Part Ⅱ Angiotensin II Receptor Blocker Associated With Less Outcome Risk in Patients With Acute Kidney Disease
May 05, 2023
Results
Clinical Characteristics of Patients
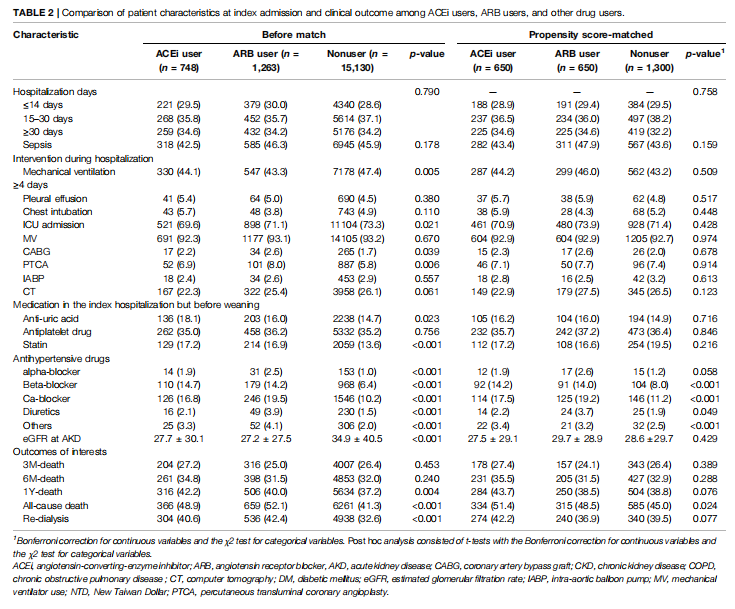
The study enrolled a total of 41,731 patients who underwent dialysis therapy during their AKI episodes within the study period, of whom 17,141 (41.1%) survival patients could be weaned off from dialysis for at least 7 days during the AKD period. Among them, there were 748 (4.36%) patients with prior ACEi use and 1,263 (7.37%) patients with prior ARB use (Figure 1). There were 116 (0.68%) patients with continuing ACEi use and 113 (0.66%) patients with continuing ARB use after weaning from AKI-D. During the AKD period, 100 new users took ARB, and 319 new users took ACEi.
The clinical characteristics of enrolled patients before index admission are shown in Table 1. The indication of RASi usage was mostly attributed to hypertension (n = 1711, 85.1%), followed by diabetes (n = 507, 25.2%), and prior cerebrovascular accident (266, 13.2%). The ACEi or ARB users before AKI-D had a higher Charlson comorbidity score (2.21 ± 2.58, 3.32 ± 2.57 vs.1.48 ± 2.23, p < 0.001) than nonusers. The levels of baseline eGFR were significantly lower in prior ACEi or ARB users (44.5 ± 37.7, 38.8 ± 41.5 vs. 50.1 ± 48.1 ml/min/1.73 m2, respectively, both p < 0.001) than those of the patients without prior ACEi or ARB.

Click here to get the benefits of Cistanche
During index hospitalization (Table 2), compared with the patients using ACEi or ARB, nonusers had significantly high ratios of using mechanical ventilation and ICU admission yet had a low ratio of receiving coronary artery bypass graft or percutaneous transluminal coronary angioplasty. The patients who were administered ACEi or ARB also were more likely to take other antihypertensive agents than nonusers during the AKD period. Additionally, compared with nonusers, ACEi and ARB users both had a high ratio of having been prescribed statins and anti-urate medications after dialysis withdrawal.
Risk of Mortality in ACEi Users, ARB Users, and ACEi/ARB Nonusers Among AKI-D Patients
The enrollees were divided into three groups: ACEi users (n = 650), ARB users (n= 650), and non-RASi users (n = 1,300) after propensity score matching (Figure 1; Supplementary Figure S1).
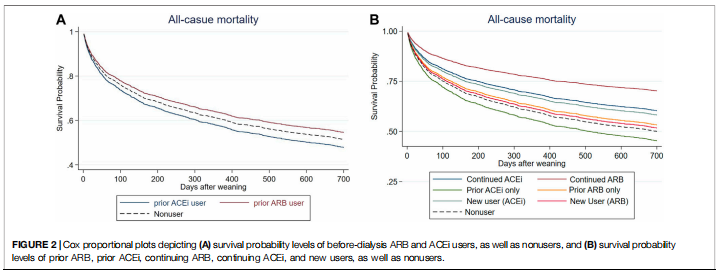
After a mean follow-up period of 1.01 ± 0.94 years (Figure 2A and Table 3), it was found that the patients who received ARB had a lower risk of all-cause mortality for post-AKI-D (adjusted hazard ratio (HR); 95% confidence interval (CI): 0.88; 0.77–1.00, p = 0.038) than non-RASi users by Cox proportional hazard analysis

for index dialysis) or continuing (both before and at the AKD period) RASi use to identify the effect of respective RASi. In a further analysis, compared with non-RASi users, patients who received continuing ARB after AKI-D had a significantly low risk for postweaning all-cause mortality (adjusted HR 0.51; 95% CI: 0.30–0.87, p = 0.013) (Figure 2B; Table 3). The beneficial HR for lower mortality in continuing ARB users showed persistent attenuation in a latency time-dependent manner from 90-days (HR, 0.20), 180-days (HR, 0.31), even to a mean follow-up of 1.23 (±1.06) years (HR, 0.51).
Hospitalization for MACE and MACE-Related Death Among ACEi/ARB Users
Regarding hospitalization for MACE, we did not identify a statistically significant protective effect for ARB relative to ACEi (adjusted HR 0.86; p = 0.145). The data did not present a statistically significant protective effect for ARB regarding MACE-related death, either (adjusted HR 0.83; p = 0.537).
Risk of Long-Term ESKD in Prior Users, Continuing Users, and Nonusers of ACEi/ARB
Prior ACEi users and prior ARB users did not have an increased risk of subsequent ESKD after using mortality as a competing risk factor compared to non-RASi users (Figure 3A, Supplementary Table S1). The patients who had continuing ACEi therapy after AKI-D could not decrease the risk of all-cause mortality and subsequent ESKD compared with the RASi nonusers even after adjusting kidney function at AKD and taking mortality as a competing risk factor (Table 3; Figure 3B).
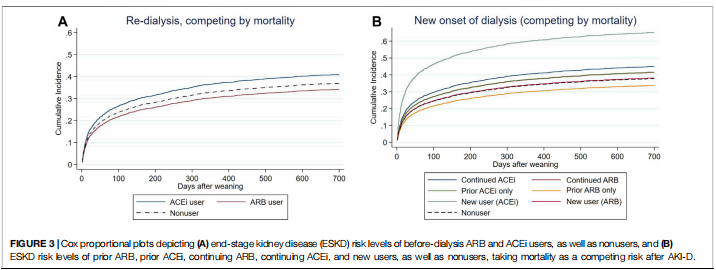
In the other compatible groups, those patients who had only prior/continuing ARB usage or only prior ACEi usage did not have an increased risk for re-dialysis following weaning from AKI-D in this matched COX proportional hazard model, taking mortality as a competing risk. However, the new users of ACEi during the AKD period had an increased risk of re-dialysis (adjusted sub-distribution hazard ratio (sHR) 2.22; 95% CI: 1.55–3.18, p = 0.037).
Sensitivity Analysis of ARB vs. ACEi
Regarding all-cause of mortality, pre-dialysis ARB users had a lower rate than ACEi users (HR, 0.824; p = 0.017). To compare differences between ACEi and ARB, we identified respective findings in the subgroup analysis (Figure 4). In patients with prior hypertension, diabetes, mechanical ventilation use, and cardiac intervention, risks of mortality were significantly lower in those taking ARB than those taking ACEi, before dialysis initiation.

Complication Analysis
We further analyzed the episodes of hyperkalemia, defined by serum potassium greater than 5.3 mmol/L (upper limit of the normal range), and found that continued use of both ARB (p = 0.070) and ACEi (p = 0.219) did not increase the risk of hyperkalemia after weaning from AKI-D. For each patient, we also collected data on eGFR in the 1 year following the withdrawal of dialysis and calculated the mean level. The mean eGFR values for ARB users, ACEi users, and nonusers are 32.7, 28.7, and 30.6, respectively. No statistically significant difference was detected (p = 0.103).
Negative Analysis
To attribute the possible health indication biases or unobserved confounders, we further identified the risk of new onset of gastrointestinal bleeding after ACEi or ARB usage. The patients with continued ACEi usage (p = 0.198) or ARB usage (p = 0.157) had similar risks of gastrointestinal bleeding after weaning from AKI-D.

Cistanche supplements
Discussion
This study is the first large population-based cohort study to investigate the role of ACEi versus ARB, respectively, in the long-term risks of mortality and re-dialysis in AKD patients weaned off from AKI-D. Nearly two-fifths of AKI-D patients could be weaned off from dialysis for at least 7 days. We showed that patients weaning from AKI-D and who had pre-AKID-continued ARB usage were associated with a lower risk of long-term all-cause mortality. Further analysis showed the benefit of ARB mainly stemmed from continuing ARB usage, but those who had continuing ACEi usage did not decrease their mortality risk. During a mean follow-up period of 1.01 years after being weaned off from AKI-D, patients with de novo ACEi usage had a higher risk of re-dialysis than those without ACEi/ARB usage, even after adjustment of their kidney function recorded at AKD. We also demonstrated that there was not an increased risk of hyperkalemia associated with using ACEi/ARB after weaning from AKI-D.
ACEi and ARB Are Different in Clinical Scenarios
In real-world practice, discontinuation of ACEi/ARB during admission and/or acute illness is common, particularly in patients with AKI-D. Both ACEi and ARB are RASi and have traditionally been considered to have similar clinical effects. Indeed, when treating patients with hypertension, heart failure, diabetes, cardiovascular disease, and chronic kidney disease, head-to-head studies revealed that antihypertensive efficacy and various clinical outcomes between ACEi and ARB were comparable (15). However, our current study provided the first evidence that their effects on reducing the risk of all-cause mortality after weaning from AKI-D may be different at the AKI to CKD transition.
We found that AKD patients who had continuing ARB usage had a lower risk of all-cause mortality, whereas there was no increased risk of re-dialysis after AKI-D/AKD. However, the mortality rate in continuing ACEi users was not remarkably lower than that in non-RASi users. Since the transition from AKI to AKD and CKD is an interconnected syndrome, our study provides a novel insight into the feasibility of various pharmacological therapies influencing the post-AKI care and even the prognosis of these patients (Ostermann et al., 2017).
In some clinical scenarios, ARB could provide better protection than ACEi does. For example, in the REACH cohort, a real-world practice, ARB was superior to ACEi in reducing cardiovascular events among high-risk patients (Potier et al., 2017); moreover, in patients who underwent coronary artery bypass grafting (CABG), the incidence of major cardiovascular events was significantly lower in ARB users during their 12-month follow-up period (Kim et al., 2020). In addition to cardiovascular protection, ARB was associated with lower rates of sepsis than ACEi in patients with chronic obstructive pulmonary disease (Lai et al., 2019); furthermore, patients who were administered ARB, rather than ACEi, had lower rates of hospitalization for sepsis, than untreated patients (Dial et al., 2014).
According to a secondary analysis of the RENAL study in critically ill patients with AKI, ACEi administration during the follow-up period was infrequent and was not associated with a statistically significant impact on patient survival (Wang et al., 2014). A previous study found that the use of RASi did not show higher rates of ESKD possibly because the deteriorated effects of ACEi could be ameliorated by ARB use (Brar et al., 2018). However, in this study, we noticed a higher risk of ESKD, as a solid outcome of kidney events in patients who were added with either de novo RAS inhibitors when they were weaned off from AKI-D.

Cistanche tubulosa
ACEi and ARB Are Different in Pharmacological Mechanisms
In patients administered with ARB or ACEi, plasma angiotensin II levels were augmented in the ARB group but no similar findings were noticed in the ACEi group (Nakamura et al., 2009); thus, ARB might facilitate the effects of angiotensin II type 2 receptor (AT2R)-mediated responses to the increased levels of angiotensin II. The anti-inflammatory effect of ARB could be more potent than that of ACEis; for example, ramipril, an ACEi, increases IL-1β and IL-10 in patients with kidney diseases (Gamboa et al., 2012). ACEi increases the plasma levels of asymmetric dimethylarginine (ADMA), an endogenous inhibitor of nitric oxide synthases as compared with valsartan, an ARB (Gamboa et al., 2015).
ACE2 expression is abundant in the kidney and is thought to protect against kidney injury (Williams and Scholey, 2018). Preclinical analysis showed inconsistent findings regarding the effects of RASi on ACE2 expression (Ferrario et al., 2005). ACEi could decrease (Hamming et al., 2008) or did not affect the activity of ACE2 (Rice et al., 2004), while ARB has been shown to increase urinary ACE2 secretion in hypertensive patients, which indicates that upregulation of ACE2 may be present in humans (Furuhashi et al., 2015). ARB could play a key role by increasing expressions of ACE2 and plasma angiotensin-(1–7) in animals and humans, thus modifying processes associated with acute inflammation and inhibiting leukocyte activation and recruitment (Simoes e Silva et al., 2013). Increased levels of angiotensin II occurring after ARB treatment, but not after ACEi, would increase substrate load on ACE2, thus leading to its upregulation (Esler and Esler, 2020). In light of our findings, continuing (reinitiating) ARB use, but neither adding new ACEi or ARB in RASi-naïve patients nor continuing (re-initiating) ACEi use after AKI-D patients weaned off from AKI-D, might be helpful to decrease subsequent all-cause mortality.
Association of Use of ACEi and ARB With Hyperkalemia
Hyperkalemia is a possible caveat of the RASi prescription. However, we found that patients given either ARB or ACEi did not have an increased risk of hyperkalemia following the AKI-D.
Study Strengths and Limitations
It is the first large-scale study to investigate the individual impact of ACEi versus ARB in patients after weaning from AKI-D via utilization of a well-maintained high-quality population cohort, in which the selection bias could be reasonably minimized. Additionally, we had a longer follow-up period than that was usually reported in clinical trials and thus enabled the evaluation of longer-term risks and benefits of RASi therapy in real-world practice.
Our study also has some limitations. First, our study was observational; therefore, the associations were not prospective, and strong causality cannot be inferred. Some important covariates, such as blood pressure, urine output, and body mass index after discharge were not available in our cohort. The observational nature of this study was an intrinsic and unavoidable limitation because the lack of randomization precluded a definite investigation of treatment advantages. Second, the treating physicians at the time of prescribing the medication had already made a risk assessment and decided that the benefits of ACEi or ARB outweighed the potential nephrotoxicity. Some patients could present AKI related to RAS inhibitors. However, we found that patients continuing the use of ACEis or ARBs among AKD patients did not have an increased risk of re-dialysis. Although eGFR dip has a strong association with subsequent progression to end-stage kidney disease (Khan et al., 2022), the current guideline illustrated “permissive kidney injury,” in terms of less than 30% increase in creatinine after initial use of ACEi or ARB (Kidney Disease, 2020).
However, it is very challenging to perform a randomized controlled trial because ACEi and ARB are standard therapeutic agents and widely used in treating hypertension, diabetic kidney disease, and congestive heart failure. Moreover, using the validated outcome of gastrointestinal bleeding that was not interfered with by ACEi/ARB, we could confirm that the selection bias was minimal, if any, from our study design. Additionally, the disease severity score after propensity score matching is similar between the study groups. Actually, regarding kidney function at baseline and the AKD period after weaning, ARB users in this study had the lowest eGFR than others before matching. Even though the ratios of comorbidities (diabetes, cerebrovascular disease, and myocardial infarction) were more severe in ARB users in our cohort, we still noticed the significant survival benefit of ARB usage.
Cistanche pills
Prospective
Given the better outcome efficacy but fewer adverse events with ARB (Messerli et al., 2018), risk-to-benefit analysis in aggregate indicates that at present there is enough evidence to support that prior or continuing ARB usage for managing the patients who could be weaned off from AKI-D. Our findings support the hypothesis that ARB may stabilize kidney outcomes and reduce mortality among patients who were weaned off from AKI-D and thus suggest the use of ARB even in the advanced stage of CKD.
In the future, prospective head-to-head comparison trials are the only ironclad way to compare the efficacy and safety of ARB objectively and to test whether the ARB outcome “paradox” really holds ironclad.
Conclusion
In conclusion, our present study revealed that prior and continuing ARB usage was associated with a lower risk of mortality after AKI-D patients were weaned off dialysis, while the use of ACEi did not have survival benefits. New users of ACEi among these AKD patients had a higher risk of re-dialysis after mortality was controlled as a competing risk. The use of ACEi or ARB during the AKD period did not increase the risk of hyperkalemia. Further prospective randomized studies are needed to verify our findings.
How to use Cistanche extract to help reduce the risks associated with acute kidney disease in patients?
Cistanche extract, a traditional medicinal herb, has been shown to exhibit the potential in reducing the risks associated with acute kidney disease (AKD). AKD is a common and severe medical condition characterized by a sudden loss of kidney function, leading to an increased morbidity rate if left untreated. The bioactive compounds in Cistanche extract have been proven to possess anti-inflammatory, antioxidant, and immunomodulatory effects, making it a promising treatment option for AKD patients. To use Cistanche extract as a supplement to reduce the risks associated with AKD, it is recommended to take it orally in an appropriate dosage, usually in the form of capsules or powder. It is important to consult with a healthcare professional before taking any herbal supplements, as they may interact with other medications and cause unwanted side effects. Additionally, maintaining a healthy lifestyle, such as following a balanced diet, staying hydrated, and avoiding smoking and alcohol consumption, can also help reduce the risks associated with AKD. In summary, while Cistanche extract shows promise in reducing the risks associated with AKD, individuals should still consult with their healthcare provider and adopt healthy lifestyle habits to optimize their overall health.
Vin-Cent Wu 1 , Yu-Feng Lin 1 , Nai-Chi Teng 2 , Shao-Yu Yang 1 , Nai-Kuan Chou 3 , Chun-Hao Tsao 3 , Yung-Ming Chen 1 , Jeff S Chueh 4,5 and Likwang Chen 2
1. Department of Internal Medicine, National Taiwan University Hospital, Taipei, Taiwan,
2. Institute of Population Health Sciences, National Health Research Institutes, Miaoli, Taiwan,
3. Department of Surgery, National Taiwan University Hospital, Taipei, Taiwan,
4. Glickman Urological and Kidney Institute, Cleveland Clinic Lerner College of Medicine, Cleveland Clinic, Cleveland, OH, United States,
5. Department of Urology, National Taiwan University Hospital, Taipei, Taiwan






