New Insights Into The Immune Functions Of Podocytes: The Role Of Complement
Oct 13, 2023
Abstract
Podocytes are differentiated epithelial cells which play an essential role in ensuring a normal function of the glomerular filtration barrier (GFB). In addition to their adhesive properties in maintaining the integrity of the filtration barrier, they have other functions, such as the synthesis of components of the glomerular basement membrane (GBM), production of vascular endothelial growth factor (VEGF), release of inflammatory proteins, and expression of complement components. They also participate in the glomerular crosstalk through multiple signaling pathways, including endothelin-1, VEGF, transforming growth factor β (TGFβ), bone morphogenetic protein 7 (BMP-7), latent transforming growth factor β-binding protein 1 (LTBP1), and extracellular vesicles. A growing literature suggests that podocytes share many properties of innate and adaptive immunity, supporting a multifunctional role in ensuring a healthy glomerulus. As a consequence, the “immune podocyte” dysfunction is thought to be involved in the pathogenesis of several glomerular diseases, referred to as “podocytopathies.” Multiple factors like mechanical, oxidative, and/or immunologic stressors can induce cell injury.

Benefits of cistanche tubulosa-strengthen immune syste
The complement system, as part of both innate and adaptive immunity, can also define podocyte damage by several mechanisms, such as reactive oxygen species (ROS) generation, cytokine production, and endoplasmic reticulum stress, ultimately affecting the integrity of the cytoskeleton, with subsequent podocyte detachment from the GBM and onset of proteinuria. Interestingly, podocytes are found to be both source and target of complement-mediated injury. Podocytes express complement proteins which contribute to local complement activation. At the same time, they rely on several protective mechanisms to escape this damage. Podocytes express complement factor H (CFH), one of the main regulators of the complement cascade, as well as membrane-bound complement regulators like CD46 or membrane cofactor protein (MCP), CD55 or decay-accelerating factor (DAF), and CD59 or defensin. Further mechanisms, like autophagy or actin-based endocytosis, are also involved to ensure podocyte homeostasis and protection against injury. This review will provide an overview of the immune functions of podocytes and their response to immune-mediated injury, focusing on the pathogenic link between complement and podocyte damage.

cistanche supplement benefits-increase immunity
Click here to view Cistanche Enhance Immunity products
【Ask for more】 Email:cindy.xue@wecistanche.com / Whats App: 0086 18599088692 / Wechat: 18599088692
Keywords Podocyte, Complement, Immune system
Background
Podocytes are highly specialized epithelial cells of the glomerulus and represent a major component of the GFB [1]. They have a complex architecture including a large cell body facing the urinary space and an interdigitating network of extensions (primary and secondary processes) terminating as (tertiary) foot processes on the GBM [2]. Normal podocyte function is guaranteed by a sophisticated actin cytoskeleton, mainly localized within the foot processes [3]. Podocytes are characterized by a highly complex architecture regulated by multiple proteins, grouped into two main podocyte structures: the slit diaphragm (SD) and focal adhesions (FA). The SD is a unique highly specialized cell–cell junction between two podocyte foot processes (Fig. 1), including key proteins like nephrin, podocin, or synaptopodin [4, 5]. The SD represents not only a size-selective barrier to prevent filtration of large macromolecules but also a signaling platform with critical functions, such as regulation of the actin cytoskeleton and initiation of signaling pathways to modulate the plasticity of foot processes [6]. FA are complex structures that are able to connect the actin cytoskeleton of foot processes to the GBM, thanks to two main molecular components: integrins and GTPases. Besides contributing to the GFB, podocytes play important functions such as synthesis and repair of the GBM (together with endothelial cells), production of VEGF, and platelet-derived growth factor (PDGF) [6–9]. Moreover, a growing literature suggests that podocytes have many functions in the innate and adaptive immune systems [10–13]. They express cytokine and chemokine receptors to respond to a variety of soluble mediators. They are also able to synthesize inflammatory mediators, such as interleukin-1 (IL-1), which may contribute to local inflammation. Evidence in the literature suggests a possible role in the adaptive immune system too, as antigen-presenting cells (APC) initiate specific T-cell responses, like dendritic cells or macrophages [14, 15]. Furthermore, podocytes express several complement components, such as complement receptor type 1 (CR1) and type 2 (CR2) and complement regulators like CD46, CD55, or CD59, and they can produce complement proteins locally, including complement component 3 (C3) and CFH [16–18]. Nevertheless, the role of complement components expressed or secreted by podocytes in the regulation of the local complement reaction is not fully understood. Podocyte injury is involved in the pathophysiology of several glomerular diseases, like immune-complex glomerulonephritis, minimal change disease (MCD), focal segmental glomerulosclerosis (FSGS), and collapsing glomerulopathy [19, 20], and evidence from the literature suggests that the complement system could be primary or secondary involved in the podocyte damage [21–23].
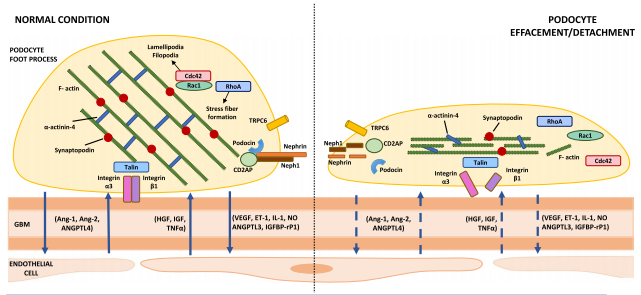
Fig. 1 Main components of the slit diaphragm and podocyte-endothelial cell cross-talk in healthy versus damaged podocytes. Podocyte slit diaphragm, glomerular basement membrane (GBM), and endothelial cells are the main components of the glomerular filtration barrier. Podocyte effacement/detachment, secondary to mechanical, oxidative, and/or immunologic triggers, is characterized by loss of silt diaphragm integrity, disruption of the actin cytoskeleton and focal adhesions, and interruption of the physiological podocyte-endothelial cell cross-talk (dashed arrows). Abbreviations: GBM, glomerular basement membrane; Ang, angiopoietin; ANGPTL, angiopoietin-like protein; IGF, insulin-like growth factor; IGFBP-rP1, insulin-like growth factor-binding protein-related protein 1; ET-1, endothelin-1; HGF, hepatocyte growth factor; IL-1, interleukin-1; NO, nitric oxide; TNF-a, tumor necrosis factor-a; VEGF, vascular endothelial growth factor
The immune podocyte: innate and adaptive functions

cistanche tubulosa-improve immune system
Increasing evidence suggests that podocytes play a role in the innate immune response because of their expression of Toll-like receptors (TLRs), especially TLR4, a subtype able to recognize bacterial lipopolysaccharide (LPS). Those receptors are upregulated in animal models of cryoglobulinemic membranoproliferative glomerulonephritis, and they could mediate glomerular damage by modulating the expression of chemokines [12]. TLRs are located on the cell surface or intracellularly and can be expressed by different types of cells, such as dendritic cells, macrophages and monocytes, fibroblasts, B and T cells, and endothelial and epithelial cells. They play an essential role in recognizing pathogen-associated molecular patterns; in particular, cell surface TLRs can mainly recognize microbial membrane components such as LPS, lipids, and proteins, while intracellular TLRs mainly recognize nucleic acids from bacteria and viruses [24]. In addition, TLRs can be activated by endogenous ligands released during stress or tissue injury, such as heat shock proteins, mRNA, and necrotic debris [25]. Cultured human podocytes constitutively express cell surface TLRs (i.e., TLR1, 2, 3, 4, 5, 6, and 10) [26], suggesting a possible role in the defense against microbial agents; however, de novo expression of intracellular TLRs subtype has also been reported in podocytes of patients with glomerular disease. In particular, puromycin aminonucleoside (PAN), commonly used to induce a nonimmune podocyte injury in vitro, can upregulate TLR9 intracellular expression and activate NF-κB and p38 MAPK in human immortalized podocytes, utilizing endogenous mtDNA as TLR9 ligand to facilitate podocyte apoptosis [27]. This would suggest a bivalent role of TLRs in podocytes, both as major players in response to foreign pathogens and mediators of podocyte damage. Moreover, podocytes can express MHC class I and II genes [28, 29], as well as B7-1 (or CD80, involved in T-cell activation) [15, 30] and FcRn (IgG and albumin transport receptor, used by podocytes to internalize IgG from the GBM) [31, 32]. In particular, MHC class II expression on podocytes is required for the development of immune-mediated renal injury, as MHC II presentation by podocytes is necessary to induce the CD4+T-cell-driven glomerular disease [14]. It is reported that these cells can act as antigen-presenting cells (APC), as they can express several macrophagic-associated markers [33, 34], and they are able to process antigens to initiate specific T-cell responses [15], supporting their multifunctional role in the immunological pathogenesis of glomerular diseases. Furthermore, the expression of functional chemokine receptors (CCR4, CCR8, CCR9, CCR10, CXCR1, CXCR3, CXCR4, and CXCR5) has been demonstrated in cultured human podocytes [35, 36]. Chemokines are small chemoattractant cytokines released by innate immune cells (i.e., neutrophils, eosinophils, macrophages, dendritic cells, natural killer cells), as well as endothelial and epithelial cells. Tey plays a central role in inflammation and immune cell recruitment by guiding circulating leukocytes to inflammation or damage sites [37, 38]. They also promote cell growth and tumor angiogenesis and are able to modulate apoptosis by binding G-protein-coupled receptors (GPCRs) on the surface of immune cells. Chemokine receptors are expressed in leukocytes, as well as non-hemopoietic cells, such as endothelial and epithelial cells [39].
CXCR1, CXCR3, and CXCR5 chemokine receptors have been identified in podocytes from kidney biopsies of patients with primary membranous nephropathy (PMN), while they were not expressed in healthy kidneys. Huber et al. suggested that podocyte CXCRs activation may contribute to GFB disruption and onset of proteinuria in PMN through hyperactivation of NADPH oxidases and oxygen radicals production [36]. Podocytes are involved in the inflammatory response of several human glomerulopathies, as suggested by their ability to produce pro-inflammatory cytokines like IL-1α and IL-1β [40, 41]. It has been reported that they can express inflammasome components, like NOD-like receptor (NLR) family proteins, which contribute to inflammatory response in the local kidney in primary glomerular diseases like lupus nephritis (LN) [42]. Podocytes are also known to secrete and/or express several complement proteins and regulators, suggesting local activation of the complement cascade. Expression of complement genes, including C1q, C1r, C2, C3, C3a receptor (C3aR), C5a receptor (C5aR), C7, CR1, and CR2, has been detected in cultured podocytes under normal physiological conditions, with increased local synthesis of complement proteins following podocyte injury [16, 17]. On the other side, complement regulators have been identified too, both membrane-bound (CD46, CD55, CD59) and soluble (CFI and CFH) forms. In particular, podocytes can express CFH locally to clear subendothelial immune complex deposits [43]. The fact that podocytes are able to produce complement components, including regulators, might have a relevant impact on podocytopathies where the complement system plays a pathogenic role. The balance between local complement activation and regulation is important to maintain the glomerular environment, as podocytes could become both target and source of injury, contributing to local complement activation and amplifying their own damage [44, 45].
Table 1 Summary of (potential and recognized) podocyte immune functions
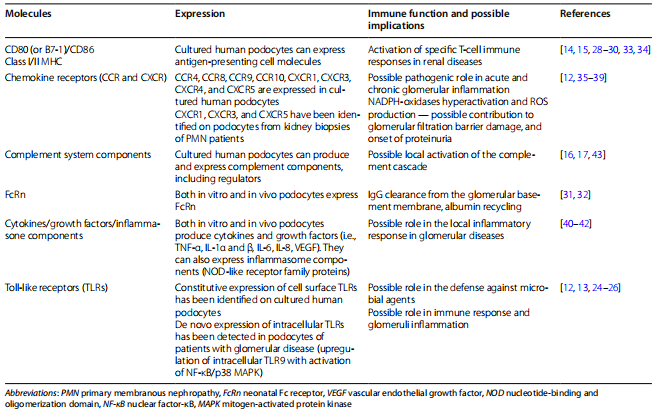
A summary of the main immune functions of podocytes is summarized in Table 1.
Podocyte and complement system
The complement system, classically described as part of the innate immune system, represents indeed a functional bridge between innate and adaptive immunity. It consists of more than 30 plasma or membrane-anchored proteins and regulators which play a role in inflammation, opsonization, and lysis of pathogens, clearance of apoptotic cells, and enhancement of both innate and adaptive immunity [46–48]. It can be activated by three different pathways, the classical, the lectin, and the alternative pathway [49, 50], which are tightly regulated by several complement components, like the membrane-bound proteins CD46, CD55, and CD59 and the soluble CFH, to prevent uncontrolled complement hyperactivation [51]. All three pathways induce a proteolytic cascade leading to a shared terminal pathway with subsequent membrane attack complex (MAC) assembly in the cell plasma membrane. Once inserted in the lipid bilayer, MAC forms a stable pore with~10 nm diameter generating several intracellular signals, which have been characterized by both in vivo and in vitro models as summarized in Table 2 [52].

cistanche supplement benefits-how to strengthen immune system
Sublytic effects of complement activation on podocyte
Mechanical, oxidative, and immunologic stress can cause podocyte damage and subsequently affect the integrity of the glomerular barrier. Complement activation with sublytic MAC formation on podocytes is an example of immunologic stress, which can trigger downstream pathways including protein kinases, lipid metabolism, cytokine production, ROS generation, growth factor signal transduction, endoplasmic reticulum stress, and the ubiquitin–proteasome system, eventually leading to disruption of the podocyte actin cytoskeleton and subsequent cell detachment [53]. More in detail, evidence suggests that a sublytic amount of MAC on the podocyte surface can induce calcium influx through the membrane pore, as well as calcium release from the intracellular storages, eventually leading to increased intracellular calcium which can activate multiple pathways, such as protein kinase signaling, and in particular protein kinases C (PKC) responsible for membrane vesiculation and internalization of MAC channels [52, 54–57], as suggested by reduction of MAC endocytosis by inhibiting PKC pathway [58]. It is well known that Ca2+ signaling in healthy podocytes is mainly mediated by angiotensin II and TRPC5 and 6 (nonselective cationic channels, downstream of angiotensin II signaling) [59]; interestingly, TRPC6 can play a dual role, as it has been shown that acute activation of this channel is able to protect podocytes from complement-mediated injury, while gain-of-function mutations/chronic hyperactivation can affect the SD and/or foot processes morphology leading to glomerular diseases, such as FSGS [60].
Table 2 Signalling pathways activated by MAC (adapted from Takano et al. (2013). Seminars in Nephrology. Reference [52]
![Table 2 Signalling pathways activated by MAC (adapted from Takano et al. (2013). Seminars in Nephrology. Reference [52] Table 2 Signalling pathways activated by MAC (adapted from Takano et al. (2013). Seminars in Nephrology. Reference [52]](/Content/uploads/2023842169/20231012123146b553f4d9ab504543a295fd3a4d4e2873.png)
induce transactivation of receptor tyrosine kinases at the plasma membrane of cultured podocytes, resulting in activation of the Ras-extracellular signal-regulated kinase (ERK) pathway and phospholipase C-γ1. Transactivated receptor tyrosine kinases could play a role in protein assembly and/or activation, inducing activation of downstream pathways, either dependently or independently of the increased cytosolic calcium levels [54, 61]. Other pathways activated by MAC formation on podocyte surface involve arachidonic acid (AA) release by cytosolic phospholipase A2-α (cPLA2), inducing AA metabolism to prostanoids, as described by Cybulsky et al. [62]. Eicosanoids can play a role in complement-mediated podocyte injury, as supported by experimental models of membranous nephropathy. Despite the exact mechanisms of glomerular damage are still unclear, cytotoxic consequences of cPLA2 activation could include the release of free fatty acids and lysophospholipids, as well as ions influx, which could ultimately affect the energy machinery [63].
ROS production has also been described in podocytes exposed to sublytic amounts of MAC; both cultured and in vivo podocytes express components of the NADPH oxidase, a complex enzyme able to reduce molecular oxygen to the superoxide anion, which is further metabolized to other ROS [52]. Lipid peroxidation and changes in the podocyte membrane composition, as well as in the GBM components, have been reported as a consequence of ROS production. Moreover, inhibition of ROS and/ or lipid peroxidation resulted in reduced proteinuria in animal models of membranous nephropathy, suggesting their pathogenic role in glomerular damage [64]. Endoplasmic reticulum (ER) stress with an accumulation of misfolded proteins and subsequent increase of the ubiquitin–proteasome system has been reported as an additional response to complement-mediated injury, as the possible protective response of podocytes to ongoing complement attack [65]. Sublytic MAC deposition on podocytes can also induce DNA damage, both in vitro and in vivo models, as demonstrated by Pippin et al. [66]. The authors also described that sublytic MAC-induced podocyte injury was associated with an increase in specific cell cycle-related genes, including p53, p21, growth-arrest DNA damage-45, and checkpoint kinase-1 and 2, leading to cell cycle arrest and podocyte growth suppression. This could explain why podocyte proliferation is limited following immune-mediated injury.
Consequences of complement activation on podocyte energy metabolism
The effects of complement activation on podocyte energy machinery are not fully understood. Brinkkoetter et al. demonstrated that podocyte metabolism is somewhat different from other types of cells, as it primarily relies on anaerobic glycolysis and the transformation of glucose to lactate [67]. More in detail, the authors showed a significantly lower mitochondrial density per cell area, compared to another type of renal cells (i.e., renal tubular cells). Also, glomeruli stained for mitochondrial enzyme superoxide dismutase 2 (SOD2) and the glycolytic enzyme pyruvate kinase M2 (PKM2) confirmed the perinuclear localization of mitochondria (and their almost complete absence in secondary and tertiary processes), while PKM2 was ubiquitous, suggesting podocyte processes as a large compartment of anaerobic glycolysis. They also used Tfam (mitochondrial transcription factor A) knockout mice to demonstrate that loss of mitochondrial transcription and lack of oxidative phosphorylation machinery do not induce podocyte disease. In addition, transient knockdown of Tfam in human podocytes significantly reduced mitochondrial respiration, while anaerobic glycolysis was significantly increased allowing a normal podocyte function. It has been demonstrated that sublytic complement-mediated injury induces reduction of intracellular ATP, in addition to reversible disruption of actin stress fibers and focal adhesions, mainly due to dephosphorylation (instead of degradation) of focal contact proteins, as described by Topham et al. using an in vitro model of rat podocytes [68]; however, the precise mechanisms need to be clarified. Also, complement activation on podocytes can cause nephrin dissociation from the actin cytoskeleton with disruption of the slit diaphragm, GFB damage, and subsequent onset of proteinuria, as suggested by the Heymann nephritis (HN) model [54, 61].
Complement‑mediated injury and podocyte response
Podocytes rely on several adaptive mechanisms to mitigate complement-mediated injury. Autophagy, a highly conserved mechanism of lysosome-mediated degradation of damaged organelles or nonfunctional proteins, is enhanced after sublytic complement damage in mouse podocytes, whereas its inhibition amplifies complement-mediated cell injury [69]. Liu et al. investigated the role of autophagy in PMN, comparing podocytes from PMN patients to cultured mouse podocytes exposed to sublytic complement activity, and they found impaired autophagy in podocytes from PMN patients, characterized by intracellular accumulation of p62 (a marker of impaired autophagy) and increase in autophagic vacuoles [70]. Podocyte-derived VEGF has also a bivalent function, as it is described that its overexpression can cause a collapsing glomerulopathy, while its inhibition is associated with GFB disruption, proteinuria, and possible development of thrombotic microangiopathy as well [71]. The putative mechanism is that, in normal conditions, VEGF signaling can regulate complement activity on podocytes and protect them from complement-mediated injury by increasing local CFH production, while its inhibition would provoke reduced levels of CFH, and podocytes would become more vulnerable to the injury.
More recently, new interesting mechanisms have been described to protect podocytes from injury, as reported by Medica et al. using a co-culture model of glomerular endothelial cells and podocytes. In particular, they demonstrated that extracellular vesicles derived from endothelial progenitor cells and involved in intercellular crosstalk (by transferring of proteins, lipids, and genetic material) are able to protect both glomerular endothelial cells and podocytes from complement (C5a)- and cytokine-mediated injury [72]. In particular, they showed that pre-stimulation of endothelial cells with extracellular vesicles prevented podocyte apoptosis and GFB disruption, and this protective effect could be mainly secondary to RNA transfer from the extracellular vesicles to damaged endothelial cells and podocytes. Despite tight surveillance of the complement system, including the activity of soluble and membrane-bound regulators, together with the protective mechanisms previously described to escape the injury, unrestricted complement activation can exceed those regulatory mechanisms, causing host tissue injury, as reported in various diseases including glomerulonephritis [73], hemolytic uremic syndrome (HUS) [74], sepsis [75], systemic lupus erythematosus [76], rheumatoid arthritis [77], organ transplant rejection [78], and age-related macular degeneration [79].
Summary and conclusions

cistanche plant-increasing immune system
Podocytes play a critical role in ensuring glomerular homeostasis. Over the years, growing literature highlighted the multiple and complex biological functions of these pericytes-like epithelial cells, which are much more than a supporting component of the GFB [1, 80–82]. Several authors described them as “immune podocytes,” to underline their properties as both innate and adaptive immune cells [10, 13, 15]. Understanding their complex biology is essential to unravel the pathogenic mechanisms of several glomerular diseases, where podocyte injury represents a common denominator.
The role of the complement system in podocyte injury has also been evaluated in a multitude of kidney disorders, such as membranous nephropathy, lupus nephritis, HUS, FSGS, and several more [45, 83–90]. The effects of complement activation on podocytes can vary based on the disease pathophysiology, as well as based on the initial trigger, which could induce lytic versus sub-lytic effects. Interestingly, podocytes have developed several protective mechanisms to escape the complement attack, such as autophagy, internalization mechanisms like endocytosis, and expression of complement regulators, and the balance between injury and defense mechanisms can ultimately determine the destiny of the podocyte cell [65, 69, 91]. Future studies, both in vitro and in vivo, are needed to better understand the role of complement activation in podocytopathies and the rationale for the use of anti-complement therapies in conditions where the complement system appears as the main driver of the disease.
References
1. Garg P (2018) A review of podocyte biology. Am J Nephrol 47(Suppl. 1):3–13
2. Miner JH (2011) Glomerular basement membrane composition and the filtration barrier. Pediatr Nephrol 26(9):1413–1417
3. Schell C, Huber TB (2017) The evolving complexity of the podocyte cytoskeleton. J Am Soc Nephrol 28(11):3166–3174
4. Kawachi H, Fukusumi Y (2020) New insight into podocyte slit diaphragm, a therapeutic target of proteinuria. Clin Exp Nephrol 24(3):193–204
5. Blaine J, Dylewski J (2020) Regulation of the actin cytoskeleton in podocytes. Cells 9(7):1700
6. Grahammer F, Schell C, Huber TB (2013) The podocyte slit diaphragm— from a thin grey line to a complex signaling hub. Nat Rev Nephrol 9(10):587–598
7. St. John PL, Abrahamson DR (2001) Glomerular endothelial cells and podocytes jointly synthesize laminin-1 and -11 chains. Kidney International. 60(3):1037–46
8. Greka A, Mundel P (2012) Cell biology and pathology of podocytes. Annu Rev Physiol 74(1):299–323
9. Byron A, Randles MJ, Humphries JD, Mironov A, Hamidi H, Harris S et al (2014) Glomerular cell cross-talk influences composition and assembly of extracellular matrix. J Am Soc Nephrol 25(5):953–966
10. Bhargava R, Tsokos GC (2019) The immune podocyte. Curr Opin Rheumatol 31(2):167–174
11. Mathieson PW (2003) What has the immune system got against the glomerular podocyte? Clin Exp Immunol 134(1):1–5
12. Banas MC, Banas B, Hudkins KL, Wietecha TA, Iyoda M, Bock E et al (2008) TLR4 links podocytes with the innate immune system to mediate glomerular injury. J Am Soc Nephrol 19(4):704–713
13. Xia H, Bao W, Shi S (2017) Innate immune activity in glomerular podocytes. Front Immunol 8:122
14. Goldwich A, Burkard M, Ölke M, Daniel C, Amann K, Hugo C et al (2013) Podocytes are nonhematopoietic professional antigen-presenting cells. J Am Soc Nephrol 24(6):906–916
15. Li S, Liu Y, He Y, Rong W, Zhang M, Li L et al (2020) Podocytes present antigen to activate specific T cell immune responses in inflammatory renal disease. J Pathol 252(2):165–177
16. Li X, Ding F, Zhang X, Li B, Ding J (2016) The expression profle of complement components in podocytes. Int J Mol Sci 17(4):471
17. Mühlig AK, Keir LS, Abt JC, Heidelbach HS, Horton R, Welsh GI, et al (2020) Podocytes produce and secrete functional complement C3 and complement factor H. Front Immunol 11:1833
18. Angeletti A, Cantarelli C, Petrosyan A, Andrighetto S, Budge K, D’Agati VD, et al (2020) Loss of decay-accelerating factor triggers podocyte injury and glomerulosclerosis. J Exp Med 217(9):e20191699
19. Kopp JB, Anders H-J, Susztak K, Podestà MA, Remuzzi G, Hildebrandt F, et al (2020) Podocytopathies. Nat Rev Dis Prim 6(1):68
20. Wiggins R-C (2007) The spectrum of podocytopathies: a unifying view of glomerular diseases. Kidney Int 71(12):1205–1214
21. Huang J, Cui Z, Gu Q-H, Zhang Y-M, Qu Z, Wang X et al (2020) Complement activation profile of patients with primary focal segmental glomerulosclerosis. PLoS ONE 15(6):e0234934
22. Couser WG (2012) Basic and translational concepts of immune-mediated glomerular diseases. J Am Soc Nephrol 23(3):381–399
23. Maillard N, Wyatt RJ, Julian BA, Kiryluk K, Gharavi A, Fremeaux-Bacchi V, et al (2015) Current understanding of the role of complement in IgA nephropathy. J Am Soc Nephrol 26(7):1503–1512
24. Kawasaki T, Kawai T (2014) Toll-like receptor signaling pathways. Front Immunol 5:461
25. Karikó K, Ni H, Capodici J, Lamphier M, Weissman D (2004) mRNA is an endogenous ligand for toll-like receptor 3. J Biol Chem 279(13):12542–12550
26. Srivastava T, Sharma M, Yew K-H, Sharma R, Duncan RS, Saleem MA et al (2013) LPS and PAN-induced podocyte injury in an in vitro model of minimal change disease: changes in TLR profile. J Cell Commun Signal 7(1):49–60
27. Bao W, Xia H, Liang Y, Ye Y, Lu Y, Xu X et al (2016) Toll-like receptor 9 can be activated by endogenous mitochondrial DNA to induce podocyte apoptosis. Sci Rep 6(1):22579
28. Coers W, Brouwer L, Vos JTWM, Chand A, Huitema S, Heeringa P et al (2008) Podocyte expression of MHC class I and II and intercellular adhesion molecule-1 (ICAM-1) in experimental pauci-immune crescentic glomerulonephritis. Clin Exp Immunol 98(2):279–286
29. Baudeau C, Delarue F, Hé CJ, Nguyen G, Adida C, Peraldi MN, et al (1994) Induction of MHC class II molecules HLA-DR, -DP and -DQ and ICAM 1 in human podocytes by gamma-interferon. Exp Nephrol 2(5):306–312
30. Reiser J, Von Gersdorf G, Loos M, Oh J, Asanuma K, Giardino L et al (2004) Induction of B7–1 in podocytes is associated with nephrotic syndrome. J Clin Investig 113(10):1390–1397
31. Akilesh S, Huber TB, Wu H, Wang G, Hartleben B, Kopp JB et al (2008) Podocytes use FcRn to clear IgG from the glomerular basement membrane. Proc Natl Acad Sci 105(3):967–972
32. Dylewski J, Dobrinskikh E, Lewis L, Tonsawan P, Miyazaki M, Jat PS, et al (2019) Differential trafficking of albumin and IgG facilitated by the neonatal Fc receptor in podocytes in vitro and in vivo. PLoS ONE 14(2):e0209732
33. Bariéty J, Nochy D, Mandet C, Jacquot C, Glotz D, Meyrier A (1998) Podocytes undergo phenotypic changes and express macrophagic associated markers in idiopathic collapsing glomerulopathy. Kidney Int 53(4):918–925
34. Mendrick DL, Kelly DM, Rennke HG (1991) Antigen processing and presentation by glomerular visceral epithelium in vitro. Kidney Int 39(1):71–78
35. Burt D, Salvidio G, Tarabra E, Barutta F, Pinach S, Dentelli P et al (2007) The monocyte chemoattractant protein-1/cognate CC chemokine receptor 2 system affects cell motility in cultured human podocytes. Am J Pathol 171(6):1789–1799
36. Huber TB, Reinhardt HC, Exner M, Burger JA, Kerjaschki D, Saleem MA et al (2002) Expression of functional CCR and CXCR chemokine receptors in podocytes. J Immunol 168(12):6244–6252
37. Charo IF, Ransohof RM (2006) The many roles of chemokines and chemokine receptors in inflammation. N Engl J Med 354(6):610–621
38. Proudfoot AEI (2002) Chemokine receptors: multifaceted therapeutic targets. Nat Rev Immunol 2(2):106–115
39. Grifth JW, Sokol CL, Luster AD (2014) Chemokines and chemokine receptors: positioning cells for host defense and immunity. Annu Rev Immunol 32(1):659–702
40. Niemir ZI, Stein H, Dworacki G, Mundel P, Koehl N, Koch B et al (1997) Podocytes are the major source of IL-1 alpha and IL-1 beta in human glomerulonephritides. Kidney Int 52(2):393–403
41. Wright RD, Beresford MW (2018) Podocytes contribute, and respond, to the inflammatory environment in lupus nephritis. American Journal of Physiology-Renal Physiology 315(6):F1683–F1694
42. Xiong W, Meng X-F, Zhang C (2020) Inflammasome activation in podocytes: a new mechanism of glomerular diseases. Infamm Res 69(8):731–743
43. Zoshima T, Hara S, Yamagishi M, Pastan I, Matsusaka T, Kawano M, et al (2019) Possible role of complement factor H in podocytes in clearing glomerular subendothelial immune complex deposits. Sci Rep 9(1):7857
44. Tipping PG (2008) Are podocytes passive or provocative in proteinuric glomerular pathology? J Am Soc Nephrol 19(4):651–653
45. Luo W, Olaru F, Miner JH, Beck LH, van der Vlag J, Thurman JM, et al (2018) Alternative pathway is essential for glomerular complement activation and proteinuria in a mouse model of membranous nephropathy. Front Immunol 9:1433
46. Dunkelberger JR, Song W-C (2010) Complement and its role in innate and adaptive immune responses. Cell Res 20(1):34–50
47. Mathern DR, Heeger PS (2015) Molecules great and small: the complement system. Clin J Am Soc Nephrol 10(9):1636–1650
48. Ricklin D, Hajishengallis G, Yang K, Lambris JD (2010) Complement: a key system for immune surveillance and homeostasis. Nat Immunol 11(9):785–797
49. Noris M, Remuzzi G (2013) Overview of complement activation and regulation. Semin Nephrol 33(6):479–492
50. Reis ES, Mastellos DC, Hajishengallis G, Lambris JD (2019) New insights into the immune functions of complement. Nat Rev Immunol 19(8):503–516
51. Zipfel PF, Skerka C (2009) Complement regulators and inhibitory proteins. Nat Rev Immunol 9(10):729–740
52. Takano T, Elimam H, Cybulsky AV (2013) Complement-mediated cellular injury. Semin Nephrol 33(6):586–601
53. Nagata M (2016) Podocyte injury and its consequences. Kidney Int 89(6):1221–1230
54. Cybulsky AV, Quigg RJ, Salant DJ (2005) Experimental membranous nephropathy redux. Am J Physiol Renal Physiol 289(4):F660–F671
55. Tegla CA, Cudrici C, Patel S, Trippe R, Rus V, Niculescu F et al (2011) Membrane attack by complement: the assembly and biology of terminal complement complexes. Immunol Res 51(1):45–60
56. Greka A, Mundel P (2011) Balancing calcium signals through TRPC5 and TRPC6 in podocytes. J Am Soc Nephrol 22(11):1969–1980
57. Cybulsky AV, Bonventre JV, Quigg RJ, Lieberthal W, Salant DJ (1990) Cytosolic calcium and protein kinase C reduce complement-mediated glomerular epithelial injury. Kidney Int 38(5):803–811
58. Carney DF, Lang TJ, Shin ML (1990) Multiple signal messengers generated by terminal complement complexes and their role in terminal complement complex elimination. J Immunol 145(2):623–629
59. Zhang L, Ji T, Wang Q, Meng K, Zhang R, Yang H et al (2017) Calcium sensing receptor stimulation in cultured glomerular podocytes induces TRPC6-dependent calcium entry and RhoA activation. Cell Physiol Biochem 43(5):1777–1789
60. Kistler AD, Singh G, Altintas MM, Yu H, Fernandez IC, Gu C et al (2013) Transient receptor potential channel 6 (TRPC6) protects podocytes during complement-mediated glomerular disease. J Biol Chem 288(51):36598–36609
61. Cybulsky AV (2011) Membranous nephropathy. Contrib Nephrol 169:107–125
62. Cybulsky AV, Takano T, Papillon J, Mctavish AJ (2000) Complement induced phospholipase A2 activation in experimental membranous nephropathy1 See Editorial by Shankland, p. 1204. Kidney Int. 57(3):1052–62
63. Bonventre JV (1992) Phospholipase A2 and signal transduction. J Am Soc Nephrol 3(2):128–150
64. Neale TJ, Ojha PP, Exner M, Poczewski H, Rüger B, Witztum JL et al (1994) Proteinuria in passive Heymann nephritis is associated with lipid peroxidation and formation of adducts on type IV collagen. J Clin Investig 94(4):1577–1584
65. Cybulsky AV (2013) The intersecting roles of endoplasmic reticulum stress, ubiquitin–proteasome system, and autophagy in the pathogenesis of proteinuric kidney disease. Kidney Int 84(1):25–33
66. Pippin JW, Durvasula R, Petermann A, Hiromura K, Couser WG, Shankland SJ (2003) DNA damage is a novel response to sublytic complement C5b–9–induced injury in podocytes. J Clin Invest. 111(6):877–85
67. Brinkkoetter PT, Bork T, Salou S, Liang W, Mizi A, Özel C, et al (2019) Anaerobic glycolysis maintains the glomerular filtration barrier independent of mitochondrial metabolism and dynamics. Cell Rep 27(5):1551–66.e5
68. Topham PS, Haydar SA, Kuphal R, Lightfoot JD, Salant DJ (1999) Complement-mediated injury reversibly disrupts glomerular epithelial cell actin microfilaments and focal adhesions. Kidney Int 55(5):1763–1775
69. Lv Q, Yang F, Chen K, Zhang Y (2016) Autophagy protects podocytes from sublytic complement-induced injury. Exp Cell Res 341(2):132–138
70. Liu WJ, Li Z-H, Chen X-C, Zhao X-L, Zhong Z, Yang C, et al (2017) Blockage of the lysosome-dependent autophagic pathway contributes to complement membrane attack complex-induced podocyte injury in idiopathic membranous nephropathy. Sci Rep 7(1):8643
71. Keir LS, Firth R, Aponik L, Feitelberg D, Sakimoto S, Aguilar E et al (2016) VEGF regulates local inhibitory complement proteins in the eye and kidney. J Clin Investig 127(1):199–214
72. Medica D, Franzen R, Stasi A, Castellano G, Migliori M, Panichi V et al (2021) Extracellular vesicles derived from endothelial progenitor cells protect human glomerular endothelial cells and podocytes from complement and cytokine-mediated injury. Cells 10(7):1675
73. Kaartinen K, Safa A, Kotha S, Ratti G, Meri S (2019) Complement dysregulation in glomerulonephritis. Semin Immunol 45:101331
74. Noris M, Mescia F, Remuzzi G (2012) STEC-HUS, atypical HUS, and TTP are all diseases of complement activation. Nat Rev Nephrol 8(11):622–633
75. Lupu F, Keshari RS, Lambris JD, Mark CK (2014) Crosstalk between the coagulation and complement systems in sepsis. Thromb Res 133:S28–S31
76. Birmingham DJ, Hebert LA (2015) The complement system in lupus nephritis. Semin Nephrol 35(5):444–454
77. Holers VM, Banda NK (2018) Complement in the initiation and evolution of rheumatoid arthritis. Front Immunol 9:1057
78. Stites E, Le Quintrec M, Thurman JM (2015) The complement system and antibody-mediated transplant rejection. J Immunol 195(12):5525–5531
79. McHarg S, Clark SJ, Day AJ, Bishop PN (2015) Age-related macular degeneration and the role of the complement system. Mol Immunol 67(1):43–50
80. Assady S, Wanner N, Skorecki KL, Huber TB (2017) New insights into podocyte biology in glomerular health and disease. J Am Soc Nephrol 28(6):1707–1715
81. Mundel P (2002) Podocyte biology and response to injury. J Am Soc Nephrol 13(12):3005–3015
82. Grahammer F (2017) New structural insights into podocyte biology. Cell Tissue Res 369(1):5–10
83. Zoja C, Buelli S, Morigi M (2019) Shiga toxin triggers endothelial and podocyte injury: the role of complement activation. Pediatr Nephrol 34(3):379–388
84. dos Santos M, Poletti PT, Milhoransa P, Monticielo OA, Veronese FV (2017) Unraveling the podocyte injury in lupus nephritis: clinical and experimental approaches. Semin Arthritis Rheum 46(5):632–641
85. Sakhi H, Moktef A, Bouachi K, Audard V, Hénique C, Remy P, et al (2019) Podocyte injury in lupus nephritis. J Clin Med 8(9):1340
86. Sharma M, Vignesh P, Tiewsoh K, Rawat A (2020) Revisiting the complement system in systemic lupus erythematosus. Expert Rev Clin Immunol 16(4):397–408
87. Bao L, Haas M, Quigg RJ (2011) Complement factor H deficiency accelerates the development of lupus nephritis. J Am Soc Nephrol 22(2):285–295
88. Ronco P, Debiec H (2020) Molecular pathogenesis of membranous nephropathy. Annu Rev Pathol 15:287–313
89. Ronco P, Plaisier E, Debiec H (2021) Advances in membranous nephropathy. J Clin Med 10(4):607
90. Cattran DC, Brenchley PE (2017) Membranous nephropathy: integrating basic science into improved clinical management. Kidney Int 91(3):566–574
91. Qi Y-Y, Zhou X-J, Cheng F-J, Hou P, Ren Y-L, Wang S-X et al (2018) Increased autophagy is cytoprotective against podocyte injury induced by antibody and interferon-α in lupus nephritis. Ann Rheum Dis 77(12):1799–1809






