PARTⅠ: Effects And Molecular Mechanism Of Pachymic Acid On Renal Ischemia Reperfusion Injury
Mar 25, 2022
Contact: joanna.jia@wecistanche.com / WhatsApp: 008618081934791
GUI-PING JIANG, YUE-JUANLIAO,L-LI HUANG,XU-JIAZENG & XIAO-HUI LIAO
Introduction
Acute kidney injury(AKI) is a common clinical disease that is caused by multiple etiologies and has a complex pathophysiological process(1). The morbidity and mortality rates for AKI are high among adults and children, thus, resulting in substantial hospitalization costs(2,3). A meta-analysis of the global incidence of AKIrevealed incidence rates of AKI was 21.6% in adults and 33.7% in children and AKI-associated mortality rates were 23.9% in adults and 13.8% in children (4). AKI occurs in ~20% of hospitalized patients, 10% of whom require renal replacement therapy, and in as much as 50% of patients in intensive care units, thus significantly increasing the length and cost of hospitalization (5).AKI is characterized by a sharp decrease in glomerular filtration rate in the short term, which is accompanied by an increase in serum creatinine, oliguria, or both; these changes can lead to chronic kidney disease or end-stage renal disease(6). Due to the variability of renal injury, the lack of reliable early biomarkers, and the heterogeneity of pathways involved in pathophysiology, there are currently no effective means of prevention and intervention except conservative treatment and renal replacement therapy(7). Thus, identifying novel treatment regimens and drugs for AKI is particularly important. The past decade has seen a wealth of research expanding the traditional view of necrosis. Recent research has challenged traditional views of cell death by identifying novel pathways in which cells die in a regulated manner, but exhibit the morphologic features of necrosis(7-9). This regulated necrosis takes several forms: Necroptosis, ferroptosis, pyroptosis, mitochondrial permeability transition-driven necrosis, and parthanatos(10). Necroptosis and ferroptosis are the most studied forms of regulated necrosis occurring in the kidney(11). A previous study demonstrated that inhibiting ferroptosis effectively ameliorates AKI (12).

cistanche pharma special for kidney
Ferroptosis is a new type of regulatory cell death, which was first proposed by Dixon et al in 2012(13). This type of cell death is iron-dependent and is accompanied by massive iron accumulation and lipid peroxidation during cell death(13). Ferroptosis serves an important regulatory role in the occurrence and development of several diseases, including tumors, neurological diseases, and AKI(14).In an in vivo study, ferrostatin 1 or 16-86 was administered 15 min before ischemia in mice with severe ischemia-reperfusion injury(IRI)-AKI, and renal tissue damage, serum creatinine, and urea all decreased in the mice 48 h after ischemia, thus, suggesting that ferroptosis has a crucial role in the pathogenesis of IRI (ischemia-reperfusion injury)(15). Therefore, regulating iron homeostasis, resisting lipid peroxidation, and inhibiting ferroptosis may provide a promising therapeutic strategy for AKI.
Pachydermic acid (PA), a lanostane-type triterpenoid from Poria cocos, has several pharmacological effects, such as antitumor, anti-inflammatory, antioxidative, hypoglycemic, sedative, and hypnotic activities(16-20).A previous study has reported that PA(Pachymic acid) can improve renal injury during sepsis in rats by inhibiting inflammatory responses and activating the nuclear factor erythroid-derived 2 like 2(NRF2)/heme oxygenase 1(HO-1) pathway through antioxidant stress (21). In addition, Poricoic acid A effectively enhances the inhibition of melatonin in the transition from AKI to chronic kidney disease following renal ischemia/reperfusion injury(22). PA(Pachymic acid) protects against AKI, however, this protective mechanism remains to be further investigated. PA(Pachymic acid) has been reported to activate NRF2in a study of renal injury in sepsis(21). Notably, the first two ferroptosis-inducing agents (RSL-3 and elastin)identified in ferroptosis studies initiate the ferroptosis cascade via inhibition of glutathione peroxidase 4(GPX4) and the cystine/glutamate transporter system (xC-/xCT), respectively, both of which are downstream targets of NRF2 (23). PA(Pachymic acid) is assumed to regulate its downstream proteins, GPX4, and xCT by activating NRF2 and interfering with ferroptosis in AKI. The present study aimed to investigate the effect of PA(Pachymic acid) on ferroptosis in mice with ischemia-reperfusion kidney injury and determine its underlying molecular mechanism of action to provide novel ideas for the treatment of clinical AKI.

Materials and methods
Reagents and animals.
PA(Pachymic acid) (white crystalline powder with purity ≥99.9%) was purchased from MedChemExpress (cat.no.HY-N0371) and stored away from light at 4C. A total of 30 C57BL/6 male mice(8-10 weeks;20-25g body weight) were purchased from Chongqing Medical University (Chongqing, China). The housing conditions were as follows:12 h light/12 h dark,~60% humidity,25°C, free access to drinking water and food, and bedding replacement every3 day; the drinking water and bedding materials were disinfected before use. All animal experiments were performed according to the Guide for the Care and Use of Laboratory Animals, issued by the National Institutes of Health in 1996(24). The present study was approved by the Institutional Animal Care and Use Committee of Chongqing Medical University (Chongqing, China; approval no.2018-019).
Ischemia-reperfusion injury-AKI model.
Mice were fed under the same conditions for 1 week to produce the model of renal ischemia-reperfusion injury, as previously described (25). The mice were anesthetized with 50-60 mg/kg of pentobarbital sodium (cat.no. P3761; Sigma-Aldrich; Merck KGaA)by intraperitoneal injection; the skin at the surgical area was wiped with 70% alcohol. The incision was positioned at the left and right sides of the spine (0.5 cm), and the incision length was 1-1.5 cm along the back. The kidneys were subsequently pulled out from the incision to expose the renal pedicle. A microaneurysm clip was used to clamp the pedicle to block the blood flow to the kidney and induce renal ischemia. Complete ischemia as indicated by a change in the color of the kidney from red to dark purple within a few seconds. After 40 min of ischemia, the microaneurysm clips were released to allow each kidney to start reperfusion, which was indicated by the change of the kidney color to red. The muscle skin was sutured after the kidney color returned to normal. The entire renal ischemia procedure was completed on a thermostatic pad, and the body temperature of the mice was kept constant at 36.5-37℃. A total of 0.8 ml warm sterile saline was administered intraperitoneally to each mouse. Each animal was placed on a heating pad until it regained full consciousness and was subsequently returned to its cage. The mice were sacrificed via cervical dislocation 24h after surgery, and the blood was collected from the orbital sinus. One-third of the renal tissue was used for histopathological analyses, and the remaining two-thirds were quickly stored at -80°℃.
Animal groups.
C57BL/6 mice were divided into five groups (n=6/group)according to a random number table, as follows: Sham group(Sham), model group(IRI), low dose group (IRI+PA)), moderate dose group(IRI+PAw), and high dose group(IRI+PAg). Animals in the three experimental treatment groups received an intraperitoneal injection with PA(Pachymic acid) at dosages of 5, 10, and 20 mg/kg. but for 3 days, before the model was induced. The sham and IRI(ischemia-reperfusion injury) groups were injected with the same volume of dimethyl sulfoxide for 3 days before model induction. The modeling method was performed as previously described (25). The bilateral renal pedicels of the mice in the sham group were exposed and treated as aforementioned, but were not clamped. The surgical incision was sutured after 40min.
Serum biochemical analysis.
The mice were sacrificed 24 h post-surgery, and 0.8-1 ml blood was collected from the orbital sinus, allowed to rest for 30 min, and centrifuged at 900xg for 10 min at 4C. The upper serum layer was collected, packed, and stored at -80°C. Serum creatinine (Scr; cat.no.c011-2-1)and blood urea nitrogen(BUN: cat.no.cO13-2-1)were assessed using reagent kits, according to the manufacturer's instructions (Nanjing Jiancheng Bioengineering Institute).
Histopathological examination.
At 24 h post-IRI(ischemia-reperfusion injury), portions of the kidney tissues were fixed in 10%neutral-buffered formalin for 24-48 h at room temperature, processed routinely by embedding in paraffin. The paraffin-fixed tissue specimens were sliced into 4 μm thick sections, which were mounted on glass slides and stained with hematoxylin for 10 min at room temperature and eosin for3 min at room temperature. A BX51 light microscope(Olympus Corporation) was used to detect the renal histopathological changes (magnification, x200, and x400). The Paller score was used to grade renal tubular necrosis in renal IRI(ischemia-reperfusion injury) (26). A total of 10 non-overlapping areas were randomly selected under high magnification using an optical light microscope (magnification, x400), and 10 renal tubules were randomly selected in each field of vision. The renal pathology sections of three mice in each experimental group were randomly selected for observation. A total of 10 visual fields were randomly selected for each pathological section, and 10 renal tubules were scored in each visual field. A total of 100 tubules was observed for each mouse, giving a total of 300 renal tubules observed in each group. The higher the score, the more serious the degree of damage of renal tubules.
Transmission electron microscopy (TEM).
Renal tissue was collected 24 h post-surgery. Following anesthesia,1 mm²renal tissue was removed in vivo and soaked in tissue fixation solution composed of 2.5% glutaraldehyde and phosphoric acid buffer solution (cat.no.G7776; Sigma-Aldrich; Merck KGaA)for 2 h at 4°℃, dehydrated in gradual ethanol (50-100%)and propylene oxide, embedded in Epon 812(cat. no. GP18010; Bejing Zhongjingkeyi Technology Co.Ltd. for 1 h at 35℃℃and solidified for 24 h at 60°C. Ultrathin sections(thickness, 50-70 nm) were placed onto 200-mesh copper grids and stained with 2% uranyl acetate for 30 min at room temperature and 0.04% lead citrate for 15 min at room temperature. The samples were viewed under an electron microscope (JEM-1400 plus, Japan Electron Optics Laboratory Co., Ltd.; magnification, x5,000 and x10,000). Images were obtained using a SlowScan charge-coupled device camera and TEM analySIS5.0 software(Olympus Soft Imaging Solutions GmbH).
Antioxidant glutathione(GSH) and lipid peroxidation malondialdehyde (MDA) detection.
Kidney tissue (50-100mg)was collected, and physiological saline solution(cat.no. B020; Nanjing Jiancheng Bioengineering Institute) was added to the tissue at a 1:9 weight(g): volume (ml) ratio. The tissue was homogenized, to produce 10% tissue homogenate, which was centrifuged at 13,800 xg for 15 min at 4℃. The supernatant was removed and the total protein content of the supernatant was determined using the bicinchoninic acid method(21). GSH(cat.no.A006-2-1) and MDA(cat.no.A003-2-2) were assessed using the reagent kits, according to the manufacturer's instructions(Nanjing Jiancheng Bioengineering Institute).
Western blotting.
Total protein was extracted from mouse kidney tissues using RIPA cell lysis buffer(cat. no. WO62-1-1; Nanjing Jiancheng Bioengineering Institute)and the protein concentrations were determined via BCA assay(cat.no. A045-4-2; Nanjing Jiancheng Bioengineering Institute).Equal amounts of proteins (50μug)were separated via SDS-PAGE(Cox-2,10.0;NRF2,10.0;xCT,12.5;HO-1, 12.5; GPx4.15.0%) and transferred onto PVDF membranes (GE Healthcare Life Sciences). The membranes were subsequently blocked with 5% skimmed milk powder for 1 h at room temperature,and incubated with primary antibodies against the following: Cyclooxygenase 2(COX-2;1:1,000; cat. no.ab179800;Abcam), GPX4(1:5,000;cat.no.ab125066;Abcam),xCT(1:2,000;cat.no.ab175186;Abcam), HO-1(1:1,000 cat.no.101147;Gentex)and NRF2 (1:1,000;cat.no.D1Z9C;CST)overnight at 4°C.Membranes were subsequently washed with TBST(0.1% Tween-20)three times. Following the primary incubation, membranes were incubated with horseradish peroxidase-conjugated secondary antibodies(1:10,000;40295G; BIOSS)for 1h at room temperature. The membranes were re-washed three times with TBST and visualized with BeyoECLPlus (cat. no. P00185; Beyotime Institute of Biotechnology), using a Chemi Doc Imaging System (Bio-Rad Laboratories, Inc.)The intensity of protein bands was determined using ImageJ v1.8.0 software(National Institutes of Health). All experiments were performed in triplicate.
Reverse transcription-quantitative(RT-g)PCR.
Total RNA was extracted from the kidneys of mice in different groups using TRIzol reagent(Takara Bio, Inc.) and cDNA was synthesized using the PrimeScript RT Reagent kit with gDNA Eraser(cat.no.RR047A; Takara Biotechnology Co., Ltd.).Relative levels of the target genes were determined via qPCR using Ultra SYBR Mixture(Takara Biotechnology Co., Ltd.). RT-qPCR was performed in a 25 ul volume with 2 μl cDNA, 400 nM each sense and antisense primer, and 12.5 μl Brilliant SYBR Green QPCR Master Mix (Takara Bio, Inc. on an ABI PRISM 7000 system(Applied Biosystems; Thermo Fisher Scientific, Inc.).The reaction was performed for 40 cycles of denaturation at 95°C for 30 sec, annealing at 53°C for 30 sec, and extension at 72℃for 10 sec. The following primer sequences were used for qPCR:GPX4 forward,5'-GCCTGGATAAGTACA GGGGTT-3' and reverse,5'-CATGCAGATCGACTAGCT GAG-3';NRF2 forward,5'-TCTTGGAGTAAGTCGAGA AGTGT-3' and reverse,5'-GTTGAAACTGAGCGAAAA AGGC-3';xCT forward, 5'-GGCACCGTCATCGGATCA G-3' and reverse,5-CTCCACAGGCAGACCAGAAAA-3';COX-2 forward,5'-TGAGCAACTATTCCAAACCAGC-3'and reverse,5'-GCACGTAGTCTTCGATCACTATC-3';HO-1 forward.5'-AAGCCGAGAATGCTGAGTTCA-3' and reverse,5'-GCCGTGTAGATATGGTACAAGGA-3'; and β-actin forward,5'-GGCTGTATTCCCCTCCATCG-3' and reverse,5'-CCAGTTGGTAACAATGCCATGT-3'.
An average value of gene expression after β-actin normalization was used as a calibrator to determine the relative levels of the target genes. Relative expression levels of the target genes were calculated using the 2-△C method (27).
Statistical analysis.
Statistical analysis was performed using GraphPad Prism 7.0(GraphPad Software, Inc.) and SPSS 23 software(IBM Corp..Data are presented as the mean± standard deviation of three independent repeats unless stated otherwise. One-way ANOVA followed by Tukey's post hoc test was used to compare differences between multiple groups. P<0.05 was considered to indicate a statistically significant difference.

Results
PA(Pachymic acid) decreases renal injury in mice with IRI(ischemia-reperfusion injury). Scr and BUN are two important indicators of renal function(5). The levels of Scr and BUN were significantly higher in the IRI(ischemia-reperfusion injury) group compared with those in the sham group(P<0.05; Fig. 1A and B). Treatment with mid-dose and high-dose PA(Pachymic acid) significantly mitigated the increases in serum creatinine and urea compared with the IRI(ischemia-reperfusion injury) group(P<0.05); however, the levels of serum creatinine and urea did not significantly differ between the light-dose PA(Pachymic acid) treatment group and the IRI(ischemia-reperfusion injury) group (P>0.05; Fig. 1A and B).
As presented in Fig. 1D, H&E staining of kidney tissues in the sham group demonstrated no significant changes in renal tissue structure. Conversely, the IRI(ischemia-reperfusion injury) group demonstrated edema and loss of renal tubular epithelial cells, tubular dilation, interstitial inflammation, inflammatory cell infiltration, collagen deposition, and deposition tube necrosis material (Fig. 1D). The Paller score, an indicator of renal tissue injury(26), was significantly higher in the IRI(ischemia-reperfusion injury) groups compared with the sham group(P<0.05; Fig. 1C). In particular, moderate-dose and high-dose PA(Pachymic acid) therapy significantly ameliorated these lesions, and the Paller score was significantly lower compared with that in the IRI(ischemia-reperfusion injury) group (P<0.05); however, there was no significant difference between the IRI(ischemia-reperfusion injury) model group and the low-dose group (P>0.05; Fig. 1C).

Figure 1. PA(Pachymic acid) alleviates renal injury following ischemia-reperfusion. (A) BUN and (B) serum creatinine were measured in mice in the Sham, IRI(ischemia-reperfusion injury), IRI+PAL, IRI+PAM, and IRI+PAH groups (40 min renal ischemia followed by 24 h of reperfusion; n=6/group). (C) Paller scores were used to grade renal tubular injury in the Sham, IRI, IRI+PAL, IRI+PAM, and IRI+PAH groups (n=3/group). (D) Hematoxylin and eosin images of the kidneys from mice in the Sham, IRI(ischemia-reperfusion injury), IRI+PAL, IRI+PAM, and IRI+PAH group. Scale bar, 50 µm. Data are presented as the mean ± standard deviation. * P<0.05 vs. sham group; # P<0.05 vs. IRI group. PA(Pachymic acid), pachydermic acid; BUN, blood urea nitrogen; IRI, ischemia-reperfusion injury; L, low dose group; M, moderate dose group; H, high dose group.
Effects of PA(Pachymic acid) on the morphological changes in mitochondria in the renal tissue of mice following renal IRI(ischemia-reperfusion injury). TEM demonstrated no significant changes in the mitochondrial structure of the renal tissue in the sham group. In the model group, ferroptosis-associated mitochondrial changes, such as decreased mitochondrial volume, increased membrane density, and decreased or absent mitochondrial cristae, were observed in renal tissue(Fig.2A). Compared with those in the model group, the mitochondria of the moderate-dose group mainly exhibited edema, and the specific changes characteristic of ferroptosis were rare(Fig.2A). However, in the high-dose PA(Pachymic acid) group, no characteristic changes of ferroptosis were observed in the mitochondria of the renal tissue, and only mild edema was exhibited(Fig. 2A). The changes in the mitochondria in the low-dose group were the same as those in the model group (Fig.2A).
Effects of PA(Pachymic acid) on GSH and MDA expression in renal IRI(ischemia-reperfusion injury). GSH is a necessary cofactor for the GPX4 function.GSH synthesis directly affects GPX4 activity.MDA is a product of lipid oxidation, which reflects the degree of intracellular lipid peroxidation and indirectly reflects the occurrence of ferroptosis(28,29). As presented in Fig. 2B, the kidney tissues had significantly lower levels of GSH in the IRI(ischemia-reperfusion injury) group compared with the sham group. Among the drug treatment groups, GSH expression was significantly higher in the IRI+PA and IRI+PAn groups compared with the IRI(ischemia-reperfusion injury) model group(P<0.05). However, GSH expression did not significantly differ between the IRI+PA, and IRI groups(P>0.05). Conversely, as presented in Fig. 2C, the tissue lipid peroxidation was greater(based on MDA levels)in the IRI(ischemia-reperfusion injury) group compared with the sham group. Among the drug treatment groups, MDA levels were significantly lower in the IRI+PA and IRI+PAu groups compared with the IRI(ischemia-reperfusion injury) group (P<0.05). Notably, no significant differences were observed in the levels of MDA between the IRI+PA, and IRI groups (P>0.05).

Figure 2. PA(Pachymic acid) decreases ferroptosis in IRI(ischemia-reperfusion injury)‑acute kidney injury. (A) Transmission electron microscopy images in the Sham, IRI(ischemia-reperfusion injury), IRI+PAL, IRI+PAM, and IRI+PAH groups (40 min renal ischemia followed by 24 h of reperfusion). Magnification, x20,000; scale bar, 1 µm. The black arrows indicate that the mitochondrial volume decreased, membrane density increased and decreased, or disappeared mitochondrial cristae were observed in renal tissue. The blue arrows indicate mitochondrial edema. The levels of (B) GSH and (C) MDA in renal tissues were assessed (n=6/group). Data are presented as the mean ± standard deviation. * P<0.05 vs. sham group; # P<0.05 vs. IRI(ischemia-reperfusion injury) group. PA(Pachymic acid), pachydermic acid; IRI, ischemia-reperfusion injury; L, low dose group; M, moderate dose group; H, high dose group; GSH, glutathione; MDA, malondialdehyde.
PA(Pachymic acid) regulates the expression of ferroptosis-related proteins in renal IRI(ischemia-reperfusion injury). In the present study, the expression levels of the ferroptosis-related proteins GPX4, xCT, and HO-1 were detected via western blotting (Fig.3A and B)and RT-qPCR (Fig.3C) analyses. The results demonstrated that the protein levels of GPX4, xCT, and HO-1 were significantly downregulated in the IRI(ischemia-reperfusion injury) group compared with the sham group (P<0.05)(Fig.3A and B). Similar results were demonstrated following RT-qPCR analysis. In addition, treatment with moderate-dose and high-dose PA(Pachymic acid) significantly increased the protein and mRNA expression levels of GPX4, xCT, and HO-1 compared with the IRI(ischemia-reperfusion injury) group(P<0.05). However, no significant differences in protein and mRNA expression levels of GPX4.xCT and HO-1 were observed between the IRI(ischemia-reperfusion injury) and low-dose PA(Pachymic acid) treatment groups (P>0.05; Fig.3B and C).
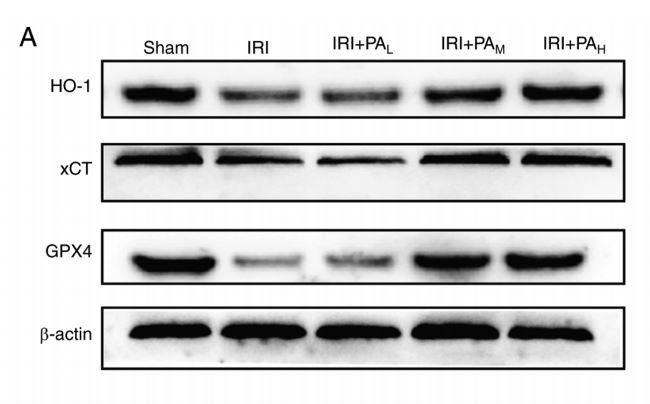
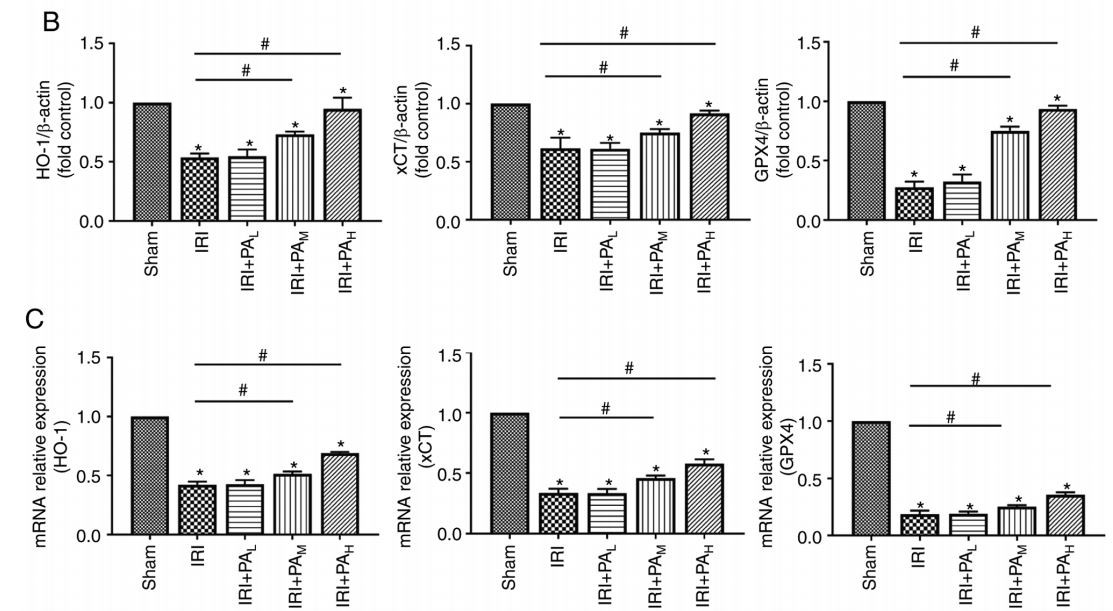
Figure 3. PA(Pachymic acid) increases the expression levels the ferroptosis-related proteins, GPX4, xCT, and HO‑1. (A) Western blotting was performed to detect the expression levels of the ferroptosis-related proteins, GPX4, xCT, and HO‑1 in IRI(ischemia-reperfusion injury)‑acute kidney injury mice (n=3). (B) Relative expression of gray values for GPX4, xCT, and HO‑1. (C) Reverse transcription‑quantitative PCR analysis was performed to detect the mRNA expression levels of the ferroptosis-related proteins (n=3). Data are presented as the mean ± standard deviation. * P<0.05 vs. sham group; # P<0.05 vs. IRI(ischemia-reperfusion injury) group. PA(Pachymic acid), pachydermic acid; GPX4, glutathione peroxidase 4; xCT, glutamate transporter system; HO‑1, heme oxygenase 1; IRI, ischemia-reperfusion injury; L, low dose group; M, moderate dose group; H, high dose group.

The results demonstrated that protein and mRNA levels of the ferroptosis-associated lipid peroxidation protein, COX-2 were significantly upregulated during IRI(ischemia-reperfusion injury)(Fig.4). Treatment with moderate-dose and high-dose PA(Pachymic acid) significantly decreased COX-2 expression compared with the IRI(ischemia-reperfusion injury) group (P<0.05), and no significant differences were observed between the IRI(ischemia-reperfusion injury) and low-dose PA(Pachymic acid) treatment groups (P>0.05; Fig.4).
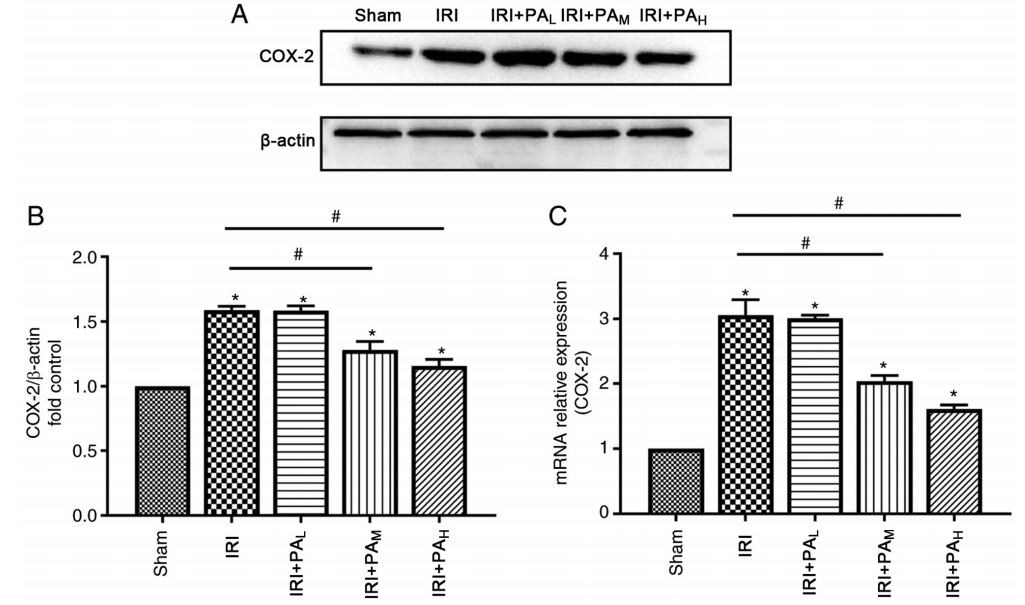
Figure 4. PA(Pachymic acid) decreases the expression levels of the ferroptosis-related protein, COX‑2. (A) Western blotting was performed to detect the expression levels of the ferroptosis lipid peroxidation marker protein, COX‑2 in IRI‑acute kidney injury mice. (B) Relative expression of gray values for COX‑2. (C) Reverse transcription‑quantitative PCR analysis was performed to detect mRNA COX‑2 expression (n=3). Data are presented as the mean ± standard deviation. * P<0.05 vs. sham group; # P<0.05 vs. IRI(ischemia-reperfusion injury) group. PA(Pachymic acid), pachydermic acid; COX‑2, cyclooxygenase 2; IRI, ischemia-reperfusion injury; L, low dose group; M, moderate dose group; H, high dose group.
PA(Pachymic acid) promotes activation of the NRF2 signaling pathway in renal IRI(ischemia-reperfusion injury).To investigate the effect of PA(Pachymic acid) on the NRF2 signaling pathway, the protein and mRNA levels of NRF2 were assessed(Fig.5). The results demonstrated that NRF2 expression was significantly lower in the IRI(ischemia-reperfusion injury) group compared with the sham group. In addition, NRF2 expression was significantly higher in the IRI+PAw and IRI+PAH groups compared with the IRI(ischemia-reperfusion injury) group (P<0.05); however, no significant differences were observed between the IRI+PA, and IRI groups(P>0.05). Furthermore, treatment with PA(Pachymic acid) also increased NRF2 expression in a dose-dependent manner (Fig.5).

Figure 5. PA(Pachymic acid) activates the NRF2 signaling pathway. (A) Western blotting was performed to detect NRF2 protein expression in IRI(ischemia-reperfusion injury)‑acute kidney injury mice. (B) Relative expression of gray values for NRF2 (n=3). (C) Reverse transcription‑quantitative PCR analysis was performed to detect mRNA NRF2 expression (n=3). Data are presented as the mean ± standard deviation. *P<0.05 vs. sham group; # P<0.05 vs. IRI(ischemia-reperfusion injury) group. PA(Pachymic acid), pachydermic acid; NRF2, nuclear factor erythroid-derived 2 like 2; IRI, ischemia-reperfusion injury; L, low dose group; M, moderate dose group; H, high dose group.






