bowel loops at the time of treatment.
The main advantage of the objective on the MU(monitor units) is the beam-on time reduction that follows from the MU(monitor units) reduction (50% reduction in this study). The value of the upper limit on the MU(monitor units) objective and its associated strength was not optimized in this study but was derived from previously investigated values in lung SABR(Stereotactic ablative body radiotherapy).35 The level of complexity achieved with VMAT and DCAT fields was similar in this previous study. This was not the case in this work as VMAT plans with the MU(monitor units) objective in use were still more complex than DCAT fields. This difference between the two studies may be due to the parameters used in the application of the MU(monitor units) objective (Max MU = 50% and strength =70 in this study as opposed to Max MU =40% and strength =80 in the previous study). Furthermore, the planning process used in this work required optimization of a plan without the MU(monitor units) objective, and subsequent re-optimization using the original MUs to derive the MU objective setting. A method involving a fixed upper limit to the MU objective per site and fraction would be of interest. Our results showing the MU(monitor units) distribution in MUO plans may be useful to start optimization directly with the second step in the optimization used in this work.
Modification to the KBP model had to be performed in 26%(16/62) of the patients. PTV and ITV were cropped and the resulting structures were used as optimization structures in 13 patients. As a result, target coverage was reduced with the use of the MU(monitor units) objective in the multi fraction group. OAR dosimetry was similar with and without the MU(monitor units) objective and all differences in dose metric medians were not statistically significant. These plans involved complex geometry but the conformity indices were lowered, isodoses were more spherical and the aperture complexity was reduced when using the MU(monitor units) objective. Allowing increased complexity for these plans might be needed to achieve the same plan quality as the original KBP plan. Utilization of an objective on the MU(monitor units) is recommended for plans with less complex target-OAR relationships, however, where there is more complex geometry, increased modulation may be required to meet plan objectives.
There are some limitations to this study. The shape of the GTV was preserved on each phase although the GTV might deform with motion. Modification to the GTV contour might affect the ITV volume resulting from the accumulation method. Consequently, the ITV may overlap with a surrounding OAR which would require modification to the KBP model. Furthermore, OAR contours used in this study were the clinical contours on the AIP image of the 4DCT. All OARs could have been delineated on each respiratory phase and accumulated on the AlP image. This process would have resulted in more patients with overlapping ITV and OAR and hence requiring manual modification of the KBP plan to meet objectives.

cistanche bodybuilding
CONCLUSION
In conclusion, kidney SABR(Stereotactic ablative body radiotherapy) plan quality is optimal by using an objective on the upper value of the MU(monitor units) with VMAT and FFF dose rate. This combination leads to similar dosimetry compared with the plan without the MU(monitor units) objective. Isodoses are more spherical with the MU objective which reduces dose splay between OAR. Moreover, the open aperture area is larger when using the MU(monitor units) objective which reduces the aperture complexity. Finally, the MUs(monitor units) are significantly reduced with the objective on the MU, which reduces the treatment time and the probability of intrafraction variation between plan and delivery.
REFERENCES
1. Siva S, Pham D, Gill S, Corcoran NM, Foroudi F.A systematic review of stereotactic radiotherapy ablation for primary renal cell carcinoma. BJU Int.2012;2012:E737-E743.
2. Siva S, Louie AV, Wagner A, et al, Pooled analysis of stereotactic ablative radiotherapy for primary renal cell carcinoma: a report from the International Radiosurgery Oncology Consortium for Kidney (IROCK).Cancer.2017;124:934-942.
3. Kunkle DA, Uzzo RG. Cryoablation or radiofrequency ablation of the small renal mass. Cancer.2008;113:2671-2680.
4. Bortfeld T, Jokivarsi K, Goitein M, Kung J, Jiang SB.Effects of intra- fraction motion on IMRT dose delivery: statistical analysis and simulations. Phys Med Biol. 2002;47:2203-2220.
5.Jiang SB, Pope C, Al Jarrah KM,Kung JH, Bortfeld T, Chen GTY. An experimental investigation on intra-fractional organ motion effects in lung IMRT treatments. Phys Med Biol.2003;48:1773-1784.
6. Duan J, Shen S, Fiveash JB, Popple RA, Brezovich IA. Dosimetric and radiobiological impact of dose fractionation on the respiratory motion-induced IMRT delivery errors: a volumetric dose measurement study. Med Phys. 2006;33:1380-1387.
7. Bortfeld T, Jiang SB, Reitzel E.Effects of motion on the total dose distribution. Sem Radiat Oncol.2004;14:41-51.
8. Court L, Wagar M, Berbeco R, et al, Evaluation of the interplay effect when using RapidArc to treat targets moving in the craniocaudal or right-left direction. Med Phys.2010;37:4-11.
9. Rao M, Wu J, Cao D, et al, Dosimetric impact of breathing motion in lung stereotactic body radiotherapy treatment using intensity-modulated radiotherapy and volumetric modulated arc therapy. Int JRadiat Oncol Biol Phys. 2011;83:251-256.
10. Ong CL, Dahele M, Slotman BJ, Wilko FARV. Dosimetric impact of the interplay effect during stereotactic lung radiation therapy delivery using flattening filter-free beams and volumetric modulated arc therapy. Int J Radiat Oncol.2013:86:743-748.
11. Gauer T, Sothmann T, Blanck O, Petersen C, Werner R.Under-re- ported dosimetry errors due to interplay effects during VMAT dose delivery in extreme hypofractionated stereotactic radiotherapy. Strahlenther Onkol. 2018;194:570-579.
12. Edvardsson A, Nordström F, Ceberg C, Ceberg S. Motion-induced interplay effects for VMAT radiotherapy. Phys Med Biol. 2018;63:743-748.
13. Rao M, Ye J, Spiegel J, Takamiya R, Mehta VK, Wu J.Interplay effect during lung SBRT with VMAT delivered by flattening filter-free beams. Int J Radiat Oncol.2014;90:S192.
14. LiX, Yang Y, LiT, Fallon K, Heron DE, Huq MS.Dosimetric effect of respiratory motion on volumetric-modulated arc therapy-based lung SBRT treatment delivered by TrueBeam machine with flattening filter-free beam.J Appl Clin Med Phys.2013;14:195-204.
15. Ong CL, Verbakel WFAR, Cuijpers JP, Slotman BJ, Senan S. Dosimetric impact of interplay effect on RapidArc lung stereotactic treatment delivery.IntJ Radiat Oncol Biol Phys.2011;79:305-311.
16. Archibald-Heeren BR, Byme MV, Hu Y, Cai M, Wang Y. Robust optimization of VMAT for lung cancer: dosimetric implications of motion compensation techniques. J Appl Clin Med Phys.2017;18:104-116.
17. Burton A, Offer K, Hardcastle N. A robust VMAT delivery solution for single-fraction lung SABR(Stereotactic ablative body radiotherapy) utilizing FFF beams minimizing dosimetric compromise. J Appl Clin Med Phys.2020;21:299-304.
18. Jiménez-Puertas S, Sanchez-Artun D, Hermida-L6pez M. Assessment of the Monitor Unit Objective tool for VMAT in the Eclipse treatment planning system. Rep Pract Oncol Radiother.2018;23:121-125.
19.Clemente S, Cozzolino M,Chiumento C, Fiorentino A,Caivano R, Fusco V. Monitor unit optimization in RapidArc plans for prostate cancer.J Appl Clin Med Phys.2013;14:52-63.
20. Ahamed S, Singh N, Gudipudi D, et al, Assessment of monitor unit limiting strategy using volumetric modulated arc therapy for cancer of hypopharynx. Physica Med.2017;35:73-80.
21. Huang BT, Lin Z, Lin PX, Lu JY, Chen CZ. Monitor unit optimization in stereotactic body radiotherapy for small peripheral non-small cell lung cancer patients.Sci Rep.2015;5:1-9.
22. Siva S, Chesson B, Bressel M, et al, TROG 15.03 phase Ⅱl clinical trial of Focal Ablative STereotactic RAdiosurgery for Cancers of the Kid-ney-FASTRACK II.BMC Cancer.2018;18:1030-1040.
23. Pan CC, Kavanagh BD, Dawson LA, et al, Radiation-associated liver injury. Int J Radiat Oncol Biol Phys.2010;76:S94-S100.
24. Dawson LA, Kavanagh BD, Paulino AC, et al, Radiation-associated kidney injury.IntJRadiat Oncol Biol Phys.2010;76:S108-S115.






