NMR-Based Metabolomics Profiling For Radical Scavenging And Anti-Aging Properties Part 2
Jun 06, 2022
Please contact oscar.xiao@wecistanche.com for more information
2.5. Classification of Herb Extracts by Principal Component Analysis
The metabolite variation between the leaves of the tested herbs was further analyzed using multivariate data analysis (MVDA). Principal component analysis (PCA), a pattern recognition method, is an unsupervised MVDA that delivers a major understanding of the association between the samples. The PCA score plots showed the separation of herbs into clusters, whereas loading plots highlighted the metabolites that provide the separation [85,86]. The PCA model exhibited good fitness (R2X=0.997)and high predictability(Q2=0.993)where the variation between R2X(cum) and Q2(cum)was less than 0.3, thus indicating that each of the herbs extracts equally and evenly contributed to the observed group separation. This observation was in alignment with Wheelock et al. [87].

Please click here to know more
The principal component analysis score plot demonstrated that the selected herbs were separated into two clusters without any remarkable outliers as shown in Figure 4A. Principal component (PC)1 exhibited the greatest sample variation, then followed by PC2. PC1 and PC2 contributed to the percentage of variance at 29.7% and 24.9%, respectively. Therefore, total variance of about 54.6% was described by these PCs. Results from the loading column plot revealed the metabolites accountable for the separation of the herbs into the positive side of PC1(V.negundo and C.longa) and the negative side of PC1 (P. minus, P. indica, O.javanica and C. caudatus)(Figure 4B).


Data from the loading column plot of PC1 discovered that the phenolic compounds, mainly the flavonoid group and phenolic acids, were accountable for the separation of the selected herbs. Quercetin (1), quercetin-3-O-rhamnoside (2), quercetin-3-O-glucoside (3), quercetin-3-O-glucuronide (4), quercetin-3-O-arabinofuranoside (5), rutin (6), mvricetin derivatives (7), catechin (8),epicatechin (9), isorhamnetin (10),astragalin (11), chlorogenic acid (12), gallic acid (13), coumaric acid (14), ascorbic acid (15), formic acid (21), fumaric acid (22), 3-methylxanthine (26) and apigenin (28) contents were mostly higher in P. minus, P. indica, C. caudatus and O.javanica as they were situated in the negative side of the plot. In contrast, V.negundo and C.longa were differentiated from the other herbs by the presence of serotonin (27) and D-limonene (29) in their extracts.
2.6. Correlation between Bioactioities and the Metabolites Using Partial Least-Squares Analysis(PLS)
In order to comprehend the association between the measured bioactivities and metabolites found in the herbs tested, PLS, as a supervised MVDA methodology, was implemented to correlate the independent variables data (NMR chemical shift of the metabolites)to the data of dependent variables, which were the inhibition of the DPPH, ABTS and ORAC assays, as well as anti-elastase and anti-collagenase activities. This methodology was applied because PLS has a great achievement in order to link the tested bioactivities with metabolites, thus can provide a model for prediction [88]. Through the PLS analysis, the relationship between bioactivities such as radical scavenging activities and anti-aging properties with metabolites in the samples could be established. Hence, metabolites that were responsible as bioactive markers could then be suggested.
The biplot is a mixture of scores and loading plots resulting from the PLS analysis, as reported by Mediani et al. [80]. A partial least-squares biplot for the radical scavenging activity (Figure5A)and anti-aging properties(Figure 5B) showed that all samples were well-separated and clustered without notable outliers. The PC1 separated the leaves of P. minus, C.caudatus, and P. indica from V. negundo, O.jaoanica, and C.longa. Based on the radical scavenging activities and anti-aging properties of biplots, the model presented good fitness (R'Y) values of 0.988 and 0.966, respectively. Meanwhile, the predictability(Q4)for radical scavenging activities and anti-aging properties were at 0.984 and 0.951, respectively.
As shown from the PLS biplot of radical scavenging activities (Figure 5A), the bioactivities (DPPH, ABTS, and ORACassay)were directed to the positive side of the biplot, which was the most active areas and closest to P.minus, P. indica and C.caudatus. In contrast, V. negundo, O.javanica, and C. longa were directed to the negative sides of the biplot, which were considered the least active areas and were further from the DPPH, ABTS, and ORAC assays, and showed a negative correlation with bioactivities. In this situation, P. mimus, P. indica, and C.caudatus have clustered apart from the least active herbs, indicating that these herbs exhibited a stronger radical-scavenging effect, thereby suggesting that these herb extracts might have contained higher levels of phenolic compounds. Among the three most active herbs, P. minus was found to be more strongly correlated with the DPPH and ABTS assay, followed by the ORAC assay.
This finding was in agreement with the in vitro results of radical scavenging activities that had been performed. The results demonstrated that P. minus exhibited a potent free radical scavenging effect as compared to the other herbs. The results thus revealed that P. minus is the most active herb for the reactive oxygen species eradicator. Significant secondary metabolites that contributed to the radical scavenging activity of P. minus were identified as quercetin, quercetin-3-O-rhamnoside, catechin, isorhamnetin, astragalin, and apigenin. oteflavonoid All these metabolites were located closer to the P. minus and also to the DPPHand ABTS radical scavenging assays. A previous study by Mediani et al.[80] also discovered that a freeze-dried sample of plants showed a higher amount of α-glucose,β-glucose, catechin, and chlorogenic acid, which might have contributed to the potent DPPH radical scavenging capacity of the herb. However, the high radical scavenging effect of P.minus could also be contributed by the unidentified metabolites in the extract.

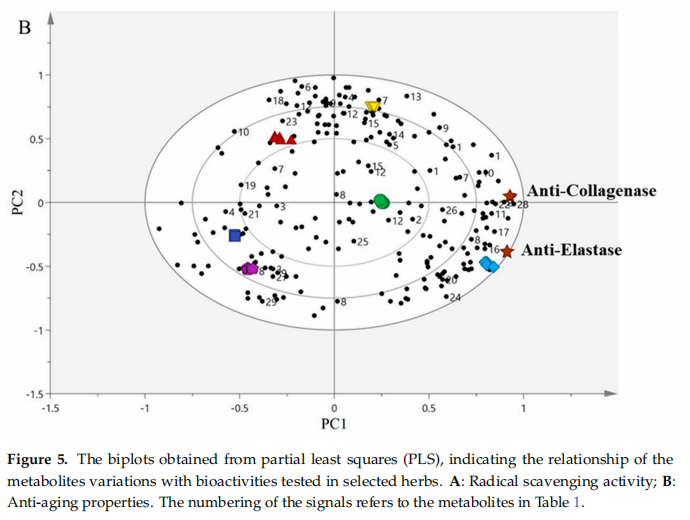
Similar findings were also found for the anti-aging properties. Figure 5B presents the biplot obtained from the PLS of anti-aging properties. The bioactivities (anti-elastase and anti-collagenase activities)were projected on the positive side of the biplot, which was the most active area and was closer to P.minus, P. indica, and C.caudatus. In contrast, V.negundo, O.javanica, and C.longa were directed to the negative side of the biplot, which was considered the least active area and was further away from the anti-elastase and anti-collagenase activities. This showed a negative or weaker correlation to the bioactivities. In this situation, P.mimus, P.indica, and C. caudatus were clustered apart from the least active herbs, indicating that these herb extracts exhibited greater elastase and collagenase inhibition. Among the three most active herbs, P. minus again was found to have a strong correlation with these anti-aging properties as compared to the other herbs.
This finding was in agreement with the in vitro results of anti-aging properties that had been performed earlier. The results revealed that P.minus was the most active herb for inhibiting elastase and collagenase enzymes. Significant secondary metabolites that contributed to the anti-aging properties of P. minus were identified as quercetin, quercetin-3-O-rhamnoside, myricetin derivatives, catechin, isorhamnetin, astragalin, and apigenin. Other metabolites such as α-glucose,β-glucose, fumaric acid, and fatty acid might also be responsible for the bioactivities. All of these metabolites were located closer to P. minus and to the anti-elastase and anti-collagenase activities.

Cistanche can anti-aging
The discrimination of these selected herbs was in agreement with the high radical scavenging activity samples especially. minus, P.India andC.caudatus, which were expected to be discriminated from the others due to their high concentration of secondary metabolites, especially flavonoids, The presence of these phenolic compounds is also believed to contribute to the greater elastase and collagenase inhibitory activities of these herbs [67-79,89-92].
The contribution of bioactive compounds of plant extracts toward free radical scavenging ability and anti-aging activity has been documented by various studies [66,72,75,79,92-101]. In addition, metabolites such as flavonoids (quercetin, kaempferol, myricetin, epicatechin, and catechin) and other phenols such as resveratrol and procyanidin B2, have been proven to significantly inhibit elastase and collagenase [69-71,79].
In the present study, the metabolite signals for variable importance in the projection(VIP) values were identified and reviewed to obtain the most significant metabolites that were correlated with the tested bioactivities. This was done to increase the integrity of the results presented here, which examined the discriminative potential of the identified metabolites. puritans vitamin c Generally, the metabolites signal with VIP>0.5 was taken into consideration to be significant for discrimination [102]. In this study, all of the metabolites contributing to the radical scavenging activities and anti-aging properties could be classified as significant discriminating metabolites since their VIP values were above than 1.0(Table 2). The two PLSbiplots model was validated using 100 random permutations, to confirm the validity (R2)and predictive(Q2) abilities of the original model with several models, comparative to the goodness of fit. The R2 illustrated that the model fitness was significant and explained the grade of Y variables in the model, whereas Q2 offered the model predictive quality similar to that reported by Eriksson et al. [103].

Generally, when the values of R2 and Q² are nearing 1, it reflects an improved presentation of the model in relation to goodness of fit and predictive quality [80]. In this study, R2 and Q2 values for both of the PLS models fell in the range of 0.951-0.988, which indicated outstanding goodness of fit(R2Y(cum)>0.8)and superior predictive ability (Q2(cum)>0.8). Results revealed that all the Y-axis intercepts of R< and Q- for the assays in radical scavenging activities and anti-aging properties were within the limits of R2<0.3 and Q2<0.05. sistanche The R2 and Q2 intercept values were in the range of 0.0367-0.0872 and-0.444 to-0.492, respectively, suggesting that both PLS models were valid and did not show overfit. Therefore, these two PLS models could be categorized as good performance models and these findings increased the reliability of the models.
2.7. Relative Quantification of Secondary Metabolites
The relative quantification of some of the secondary metabolites that had been identified from the selected herbs is shown in Figure 6. These metabolites were found mostly higher in the most active herbs such as P. minus, were located on the positive side of the biplots, and were closer to almost all the bioactivities tested. what is cistanche These results revealed that secondary metabolites especially from the flavonoid group of compounds that are present in high amounts in P. minus might have contributed to the free radical eradicator and anti-aging properties of this herb?

When comparing to the different structures of the flavonoid that could have contributed to their effectiveness, it was noted that the hydroxylation pattern in the B-ring might be one of the important factors for the inhibition effect of the metabolites on aging enzymes activity[80]. Sim et al.[104] also revealed that at both the protein and mRNA level, the inhibitory effect of these flavonoids became powerful with a growing number of groups in the B-ring, and they examined the structure-activity association of some flavonoids in on MMP-1 gene expression in UV-Airradiated human dermal fibroblasts.
3. Materials and Methods
3.1. Chemicals and Reagents
Deuterated methanol-d4(CH3OH-d4), non-deuterated KH2PO4, sodium deuterium oxide (NaOD), trimethyl silyl propionic acid-d4 sodium salt (TSP), ethanol, and methanol were supplied by Merck Millipore International (Darmstadt, Germany). Quercetin, phosphate buffer, 2,2-diphenyl-1-picrylhydrazyl (DPPH),2,2-azinobis(3-ethyl-benzothiazoline-6-sulfonic acid)[ABTS],Trolox,potassium persulfate,2,2'-azobis(2-amidinopropane)[AAPH, epigallocatechin gallate (EGCG),HEPES buffer, elastase enzymes,N-methoxy succinyl-Ala-Ala-Pro-Chloro,N-Methoxysuccinyl-Ala-Ala-Pro-Val-p-nitroanilide and deuterium oxide (D2O) were supplied by Sigma(Aldrich, Germany).

3.2. Plant Material and Sampling
Fresh leaves of O.jacanica, P. minus, and C.longa were collected from Felda Sungai Koyan Satu, Raub, and Pahang. V.negundo leaves were obtained from the Institute of Bioscience, Universiti Putra Malaysia. Fresh leaves of P. indica were harvested at University Agriculture Park, Universiti Putra Malaysia, and fresh leaves of C. caudatus leaves were collected at Agricultural Institute, Serdang, Selangor. A voucher specimen of these herbs was placed at the herbarium, Institute of Bioscience, Universiti Putra Malaysia, and each specimen was validated by the botanist. Anti aging cistanche All the leaves were harvested consistently in the morning, on sunny days to ensure the reliability of metabolites content. The plot at the open field for each herb was separated into six parts and each sample was collected from each segment as six replicates.
3.3.Sample Preparation
Once harvested, the fresh leaves were washed under running tap water to remove all residues, dried with laboratory tissue paper, and instantly frozen with liquid nitrogen to stop all the enzymatic reactions and preserved the metabolites prior to lyophilization. The samples were then dried in a LABCONCO(Kansas City, MO, USA) freeze dryer until consistent weight and moisture content reached below 10%. All the dried samples were ground using a laboratory blender to a fine powder and sieved using a laboratory test sieve(ENDECOTTS LTD.London, England) sized 300 um to obtain the uniform size. The powdered samples were vacuum-packed in an aluminum packaging to protect them from light exposure and humidity and stored at-80°C prior to analyses.
3.4. Extraction
The extraction procedure described by Mediani et al. [105] was followed with some modifications. Briefly, 10 g of each powdered sample were immersed in 100 mL 60% ethanol in an amber conical flask and sonicated for1 h using an ultrasonic bath sonicator (WiseClean, model WUC-D10H, Seoul, Korea) under controlled temperature (below 40°C). The samples were filtered through Whatman filter paper no.1 and the remains were re-extracted twice and filtered after the first extraction was completed. The extracts were then pooled and concentrated using a rotary evaporator under a vacuum at 40°C. The derived viscous substances were then freeze-dried using a LABCONCO freeze dryer to ensure the comprehensive elimination of water and stored at-80°C until further use. Lastly, the dried crude extracts were diluted to the necessary concentrations for all the investigations conducted.
3.5. DPPH Radical Scavenging Activity
The radical scavenging activity of the samples was determined by following the technique developed by Kong et al.[106], which was developed from a modified method of Brand-Williams et al. [107]with slight modification. Briefly, 50uL sample extracts in methanol at different concentrations(0 to 500 ug/mL) were added with195uLfreshly prepared0.2mMmethanolic2,2-diphenyl-1-picrylhydrazyl (DPPH)solution and stored. Alltestsamples were preparedin a 96 wellplate. Thedecolourizingprocess was recorded at 515 nm using a spectrophotometer (Biotek EL 800 microplate reader, Bio-Tek, Winooski, VT, USA) after 60 min incubation in darkness and compared to positive control and blank samples. The percentage of radical scavenging activity was measured according to the following equation:
![]()
where A control is the absorbance of control without plant extracts and Simple is the absorbance of plant extracts.
The plant extracts or positive control concentrations that scavenged 50% of stable free radical DPPH were calculated as the ICs using the linear graph of radical scavenging activity percentage against
plant extracts/positive control concentrations. Lower ICso indicated higher antioxidant activity. All experiments were run in six replicates with Trolox and quercetin as positive controls.
3.6.ABTS Radical Scavenging Assay
For ABTS radical scavenging assay, the analysis was conducted following the procedure described by Arnao et al.[108] with some amendments. The stock solutions prepared were 7 mM ABTS+ solution and 2.45 mM potassium persulfate solution. In order to prepare the working solution, the two stock solutions were mixed in the same quantities and left to react in the dark for 16 h at room temperature. Then, the working solution was diluted with distilled water to acquire an absorbance of 0.700±0.005 units at 734 nm using a spectrophotometer (UV-1650PC spectrophotometer, Shimadzu, Kyoto, Japan)and known as ABTS+ solution. The ABTS+ solution was freshly prepared for every assay. This ABTS+solution (900 μL) was allowed to react with 100 μL of herb extracts for 2 min. The absorbance was then read at 734 nm using the spectrophotometer. The standard curve comprising 3.1 ug/mL to and 50 ug/mL Trolox was developed, and the results were expressed as mg Trolox Equivalent Antioxidant Capacity/g sample (mg TEAC/g sample).
3.7. ORAC Radical Scavenging Assay
An ORAC assay to measure the peroxyl radical scavenging efficacy was implemented as stated by Huang et al. [109] using The FLUOstar OPTIMA microplate fluorescence reader (BMG LABTECH, Ortenberg, Germany). Each herb extract and Trolox(standard) were prepared in 75 mM phosphate buffer solution(PBS) pH 7.4. In 96-well black microplate, a total of 150 μL fluorescein (10 nM dissolved in PBS)was added followed by 25 μL of Trolox, plant extracts, or PBS as a blank. These solutions were pipetted in triplicate wells. The microplate was incubated for 15 min at 37 °C and covered with a lid. The fluorescence was measured with excitation wavelength at 458 nm and emission wavelength at 520 nm. The background signal was determined by taking the measurement every 90 s.
After that,25 uL freshly prepared 2,2'-azobis(2-amidinopropane)(AAPH,240 mM in PBS) was introduced by on-board injectors. The decay of fluorescence was then taken up to 90 min using the same excitation and emission wavelengths. The evaluation was done for the areas under the curve for samples (fluorescence versus time) minus the area under the curve for the blank and compared to a standard curve (25-400 μM Trolox). The ORAC values related to the Trolox were calculated using the equation as follows:
![]()
3.8.Elastase Inhibition Assay
The elastase inhibitory activity was determined according to the technique described by Krause et al.[110], with minor modifications by Ndlovu et al. [75]. The sample wells contained 25 μL 0.1 M HEPES buffer (pH7.5),25 μL herb extract (100 ug/mL), and 25 μL elastase enzyme (1 ug/mL). The blank wells only contained 75 μL HEPES buffer and the negative control wells contained 50 μL HEPES buffer and 25 μL elastase enzyme. The positive control wells contained 25 μL HEPES buffer,25 μL N-methoxy succinyl-Ala-Ala-Pro-Chloro(10 ug/mL), and 25μL of elastase enzyme. The solvent control wells contained 25 μL HEPES buffer,25 μL of 10% methanol, and 25 μL elastase enzyme. Blank controls for the herb extract(for color controls of every extract tested) contained 150 μL HEPES buffer and 25 uL of the herb extract. The micro-well plate was then incubated for20 min at room temperature. Following this,100μL substrate (N-Methoxysuccinyl-Ala-Ala-Pro-Val-p-nitroanilide,1 mM) was added. Then the plates were incubated for an additional 40 min at 25 ℃. Following incubation, the absorbance was read using a spectrophotometer(Biotek EL800microplate reader) at 405 nm. Percentage inhibition of the herb extracts was calculated using the equation as follows:
where A control is the absorbance of buffer with elastase and solvent and Atest is the absorbance of buffer, elastase, and herb extract or N-Methoxysuccinyl-Ala-Ala-Pro-Chloro.
3.9. Collagenase Inhibition Assay
The collagenase inhibition activity was conducted following the method of Van-Wart and Steinbrink [111] (1981) with modifications by Madrone et al. [92]. The test was implemented in 50 mM TES buffer (pH7.4 with 0.36 mM CaCl2). Collagenase enzyme from Clostridium histolyticum (ChC-EC.3.4.23.3) was prepared at the concentration of 0.8 units/mL (dissolved in TES buffer stock solution). The synthetic substrate N-[3-(2-furyl) acryloyl]-Leu-Gly-Pro-Ala (FALGPA) was prepared at a concentration of 2 mM in TES buffer stock solution. The total volume of 150 μL for the final reaction mixture contained 46.3 μL TESbuffer, 60 μL FALGPA (0.8 mM FALGPA final concentration),18.7 μL collagenase enzyme (0.1 units/mL final concentration), and 25μL herb extracts (100 μg/mL). Herb extracts and enzymes in TES buffer were incubated at room temperature for 15 min before adding the substrate to initiate the chemical reaction. Negative controls were carried out with a TES buffer. After adding the substrate, the absorbance was immediately read at 340 nm using a spectrophotometer (Benchmark Plus Microplate, Bio-Rad 170-6930, Bio-Rad, Hercules, CA, USA)in 96 well microtiter plates and continuously measured for another 20 min. Positive control was performed using epigallocatechin gallate(EGCG) at 12.5ug/mL. Percentage inhibition of samples was calculated using the equation as follows:

where A control is the absorbance of TES buffer and Atest is the absorbance of TES buffer, collagenase enzyme, and plant extract or FALGPA.
3.10.Metabolite Profiling Using 'H-NMR Measurement
The metabolite profiling using H-NMR of selected herbs was performed based on the protocol described by Kim et al. [47] with slight modifications. Crude herb extracts (25mg) were transferred into an mL microcentrifuge tube. A mixture of methanol-d4 and KHPO4 buffer in Dao (pH 6.0)containing 0.1% trimethyl silypropionic acid sodium salt (TSP) was added to the herb samples with a total volume of 0.7mL at (1:1)ratio. The microcentrifuge tubes containing herb extracts were then vortexed for 1 min at room temperature followed by ultrasonication for 15 min. Then, the mixture was centrifuged for 10 min at 5678g to separate the supernatant from any unsolvable materials. Next, 0.6mL clear supernatant was incorporated into NMR tubes and subjected to 'H-NMR analysis. The analysis of 'H-NMR was accomplished via a 500 MHz Varian INOVA NMR spectrometer (Varian Inc., Palo Alto, CA, USA)which operated at a frequency of 499.887 MHz and spectra were recorded at 26°C. Every single spectrum comprised 64 scans with 3.53 min of acquisition time and a width of 20 ppm. The data was analyzed using Chenomx software(v.6.2)(Clhenomx Inc, Edmonton, AB, Canada) to conduct phasing and baseline correction with a consistent setting. Multivariate data analysis was conducted using SIMCA software(version 13.0, Umetrics, Umea, Sweden). 3.11. Bucketing of 'H-NMR Spectra
Bucketingof H-NMR spectra were implemented by using Chenomxsoftware(v.6.2, Edmonton, AB, Canada). All spectra were binned from0.5-10.0 pprn region with the same parameters. The parameters included a spectral width of §0.04 which obtained a total of 245 integrated regions for each NMR spectrum. The chemical shift for water and residual methanol-d at δ4.70-4.90 and δ3.27-3.35 respectively were eliminated. The uniform binned data was then subjected to multivariate data analysis (MVDA).
3.12. Relative Quantification of Metabolites
The identified metabolites were examined for their relative quantification which was calculated based on the mean peak area of the signals after binning of LH-NMR spectra.
3.13. Statistical Analysis
All outcomes of six replicates were expressed as the means ± standard deviation. For the measurements of radical scavenging activities, inhibition of elastase and collagenase activities, and relative quantification of metabolites, statistical analyses were conducted using Minitab 16(Version 16, Minitab Inc., State College, PA, USA). Analyses of variance (ANOVA) were applied to examine for the significant differences between the means with p<0.05 considered as significantly different. Principal component analysis (PCA) and partial least square (PLS) from the multivariate data analysis (MVDA), were implemented using SIMCA-P software(v. 13.0, Umetrics, Umeå, Sweden)using the Pareto scaling method after the binning of NMR spectra was completed. The correlation between metabolites components and ICs0 values for DPPH radical scavenging activity was converted to 1/IC5so in order to acquire the same trend as the functional properties activity.

4. Conclusions
The application of H-NMR analyses coupled with MVDA was successful in investigating the variation in the metabolites of the selected herbs. The PCA score plot showed a distinct separation of the herbs according to their clusters. This study revealed that P. minus possessed the highest radical scavenging effect through DPPH and ABTS assays and exhibited the highest anti-aging activities. The two biplots of PLS analysis further validated this result, since a strong association was found between the metabolites identified in P. minus and the bioactivities tested. The active metabolites believed to contribute to the radical scavenging activities and anti-aging properties of P.minus have included quercetin, quercetin-3-O-rhamnoside, myricetin derivatives, catechin, isorhamnetin, astragalin, and apigenin. Therefore, it can be assumed that these metabolites were responsible for the potent radical scavenging effect and high anti-aging properties of P. minus. These metabolites were mainly from the flavonoids group, in particular flavonols. Therefore, it can be suggested that the metabolites from P. minus could be a promising free radical eradicator and aging enzymes inhibitor that can be used for delaying aging symptoms and for the treatment of aging-associated chronic diseases. These findings will aid to establish the potency of P. minus as a potential natural source of anti-aging agents and as a natural free radical eradicator.
This article is extracted from Molecules 2019, 24, 3208; doi:10.3390/molecules24173208 www.mdpi.com/journal/molecules






