PART Ⅱ: Effect Of Cistanche Tubulosa Extracts On Male Reproductive Function in Streptozotocin-Nicotinamide-Induced Diabetic Rats
Mar 04, 2022
Contact: Audrey Hu Whatsapp/hp: 0086 13880143964 Email: audrey.hu@wecistanche.com
Zwe-Ling Kong ® Athira Johnson® Fan-Chi Ko, Jia-Ling He and Shu-Chun Cheng
Department of Food Science, National Taiwan Ocean University, Pei-Ning Road, Keelung 20224, Taiwan; athirajohnson07@gmail.com (A.J.); makeadrea@hotmail.com (F.-C.K.); k46255@gmail.com (J.-L.H.); riceroll826@gmail.com (S.-C.C.)
* Correspondence: kongzl@mail.ntou.edu.tw; Tel.: +886-2-2462-2192; Fax:+886-2-2463-4203
Abstract: Diabetes is a chronic disorder characterized by hyperglycemia due to decreased levels of insulin or the inefficiency of the tissue to use it effectively. Infertility is known as a major outcome of diabetes and affects the male reproductive system by causing sperm impairment and gonadal dysfunction. Cistanche tubulosa is a parasitic plant that has the capacity to improve memory, immunity, and sexual ability, reduce impotence, and minimize constipation. This study was focused on the investigation of the anti-inflammatory and protective effects of echinacoside (ECH) in Cistanche tubulosa extract (CTE) on the male reproductive system of diabetic rats. The antioxidant, anti-inflammatory, and protective effects of CTE were evaluated by both in vitro and in vivo methods. The in vitro results show that the ECH inhibited reactive oxygen species (ROS) production and improved StAR, CYP11A1, CYP17A1, and HSD17p3 protein expression. The in vivo analysis was carried out with three doses of echinacoside (ECH) (80, 160, and 320 mg/kg) in CTE. In total, 0.571 mg/kg of rosiglitazone (RSG) was administered as a positive control. Diabetes was induced by streptozotocin (STZ) (65 mg/kg) and nicotinamide (230 mg/kg) in combination with a high-fat diet (45%). The in vivo studies confirmed that the ECH improved blood sugar levels, insulin resistance, leptin resistance, and lipid peroxidation. It can restore kisspeptin 1 (KiSS1), G protein-coupled receptor GPR 54, suppressor of cytokine signaling 3 (SOCS-3), and sirtuin 1 (SIRT1) messenger ribonucleic acid (mRNA) expression in the hypothalamus and recover sex hormone level. Thus, this study confirmed the antioxidant, anti-inflammatory, and steroidogenesis effects of CTE.
Keywords: diabetes; infertility; Cistanche tubulosa extract (CTE); echinacoside (ECH); anti-inflammatory activity; antioxidant activity; steroidogenesis effects
Protective effects of echinacoside (ECH) from Cistanche tubulosa
3. Results
3.1.In Vitro Analysis
3.1.1.Comparison of Antioxidant Activities of ECH, CTE, and RES
Long-term high oxidative stress and chronic inflammation are the main causes of diabetic complications [16]. CTEs contain various phenylethanoid glycosides such as echinacoside (ECH) and acteoside, with the potential for antioxidant activity [17]. DPPH radical scavenging assay was conducted for evaluating the antioxidant activity of ECH. Trolox and resveratrol (3,5,4'-trihydroxystilbene, RES) were taken as the standard and positive control respectively. As shown in Figure 1, ECH showed better radical scavenging activity and the activity was significantly higher than that of the positive control (RES).
3.1.2.Cell Viability of ECH on LC-540 and TM3 Leydig Cells
MTT assay was performed for evaluating the cell viability of different concentrations of ECH. LC-540 Leydig cells and TM3 Leydig cells showed more than 80% of viability after being treated with ECH (Figure 2). As compared to the other concentrations, a 100-|dM concentration (dilutions of 100 rM in 2% FBS) of ECtf showed better cell viability in ffM3 Leydig cells. The result indicated that the increased concentration of ECH does not contribute to any significant toxicity to the cells.

3.1.3.Effect of ECH on AGE-Induced Superoxide Production by NBT Assay in LC-540 and TM3 Leyd ig Cells
AGEs cause oxidative damage in the body due to the oxidation of glucose and the formation of free radicals such as O2-, hydroxyl radicals, and carbonyl groups [18]. After being stimulated with 50 Rg/mL of AGEs, the LC-540 (Figure 3a) and TM3 (Figure 3b) c ells were treated with ECH and RES (5 /M and 10 tsp each). The results showed that the production of superoxide anion was increased in the control group (stimulated AGOs), but the production of superoxide anion was decreased after treatment with ECH and RES. From Fig ure 3, ii was understood that the ECH and RES reduce the level of superoxide production and protect the cells from oxidative damage. In the case of LC-540 cells, 10 ECH showed almost similar activity with the normal group. The superoxide production in both cells was decreased with an increase in the concentration of both ECH and RES.
3.1.4.Effect of ECH on H2O2 Production in AGE-Stimulated LC-540 Leydig Cells
DCFH-DA assay was executed for detecting the presence of oxidative species. After entering into the cells, the fluorescent dye DCFH-DA is oxidized by intracellular H2O2 and forms dichlorofluorescein (DCF). As shown in Figure 4, there was a significant increase in intracellular H2O2 production in the AGE-stimulated group (control) whereas the addition of 10 ECH and RES significantly reduced the H2O2 production in cells. Administration of 10 ECH resulted in only about 47.1% of H2O2 production, significantly lower than in the control group.
3.1.5. Effect of ECH on RAGE and NF-kB Protein Expression Levels in AGE-Stimulated LC-540 Leydig Cells
Western blot analysis was performed to confirm the presence of RAGE in LC-540 Leydig cells. The effect of ECH on RAGE (Figure 5a) and NF-kB (Figure 5b) protein expression levels in AGEs-stimulated Leydig cells were shown in Figure 5. The results showed that 50 /mL concentration of AGEs induced higher RAGE and NF-kB expression in LC-540 Leydig cells and 10 concentrations of ECH and RES has significantly reduced the expression of RAGE and NF-kB. The expression of NF-kB was significantly lower in RES and ECH than in RAGE antagonists. So, the results confirmed that the ECH reduced the level of inflammation by decreasing the level of RAGE and NF-kB.
3.1.6. Effect of ECH on the Testosterone Synthesis pathway in AGE-Stimulated LC-540 Leydig cells.
The process of spermatogenesis and male infertility depends upon the presence of testosterone. As shown in Figure 6, the expressions of StAR (Figure 6a), CYP11A1 (Figure 6b), CYP17A1 (Figure 6c), and HSD17p3 (Figure 6d) proteins were significantly decreased in AGE-stimulated LC-540 Leydig cells (control group). The expressions of StAR, CYP11A1, CYP17A1 and HSD17p3 proteins were significantly increased when the RAGE antagonist, RES, and ECH were a dded. The increased expression of StAR and CYP11A1 proteins were seen in both the ECH- and RES-treated groups. The expression level of CYP17A1 was almost similar in RES and ECH groups. The expressions of the HSD17&3 protein in ECH-treated Leydig cells were much higher than those of the RES and RAGE antagonist-treated cells. Increased expression of StAR, CYP11A1, CYP17A1, and HSD17p3 proteins indicated the normal production of testosterone.
3.2.In Vivo Analysis
3.2.1. Effects of CTE on Body Weight and Calorie Intake
After 6 weeks of experimentation, the diabetic group (HFD-DM) showed higher body weight than the control group. HFD-DME4 group showed a lower body weight than the HFD-DM and HFD-DMER groups (Figure 7a). The calorie intake of the control group was significantly lower than in the other groups. There was no significant difference between the calorie intakes of the other five groups (Figure 7b).
3.2.2.Oral Glucose Tolerance Test (OGTT) to Determine the Success) 1 Inductien of Diabetes
The oral glucose tolerance test (OGTT) is used as a promising tool for detecting diabetic Mellitus. An increased level of glucose in the blood indicates a diabetic condition. As shown in Figure 8a, the plasma glucose level was lower in CTE groups than in the DM group at 0, 30, 90, and 120 min. Further, the area under the curve (AUC) of plasma glucose concentration showed that in CTE and RSG groups.3.2.3.Total Plasma Glucose, Cholesterol, and Triglyceride Contents
The plasma fasting blood glucose level was higher in the DM group and lower in the DME2 group (except control) than others. There was no significant difference in the total cholesterol between the groups except for the DME4 group. In the DME4 group, the cholesterol level was lower than the others. The level of triglycerides was higher in DM group and lower in DME4 group and the triglyceride content was decreased with increase in the concentration of CTE (Table 2). The results show that the level of plasma glucose, cholesterol, and triglycerides was higher in DM group and the level was significantly decreased on treatment with CTE.
3.2.4.Plasma Insulin Levels, Plasma Leptin Level, and Homeostasis Model Assessment-Insulin Resistance (HOMA-IR) Values
The levels of plasma insulin, leptin, and HOMA-IR values are shown in Table 3. The DM group had higher plasma insulin and plasma leptin levels than the control group. The HOMA-IR index was also significantly higher in the DM group. The plasma insulin, leptin, and HOMA-IR values were decreased with an increase in the concentration of CTE. Plasma leptin was significantly reduced in CTE groups but the RSG drug group (DMR) did not show any significant difference from the DM group.
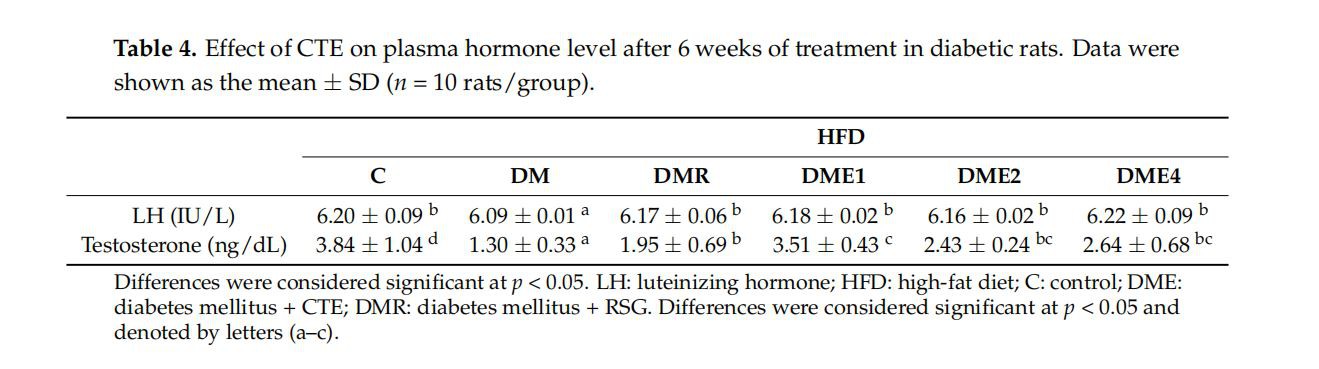
3.2.5.Effect of CTE on Plasma LH and Testosterone Levels in Diabetic Rats
As shown in Table 4, the concentrations of testosterone in diabetic rats (DM) were significantly decreased, while the concentrations of testosterone were significantly increased at various doses of CTE. Further, the results showed a slight decrease in the level of LH in the DM group as compared to DMR, DME1, DME2, and DME4. The LH production was higher in the DME4 group.
3.2.6.Effect of CTE on the Sperm Parameters of Diabetic Rats
The experimental results showed that the DM group had a significant decrease in sperm number and motility than the control group whereas the sperm abnormality rate was significantly increased in the DM group. Interestingly, the sperm number, sperm motility, and the sperm abnormality rate of the DMR group improved but the sperm motility does not reach a significant level as compared to DME4. DME2 showed better sperm count than all of them and the motility rate was significantly increased in the DME4 group. There was no significant difference between the numbers of abnormal sperm betwee n RSG- and CTE-treated groups (Table 5).
3.2.7.Effect of CTEon the Morpholo of Seminiferous Tubules in Diabetic Rats
Figure 9 shows the H&E staining of the testis section. The black arrow denotes the Leydig cell and the white arrow denotes the Sertoli cell. Both the Leydig cell and the Sertoli cell in the DM group showed significant atrophy and a cavity was seees in the lumen. The structure of Leydig cells and Sertoli were restored inCTE- andRSG-treatedgroups. The thictasessof the seminiferous tubulewas higher in the CTEand RSGgroups than in the DM group.

3.2.8.Effect of CTE on KiSS1, GPR54, SOCS-3, and SIRT1 mRNAs in the Hypothalamus of Diabetic Rats
The expression of KiSS1 (Figure 10a), GPR54 (Figure 10b), SOCS-3 (Figure 10d), and SIRT1 (Figure 10c) were shown in Figure 10. The mRNA expression of KiSS1 and receptor GPR54 in diabetic rats was significantly lower than in the control group. The KiSS1 and GPR 54 mRNA expression level in DMR, DME1, DME2, and DME4 was significantly increased. In particular, the GPR54 mRNA expression was significantly increased in DME4 and was almost similar to the control group.
This experiment further explored the amount of SOCS-3 and SIRT1 mRNAs in the hypothalamus of rats. The expression of SOCS-3 mRNA in diabetic rats was increased significantly indicating that the leptin impedance was more serious. The DMR, DME1, DME2, and DME4 groups showed significant improvements when compared to the diabetic group. The SIRT1 mRNA expression in the DM group was decreased

3.2.9.Effect of CTE on Antioxidant Enzymes in Plasma and Testis of Diabetic Rats
Table 6 shows that the plasma SOD activity, GPx activity, and catalase activity of diab etic rats were decreased significantly and the activities were increased in CTE- and RSG-treated groups. In addition to this, DM E4 showed significant improvements in GPx activity than others. Results showed that the SOD and catalase activities in the diabetic rats were significantly decreased after six weeks. The SOD and catalase activity of the DMR group does not reach a significant level. The CTE-treated groups showed significant improvements in the SOD and catalase activity. The increased activities of catalase and SOD in CTE groups were also seen in testis (Table 7). The highest activity of SOD and catalase were observed in the DME1 group and DME2 groups respectively.

3.2.10.Effects of CTE on Oxidative Stress and Inflammation in the Plasma and Testis of Diabetic Rats
Nitric oxide (NO) production in plasma (Figure 11a) and testis (Figure lib) were shown in Figure The production of NO in the DM group was significantly increased both in testis and plasma as compared to the control group. A gradual reduction of NO production was observed in DME1, DME2, and DME4 groups (in plasma). The DMR group als o showed a significant reduction in NO production. In the case of the testis, there was a slight reduction of NO production was observed in CTE groups, whereas the DMR group does not significantly reduce NO production.
As shown in Figures 12 and 13, the level of TNF-a and IL-6 were significantly increased in diabetic rats (both in plasma and testis), indicating that the inflammation is more serious. In plasma, the level of TNF-a is similar in the CTE- and RSG-treated groups (Figure 12a). The CTE groups significantly (especially DME2) reduced the level of TNF-a in the testis (Figure 12b). The level of IL-6 was significantly reduced in the plasma of CTE and RSG groups (Figure 13a). In the testis, there was a trend to a re duction in the level of IL-6, but this did not reach a significant level (Figure 13b).
3.2.11.Effects of CTE on Oxidative Stress and Inflammation in Spermatozoa of Diabetic Rats I nduced by High-Fat Diet
Figure 14 shows the superoxide anion content in rat sperm. The results showed that the superoxide anion production in the sperm of diabetic rats increased significantly and there was no significant improvement was observed in the DMR group. DME1 and DME4 groups showed significantly reduced production of superoxide anion.
3.2.12. Effects of CTE on Lipid Peroxidation in Spermatozoa of Diabetic Rats Induced by High-Fat Diet
Studies have been shown that lipid peroxidation was increased in both type 1 and type 2 diabetes patients [19]. The malondialdehyde (MDA) level in plasma, sperm, and testis was shown in Table 8. MDA level in plasma, testis, and sperm of the DM group was significantly higher and the treatment with CTE and RSG reduced the production of MDA. A significant reduction was observed in DME4 and DMEI groups in plasma. This study found that the diabetic rats not only increased the degree of lipid peroxidation in plasma but at so increased in testis and sperm.

Effects of Cistanche tubulosa extract: Anti-Oxidative Stress and Inflammation
4. Discussion
Diabetes is a chronic disease associated with a high level of sugar in the blood. The imbalance between antioxidant and ROS levels will lead to the condition called oxidative stress. Superoxide, hydroxyl radical, hydrogen peroxide, nitric oxide, and singlet oxygen are some of the examples of ROS that will con tribute diabetes condition via oxidative stress [20]. Cittanche tubulosa is a desert plant that contains active components such as polysaccharides, oligosaccharide s, phenylethanoid glycosides (echinacoside, verbascoside), palmitic acid, linoleic acid, iridoids, alditols, and lignans. This plant is capable of producing anti-inflammatory, neuroprotective, antibacterial, antiviral, anti-oxidative, anti-tumor, and immunomodulatory effects [21]. Literature studies show that the phenylethanoid glycosides from Cistanche tubulosa are the major reason for the antioxidant activity [22]. Resveratrol (RES) and rosiglitazone (RSG) were taken as the positive control for in vitro and in vivo studies respectively. RSG is a powerful insulin sensitizer and has an affinity towards the isoform of peroxisome proliferator-activated receptor (PPARc). It controls hyperglycemia in diabetic patients [23]. RES has natural antioxidant activity, acts as a vasodilator, regulating lipoprotein metabolism, inhibiting platelet aggregation, and preventing cancer [24,25]. Our investigation indicated that the ECH shows better radical scavenging activity than CTE and RES (Figure 1). It is also understood that the ECH does not cause any significant toxicity to LC-540 and TM3 Leydig cells (Figure 2).
AGEs were used to induce stress conditions in Leydig cells. The production of AGEs, inflammation, oxidative stress and diabetes are interconnected. The hyperglycemic condition of diabetes promotes cell injury through the production of AGEs and oxidative stress. AGEs induce p cell toxicity, which further promotes immune cell recruitment and cell death. An elevated level of AGEs promotes the expression of two types of receptors called RAGE and AGER1 [26]. The dripping and subsequent transfer of an electron from the mitochondrial respiratory chain to molecular oxygen during oxidative stress results in the formation of a superoxide anion. Our results show that the production of superoxide anion induced by AGEs was increased in the control group and further improvements were observed with both ECH and RES treatments. According to our study, it was understood that the superoxide (Figure 3) and H2O2 (Figure 4) production induced by AGEs was decreased in the presence of ECH and RES.
NF-kB is known as the important mediator of inflammation associated with diabetes [27]. The expression of NF-kB leads to p cell dysfunction and cell death. The activation of NF-KB by oxidative stress stimulates the pro-inflammatory response, upregulation of endothelin, and apoptosis [28]. The expression of RAGE and NF-kB were increased in AGE-stimulated groups and the subsequent reduction was observed in ECH- and RES-treated cells (Figure 5).
Testosterone is an anabolic steroid and primary male sex hormone synthesized from cholesterol. The process starts with the oxidative cleavage of cholesterol's side chain by the cholesterol side-chain cleavage gene (CYP11A). This gene is localized in the mitochondrial membrane and converts the cholesterol into pregnenolone. Next, the CYP17A1 gene from the endoplasmic reticulum removes two extra carbon atoms and produces multiple C19 steroids. In addition to this, the pregnenolone is oxidized to form androstenedione/progesterone by hydroxysteroid dehydrogenase (3-p-HSD). Finally, testosterone is produced by the reduction of the keto group in the 17th carbon position of androstenedione by 17-beta-hydroxysteroid dehydrogenase (17-p-HSD). Leydig cells are involved in the major production of testosterone. The transfer of cholesterol into the inner mitochondrial membrane requires the action of steroidogenic acute regulatory protein (StAR). Present studies show that the expression of StAR, CYP11A1, CYP17A1, and HSD17p3 were decreased in AGE-treated cells (control) and there was a tremendous increase was shown in ECH- and RES-treated cells (Figure 6). Thus, the results show that the production of testosterone was increased in the ECH- and RES-treated groups.
During a diabetic condition, the transport of glucose to organs is limited and as a result, the glucose levels increase [29]. Hence, the hyperglycemic condition is one of the important markers for diabetic detection. Our studies show that the plasma glucose level was increased in diabetic groups and the AUC of the DM group was significantly higher than the other groups (Figure 8). Along with plasma glucose, the total cholesterol and triglyceride content was significantly increased in the DM group (Table 2). Atherosclerosis is facilitated by an increased level of cholesterol and triglycerides [30]. However, the level of cholesterol and triglycerides was decreased in CTE- and RSG-treated groups. The triglyceride content of the DME4 group reached an almost similar value to the control group.
Insulin is a hormone produced by the p cells of the pancreas and functionalized to control blood sugar level in the body while leptin is a hormone produced by the adipocytes and are capable of regulating food intake and energy utilization. Studies indicated that leptin is involved in the pathophysiology of obesity and there is a positive interaction between leptin and insulin [31]. Our studies show that the level of plasma inulin and leptin were higher in the DM group and the insulin level was decreased with an increase in the concentration of CTE. The RSG group does not show any significant difference from the DM group. The insulin resistance and p cell function were assessed by
HOMA-IR method. The HOMA-IR value increased in the DM group and was significantly different from other groups (Table 3).
DM affects the reproductive function via the hormonal alternation in the HPG axis and studies revealed that the insulin expression in the testis is also affected by diabetes. It is characterized by Sertoli cell vacuolization, increased DNA fragmentation, impaired spermatogenesis, and increased germ cell depletion. Oxidative stress also contributes to abnormalities in reproductive function [32]. The process of formation of sperm in the male reproductive organ (testes) is called spermatogenesis. The testis is composed of tightly coiled tubules called seminiferous tubules. Sertoli cells are seen in the walls of the seminiferous tubule and provide nourishment to the immature sperm. The investigation on sperm parameters indicated that the number of sperm and motility decreased and the abnormalities increased in DM group (Table 5). An opposite effect was observed in the CTE- and RSG-treated groups. Both the Leydig cell and the Sertoli cells in the DM group showed significant atrophy and the cavity was seen in the lumen. The thickness of the seminiferous tubule also decreased in the DM group. An improved result was observed in CTE- and RSG-treated groups (Figure 9). In addition to this, the level of LH and testosterone decreased in the DM group and the levels increased in CTE-treated groups (Table 4). In men, low serum testosterone and lower LH pulse frequency were often associated with obesity and diabetes mellitus type 2 [33].
Kisspeptins encoded by the KiSS1 gene are known as the potent stimulator of the HPG axis and any mutation in the kisspeptin gene will lead to low levels of sex steroids and gonadotropin. Studies show that in STZ-induced diabetic rats, the Kiss1 mRNA levels were decreased [33]. The initiation and maintenance of mammalian infertility are connected with G-protein coupled receptor 54 (GPR54). The mutation in GPR 54 is characterized by the absence of sexual maturation and low levels of gonadotropic hormones (LH and FSH). Pro-inflammatory cytokines such as IL-6 and TNF-a up-regulate the expression of the suppressor of cytokine signaling 3 (SOCS3) implicated in inflammation-mediated insulin resistance in the liver and adipocytes [34]. SIRT 1 is a gene associated with the regulation of several aging diseases. This gene is prominently expressed in the p cells of the pancreas and regulates insulin secretion and prevents apoptosis. Current studies indicated that the expression of KiSS1, GPR54, and SIRT1 decreased in the DM group, but the expression increased in CTE-treated groups (Figure 10). An increase in the SOCS-3 expressions in the DM group indicated the inflammatory condition. The first molecular link identified between obesity and inflammation was TNF-a. Hence, an increased level of TNF-a is an indicator of inflammation. The decreased level of pro-inflammatory cytokines such as TNF-a and IL-6 were seen in CTE-treated groups (Figures 12 and 13).
The imbalance between ROS and antioxidants leads to the diabetic condition. Superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPX) are known as the primary antioxidants responsible for maintaining the optimum ROS level [35]. From the results, it was found that the activity of antioxidants was significantly lower in the DM group. CT-treated groups showed improvements in the production of antioxidants. The RSG-treated group did not show any significant improvements in antioxidant activity (Tables 6 and 7).
Nitric oxide (NO) is known as an important ROS that contributes to inflammation. Our studies point out that the level of NO was decreased in CTE-treated groups (Figure 11). The results showed that the superoxide anion content in the sperm of diabetic rats increased significantly and there was no significant improvement after the administration of RSG. The production of superoxide was reduced in CTE groups.
The determination of MDA is very useful for evaluating lipid peroxidation. Lipid peroxidation is the process of oxidation in lipids and finally results in cell damage. MDA is produced as the result of lipid peroxidation of polyunsaturated fatty acids. Studies show that MDA level is correlated with age and fasting blood glucose level [36]. The current study indicated that the CTE improves lipid peroxidation in plasma, testis, and sperm (Table 8).

Cistanche tubulosa extract
5. Conclusions
The oxidative stress during diabetic conditions disturbs the male reproductive system by sperm impairment and gonadal dysfunction. Cistanche tubulosa is a desert plant widely accepted in Chinese medicine due to its pharmacological effects. Echinacoside (ECH) is the main constituent of CTE responsible for antioxidant and anti-inflammatory activities. Our in vitro results indicated that the ECH restored the testosterone synthesis pathway and lowered the level of NF-kB and RAGE protein expression. ECH effectively inhibited the production of superoxide anion and H2O2 in Leydig cells. The in vivo studies revealed that the ECH reduced the levels of cholesterol, triglycerides, TNF-a, and IL-6. In addition to this, the mRNA expressions in the hypothalamus of the diabetic rats were significantly improved. It is also of note that the ECH reduced lipid peroxidation and improved insulin resistance in diabetic male rats. The antioxidant activities increased both in plasma and testis. Therefore, our studies suggested that the ECH provided effective protection against reproductive dysfunction in STZ-induced diabetic male rats.
Author Contributions: F.-C.K. performed investigation, formal analysis, and software analysis. A.J. and J.-L.H. participated in the data curation and drafted the manuscript. Z.-L.K. and A.J. conceived the manuscript review and edited it. Z.-L.K. formulated the methodology and supervised the whole experiment. S.-C.C. participated in formal analysis, investigation, and validation. All authors contributed toward data analysis, drafting, and critically revising the paper, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding: This work was financially supported by the Center of Excellence for the Oceans, National Taiwan Ocean University, from the Featured Areas Research Centre Program within the framework of Higher Education Sprout Project by the Ministry of Education (MOE), Taiwan.
Conflicts of Interest: The authors declared no conflict of interest.
References
1. World Health Organization. Global Reports on Diabetes. 2017. Available online: http://www.who.int/ news-room/fact-sheets/detail/diabetes (accessed on 25 August 2018).
2. Haynes, N.E.; Corbett, W.L.; Bizzarro, F.T.; Guertin, K.R.; Hilliard, D.W.; Holland, G.W.; Kester, R.F.; Mahaney, P.E.; Qi, L.; Spence, C.L.; et al. Discovery, structure-activity relationships, pharmacokinetics, and efficacy of glucokinase activator (2R)-3-cyclopentyl-2-(4-methane-sulfonyl phenyl)-N-thiazol-2-yl- propionamide (RO0281675). J. Med. Chem. 2010, 53, 3618-3625. [CrossRef] [PubMed]
3. Agbaje, I.M.; Rogers, D.A.; McVicar, C.M.; McClure, N.; Atkinson, A.B.; Mallidis, C.; Lewis, S.E. Insulin dependant diabetes mellitus: Implications for male reproductive function. Hum. Re-prod. 2007, 22,1871-1877. [CrossRef] [PubMed]
4. Fouchecourt, S.; Metayer, S.; Locatelli, A.; Dacheux, F.; Dacheux, J.L. Stallion epididymal fluid proteome: Qualitative and quantitative characterization; Secretion and dynamic changes of major proteins1. Biol. Reprod. 2000, 62,1790-1803. [CrossRef] [PubMed]
5.Vernet, P.; Aitken, R.J.; Drevet, J.R. Antioxidant strategies in the epididymis. Mol. Cell. Endocrinol. 2004,216, 31-39. [CrossRef] [PubMed]
6.Szkudelski, T. Streptozotocin-nicotinamide-induced diabetes in the rat. Characteristics of the experimental model. Exp. Biol. Med. 2012,237, 481-490. [CrossRef] [PubMed]
7.Zheng, S.; Jiang, X.; Wu, L.; Wang, Z.; Huang, L. Chemical and genetic discrimination of Cistanches herba based on UPLC-QTOF/MS and DNA barcoding. PLoS ONE 2014, 9, e98061. [CrossRef] [PubMed]
8. Li, J.; Li, J.; Aspire, A.; Gao, L.; Huo, S.; Luo, J.; Zhang, F. Phenylethanoid glycosides from Cistanche tubulosa inhibits the growth of B16-F10 Cells both in vitro and in vivo by induction of apoptosis via mitochondria-dependent Pathway. J. Cancer 2016, 7,1877-1887. [CrossRef] [PubMed]
9. Zhao, Q.; Gao, J.; Cai, D. Neurotrophic and neurorescue effects of echinacoside in the subacute MPTP mouse model of Parkinson's disease. Brain Res. 2010,1346, 224-236. [CrossRef] [PubMed]
10. Yoshikawa, M.; Matsuda, H.; Morikawa, T.; Xie, H.; Nakamura, S.; Muraoka, O. Phenylethanoid aminoglycosides and acylated oligosugars with vasorelaxant activity from Cistanche tubulosa. Bioorg. Med. Chem. 2006, 14, 7468-7475. [CrossRef] [PubMed]
11. Jiang, Z.; Wang, J.; Li, X.; Zhang, X. Echinacoside and Cistanche tubulosa (Schenk) R. wight ameliorate bisphenol A-induced testicular and sperm damage in rats through gonad axis regulated steroidogenic enzymes. J. Ethnopharmacol. 2016,193, 321—328. [CrossRef] [PubMed]
12. Placer, Z.A.; Cushman, L.L.; Johnson, B.C. Estimation of product of lipid peroxidation (malonyl dialdehyde) in biochemical systems. Anal. Biochem. 1966,16, 359-364. [CrossRef]
13. Younglai, E.V.; Holt, D.; Brown, P.; Jurisicova, A.; Casper, R.F. Sperm swim-up techniques and DNA fragmentation. Hum. Reprod. 2001,16,1950-1953. [CrossRef] [PubMed]
14. Yokoi, K.; Uthus, E.O.; Nielsen, F.H. Nickel deficiency diminishes sperm quantity and movement in rats. Biol. Trace Elem. Res. 2003, 93,141-153. [CrossRef]
15.Aebi, H. Catalase in vitro. Methods Enz^mol. 1984,105,121-126. [CrossRef] [PubMed]
16. Brownlee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001, 414, 813. [CrossRef] [PubMed]
17.Shimoda, H.; Tanaka, J.; Takahara, Y.; Takemoto, K.; Shan, S.J.; Su, M.H. The Hypocholesterolemic Effects of Cistanche tubulosa Extract, a Chinese Traditional Crude Medicine, in Mice. Am. J. Chin. Med. 2009, 37, 1125-1138. [CrossRef] [PubMed]
18. Baynes, J.W.; Thorpe, S.R. Role of oxidative stress in diabetic complications: A new perspective on an old paradigm. Diabetes 1999, 48,1-9. [CrossRef] [PubMed]
19. Akkus, I.; Kalak, S.; Vural, H.; Caglayan, O.; Menekse, E.; Can, G.; Durmus, B. Leukocyte lipid peroxidation, superoxide dismutase, glutathione peroxidase and serum and leukocyte vitamin C levels of patients with type II diabetes mellitus. Clin. Chim. Acta 1996, 244, 221-227. [CrossRef]
20. Matough, F.A.; Budin, S.B.; Hamid, Z.A.; Alwahaibi, N.; Mohamed, J. The role of oxidative stress and antioxidants in diabetic complications. Sultan. Qaboos Univ. Med. J. 2012, 12, 5-18. [CrossRef] [PubMed]
21. Gu, C.; Yang, X.; Huang, L. Cistanches herba: A neuropharmacology review. Front. Pharmacol. 2016, 7,1-10. [CrossRef] [PubMed]
22. Xiong, W.T.; Gu, L.; Wang, C.; Sun, H.X.; Liu, X. Anti-hyperglycemic and hypolipidemic effects of Cistanche tubulosa in type 2 diabetic db/db mice. J. Ethnopharmacol. 2013,150, 935-945. [CrossRef] [PubMed]

Cistanche tubulosa supplements







