Immunological Characteristics Govern The Transition Of COVID-19 To Endemicity
Jul 24, 2023
Humans have regularly been threatened by emerging pathogens that kill a substantial fraction of all people born. Recent decades have seen multiple challenges from acute virus infections, including severe acute respiratory syndrome (SARS), Middle East respiratory syndrome (MERS), Hendra, Nipah, and Ebola. Fortunately, all were locally contained. When the containment is not immediately successful, as is likely for the novel beta coronavirus severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (1, 2), we need to understand and plan for the transition to endemicity and continued circulation, with possible changes in disease severity owing to virus evolution and buildup of host immunity and resistance.
Viral infection is a great medical challenge facing the world today, characterized by widespread morbidity and high transmissibility. Acute viral infection, especially pneumonia, has become a global public health problem, seriously affecting people's health and safety.
Immunity is one of our most important weapons in the fight against viral infections. Human immunity refers to the body's resistance to pathogens, including innate immunity and acquired immunity. Innate immunity refers to the natural immune system of the human body, which can strike pathogens promptly; acquired immunity refers to the immune response produced by the body after exposure to pathogens.
Acute viral infection can significantly affect the body's immunity. On the one hand, the virus will enter the body through various channels, disrupt the normal immune defense of the human body, destroy the balance and function of the immune system, and cause the body's immunity to decline. On the other hand, the inflammatory response and immune response after virus infection have caused a great burden on the human body, consuming a lot of energy and nutrients, resulting in fatigue and weakness, and further decline in immunity.
Therefore, improving immunity is one of the effective ways to prevent acute viral infection. Many researchers have found that a healthy diet, moderate exercise, a good attitude, and adequate sleep can enhance immunity. In addition, people can prevent certain viral infections by getting vaccinated. It is believed that with the continuous advancement of science and technology, we will have better progress in the prevention and treatment of acute viral infections.
In short, one should maintain a positive attitude to face acute viral infection. Only by strengthening health education and enhancing personal immunity can we more effectively prevent and control virus infection and ensure people's health and safety. From this point of view, we need to improve our immunity. Cistanche can significantly improve immunity, because Cistanche is rich in various antioxidant substances, such as vitamin C, carotenoids, etc. These ingredients can scavenge free radicals, reduce oxidative stress, and improve the resistance of the immune system.

Click cistanche tubulosa benefits
SARS-CoV-2 is an emerging virus that causes
COVID-19. The virus has a high basic reproductive number (R0) and is transmissible
during the asymptomatic phase of infection,
both of which make it hard to control (3). However, there are six other coronaviruses with
known human chains of transmission, which
may provide clues to future scenarios for
the current pandemic. There are four human
coronaviruses (HCoVs) that circulate endemically around the globe; these cause only mild
symptoms and are not a considerable public
health burden (4). Two other HCoV strains,
SARS-CoV-1 and MERS-CoV, emerged in recent decades and have higher case fatality ratios
(CFRs) and higher infection fatality ratios (IFRs)
than COVID-19 but were contained and thus
never spread widely (5, 6).
We propose a model to explore the potential changes in both transmission and disease severity of emerging HCoVs through the transition to endemicity. We focus on SARSCoV-2 and discuss how the conclusions would differ for emerging coronaviruses more akin to SARS-CoV-1 and MERS-CoV.
We hypothesize that all HCoVs elicit immunity with similar
characteristics, and the current acute public
health problem is a consequence of epidemic
emergence into an immunologically naïve
population in which older age groups with
no previous exposure are most vulnerable to
severe disease. We use our estimates of immunological and epidemiological parameters
for endemic HCoVs to develop a quantitative
model for endemic transmission of a virus with
SARS-CoV-2–like characteristics, including the
age dependence of severity. Our model explicitly considers three separate measures for
immune efficacy that wane at different rates
(fig. S1).
Building on ideas from the vaccine modeling literature, we suggest that immunity may
protect in three ways (7). In its
most robust form, sterilizing immunity can
prevent a pathogen from replicating, thereby
rendering the host refractory to reinfection.
We term this property immune efficacy concerning susceptibility (IES). If immunity
does not prevent reinfection, it may still attenuate pathology due to reinfection (IEP)
and/or reduce transmissibility or infectiousness (IEI). Indeed, experimental reexposure
studies on endemic HCoVs provide evidence
that the three immune efficacies do not wane at
the same rate (8, 9). Callow et al.’s experimental
study (8) shows that reinfection is possible
within one year (relatively short IES); however,
upon reinfection, symptoms are mild (high
IEP) and the virus is cleared more quickly
(moderate IEI). Details on the derivation of
the model can be found in section 2 of the
supplementary materials (SM).

We reanalyze a detailed dataset that estimates age-specific seroprevalence based on both immunoglobulin M (IgM; acute response) and IgG (long-term memory) against
all four circulating HCoVs in children and
adults (10) to estimate parameter ranges for
transmission and waning of immunity (Fig. 1A).
The rapid rise in both IgM and IgG seroprevalence indicates that primary infection with
all four endemic HCoV strains happens early
in life, and our analysis of these data gives us
an estimate for the mean age of primary infection (MAPI) between 3.4 and 5.1 years, with
almost everyone infected by age 15 (see SM
section 1 for details).
The absence of detectable
IgM titers in any individual over the age of
15 years suggests that reinfection of adults
causes a recall response, indicating that while
HCoV-specific immunity may wane, it is not
lost. Whether immunity would wane to naïve
levels in the absence of high pathogen circulation remains an open question.
For most people to be infected so early in life—younger even than measles in the prevaccine era—the attack rate must exceed transmission from primary infections alone. The model shows that a high attack rate can arise from a combination of high transmissibility from primary infections (i.e., high R0), waning of sterilizing immunity, and substantial transmission from reinfections in older individuals. The rapid waning of sterilizing immunity is also reported in experimental HCoV infections of humans, which showed that reinfection is possible 1 year after an earlier infection, albeit with milder symptoms (IEP) and a shorter duration (IEI) (8). Figure 1B shows the plausible combinations of waning immunity and transmission from reinfected individuals that are required to produce the MAPI observed in Fig. 1A, based on steady-state infection levels (see SM section 2.1 for details). Table 1 shows the ranges of the parameters used in our simulations.
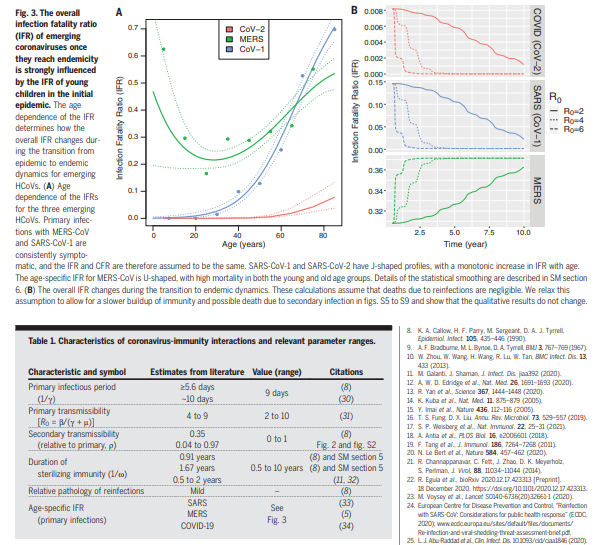
At the beginning of an outbreak, the age distribution of cases mirrors that of the population (Fig. 2A). However, once the demographics of infection reaches a steady state, our model predicts that primary cases occur almost entirely in babies and young children, who, in the case of COVID-19, experience a low CFR and a concomitantly low IFR. Reinfections in older individuals are predicted to be common during the endemic phase and to contribute to transmission, but in this steady-state population, older individuals, who would be at risk for severe disease from a primary infection, have acquired disease-reducing immunity after infection during childhood. The top panel of Fig. 3B illustrates how the overall IFR for SARSCoV-2 drops drastically, eventually falling below that of seasonal influenza (~0.001) once the endemic steady state is reached.
The time it takes to complete the shift in IFR as endemicity develops depends on both transmission (R0) and loss of immunity [waning of sterilizing immunity (w) and transmissibility of reinfections (r)], as shown in Fig. 2B and Fig. S4. The transition from epidemic to endemic dynamics is associated with a shift in the age distribution of primary infections to lower age groups (Fig. 2A).
This transition may take anywhere from a few years to a few decades, depending on how quickly the pathogen spreads. The rate of spread, measured by R0, is determined by a combination of viral properties and the frequency of social contacts and may therefore be reduced by social distancing. The top panel in Fig. 2A shows the effect of reducing R0 to 2, whereas the middle and bottom panels show the dynamics for higher R0, which are more akin to those of SARS-CoV-2 in the absence of control measures.

If the transmission is high, the model predicts a high caseload and high death rate in earlier years following emergence (Fig. 2 and Fig. S5). We see that, as might be expected, longer-lasting sterilizing immunity slows down the transition to endemicity (Fig. 2B). These results are robust to a more biologically realistic distribution for the duration of sterilizing immunity and the possibility that the generation of protective immunity requires more than one infection (see SM section 3 and figs. S5 to S9).
Slowing down the epidemic through social distancing measures that reduce R0 to close to 1 flattens the curve, thus delaying infections and preventing most deaths from happening early on, affording critical time for the development of an effective vaccine (fig. S10). If vaccine-induced IES and IEP immunity is similar to that induced by HCoV infections, the vaccine may usher in the endemic regime more quickly. The model code (see the acknowledgments) provides a flexible scaffolding for studying alternative vaccination scenarios. Notably, the model predicts that once the endemic state is reached, mass vaccination may no longer be necessary to save lives (see SM section 4 and fig. S11).
We can extend our predictions to two other
potentially emerging coronavirus infections,
SARS and MERS. Our model predicts that in
the endemic state, the IFR of a circulating HCoV
depends primarily on the severity of childhood
infections. In the case of SARS-CoV-1, which
is more pathogenic than SARS-CoV-2, we still
expect a low disease burden in the endemic
phase, because SARS-CoV-1, like SARS-CoV-2,
has a low IFR in young people (Fig. 3). However, data suggest that not all emerging HCoVs
follow this optimistic pattern; the overall IFR
of an endemic MERS-like virus would not decrease during the transition to endemicity,
as seen in Fig. 3B, and this is because disease
severity (and IFR) is high in children, the age
group expected to experience the bulk of
primary cases during the endemic phase. In
the endemic phase, a vaccination program
against MERS would therefore be necessary
to avoid excess mortality (fig. S11).
The key result from our model framework
that explicitly recognizes that functional Immunity to reinfection, disease, and shedding
is different is that, in contrast with infections
that are severe in childhood, SARS-CoV-2 could
join the ranks of mild, cold-causing endemic
HCoVs in the long run.
A critical prediction is that the severity of emergent HCoVs, once they reach endemicity, depends only on the severity of infection in children (Fig. 3) because all available evidence suggests that immunity to HCoVs has short IES and moderate IEI, leading to frequent reinfection throughout adulthood (11, 12), but strong IEP such that childhood infection protects from pathology upon reinfection in adulthood, as evidenced by the rarity of severe infections or detectable IgM titers in adults. Strain-specific virulence factors, such as the shared cellular receptor, angiotensin-converting enzyme 2 (ACE-2), to which SARS-CoV-1, SARS-CoV-2, and the endemic strain NL63 all bind (13–16), may affect the CFR during the emergence phase but have little impact on the severity of disease in the endemic phase. Because the four endemic HCoVs have been globally circulating for a long time and almost everyone is infected at a young age, we cannot ascertain how many pathologies would result from a primary or even a secondary case of any of these in an elderly or otherwise vulnerable person.
The key insights come from how our model explicitly incorporates different components of immunological protection concerning susceptibility, pathology, and infectivity (IES, IEP, and IEI, respectively) and their different rates of waning. In our analysis, we hypothesized that these components of immunity for SARS-CoV-2 are comparable to those of endemic HCoVs, and this needs to be determined. Additionally, during the transition to endemicity, we need to consider how the immune efficacies depend on primary and secondary infections across ages (17) and how responses differ between vaccination and natural infection.

Longitudinal analysis of SARS patients provides an opportunity to measure the durability of immune memory in the absence of reexposure. The only long-term study we know of that follows SARS-CoV-1–specific antibodies suggests that they wane faster than antibodies to other live viruses and vaccines such as measles, mumps, rubella, and smallpox (18) and fall below the threshold of detection in 6 years (19). In contrast to antibody responses, memory T cells persist for much longer periods (19, 20) and confer protection in animal model systems (21).
We further consider the effects of strain variation both for natural infection and vaccination. Strain variation and antibody escape may occur in endemic strains (22); however, the fact that symptoms are mild suggests that immunity induced by previously seen strains is nonetheless strong enough to prevent severe disease. Indeed, among HCoVs, frequent reinfections appear to boost immunity against related strains (12). However, the effect of strain variation may differ for vaccine-induced immunity, especially in light of the narrower epitope repertoire of many currently authorized vaccines.
If frequent boosting of immunity by ongoing virus circulation is required to maintain protection from pathology, then it may be best for the vaccine to mimic natural immunity insofar as preventing pathology without blocking ongoing virus circulation. Preliminary results suggest the adenovirus-based vaccine is better at preventing severe than mild or asymptomatic infections (23), and it will be important to collect similar data for the other vaccines. Should the vaccine cause a major reduction in transmission, it might be important to consider strategies that target delivery to older individuals for whom infection can cause higher morbidity and mortality, while allowing natural immunity and transmission to be maintained in younger individuals.
During the transition to endemicity, primary SARS-CoV-2 infections will frequently occur in older individuals, and we need to determine whether immunity induced by infection or vaccination in adulthood is similar to that produced by natural infections in childhood. Thus far, there have been few reinfections reported with SARSCoV-2, and disease severity has varied (24); the only population-level study of reinfection that we are aware of estimates a low rate of reinfection in the first 6 months after primary infection and mild disease upon reinfection (25), but further analysis and monitoring are vital.
The findings presented here suggest that using symptoms as a surveillance tool to curb the spread of SARS-CoV-2 will become more difficult, as milder reinfections increasingly contribute to chains of transmission and population-level attack rates. In addition, infection or vaccination may protect against disease but not provide the type of transmission-blocking immunity that allows for shielding (26) or the generation of long-term herd immunity (2).
The details of the change in overall IFR through the transient period will be affected by a wide array of factors, such as age-specific human contact rates (27) and susceptibility to infection (28) as well as improvement in treatment protocols, hospital capacity, and virus evolution. The qualitative result of mild disease in the endemic phase is robust to these complexities, but quantitative predictions for the transient phase will depend on careful consideration of these realities and how they interact with the dynamics of infection and components of immunity (29).

The changes in the IFR over time predicted by the model have implications for vaccination strategy against current and future emerging HCoVs. Social distancing and an effective vaccine are critical for control during a virgin epidemic and the transition out of it, but once we enter the endemic phase, mass vaccination may no longer be necessary. The necessity for continual vaccination will depend on the age dependence of the IFR. If primary infections of children are mild (as for SARS-CoV-1 and SARS-CoV-2), continued vaccination may not be needed as primary cases recede to mild childhood sniffles. If, on the other hand, primary infection in children is severe (as for MERS), then vaccination of children will need to be continued.
From an ecological and evolutionary perspective, our study opens the door to questions regarding the within-host and between-host dynamics of human immunity and pathogen populations in the face of immune efficacies with different kinetics. It also opens the question of how these immune efficacies interplay with strain cross-immunity, which is likely relevant within the alpha- and beta-coronaviruses. Considering data and model predictions from emergence through endemicity of HCoVs revealed a framework for understanding immunity and vaccination that may apply to a variety of infections, such as respiratory syncytial virus and seasonal influenza, which share similar age distributions and immune responses.
REFERENCES AND NOTES
1. S. Cobey, Science 368, 713–714 (2020).
2. H. E. Randolph, L. B. Barreiro, Immunity 52, 737–741 (2020).
3. C. Fraser, S. Riley, R. M. Anderson, N. M. Ferguson, Proc. Natl. Acad. Sci. U.S.A. 101, 6146–6151 (2004).
4. S. Su et al., Trends Microbiol. 24, 490–502 (2016).
5. M. Salamatbakhsh, K. Mobaraki, S. Sadeghimohammadi, J. Ahmadzadeh, BMC Public Health 19, 1523 (2019).
6. S. Ruan, Lancet Infect. Dis. 20, 630–631 (2020).
7. M. E. Halloran, I. M. Longini, C. J. Struchiner, Design and Analysis of Vaccine Studies (Statistics for Biology and Health, Springer, 2010).
8. K. A. Callow, H. F. Parry, M. Sergeant, D. A. J. Tyrrell, Epidemiol. Infect. 105, 435–446 (1990).
9. A. F. Bradburne, M. L. Bynoe, D. A. Tyrrell, BMJ 3, 767–769 (1967). 10. W. Zhou, W. Wang, H. Wang, R. Lu, W. Tan, BMC Infect. Dis. 13, 433 (2013).
For more information:1950477648nn@gmail.com






