Part Ⅰ Hydrogen: A Novel Treatment Strategy in Kidney Disease
May 23, 2023
Abstract
1. background
Hydrogen is a chemical substance that has yet to be widely used in medicine. However, recent evidence indicates that hydrogen has multi-faceted pharmacological effects such as antioxidant, anti-inflammatory, and antiapoptotic properties. An increased number of studies are being conducted on the application of hydrogen in various diseases, especially those affecting the renal system.
2. summary
Hydrogen can be inhaled, as a gas or liquid, and can be administered orally, intravenously, or locally. Hydrogen can rapidly enter sub-organelles such as mitochondria and the nucleus by simple diffusion, producing reactive oxygen species (ROS) and triggering DNA damage. Hydrogen can selectively scavenge hydroxyl radical (•OH) and peroxynitrite (ONOO−), but not other reactive oxygen radicals with physiological functions, such as peroxy anion (O2 −) and hydrogen peroxide (H2O2). Although the regulatory effect of hydrogen on the signal transduction pathway has been confirmed, the specific mechanism of its influence on signal molecules remains unknown. Although many studies have investigated the therapeutic and preventive effects of H2 in cellular and animal experiments, clinical trials are few and still far behind. As a result, more clinical trials are required to investigate the role of hydrogen in kidney disease, as well as the effect of its dose, timing, and form on the overall efficacy. Large-scale randomized controlled clinical trials will be required before hydrogen can be used to treat renal illnesses.
3. Key Messages
This article reviews the mechanisms of hydrogen in the treatment of renal disease and explores the possibilities of its use in clinical practice.
Keywords
Hydrogen; Kidney disease; Anti-inflammatory; Antioxidant; Cell death.

Click here to get the Cistanche benefits
Introduction
Kidney disease is one of the most common diseases in humans. Common types of kidney disease include acute kidney injury (AKI), renal fibrosis, polycystic kidney disease, and renal cell carcinoma. There are many treatment options available for kidney diseases, including drug therapy, kidney dialysis, and kidney transplantation. In recent years, increasing attention has been paid to research using hydrogen to treat these diseases. Hydrogen exhibits strong reducing properties and is frequently used as a reducing agent in chemical reactions.
Therapeutic applications of H2 were first reported in 1975 when tumors’ size reduced significantly when hairless albino mice with squamous cell carcinoma were exposed to a mixture of 2.5% oxygen and 97.5% hydrogen at a total pressure of 8 atmospheres for up to 2 weeks [1]. In 2007, Ohsawa et al. [2] proposed for the first time that hydrogen gas had antioxidant and antiapoptotic properties that could protect the brain against ischemia-reperfusion (I/R) injuries (IRIs) and stroke by selectively neutralizing hydroxyl radicals. Hydrogen has attracted much attention due to its potential therapeutic effect on kidney diseases. Many clinical and experimental models of kidney disease, as well as accumulating evidence in other biomedical domains, have demonstrated that hydrogen can act as a scavenging agent to selectively alleviate reactive oxygen species (ROS). Inhalation of hydrogen gas or use of hydrogen-containing aqueous solution can exert a powerful cellular protective effect.
In this review, we will focus on the physiological roles of hydrogen in kidney diseases, its potential as a therapeutic strategy, and the mechanisms that may be implicated in its protective effects. In addition, the findings of recent studies on hydrogen in various kidney disease models are discussed.

Action Mechanisms
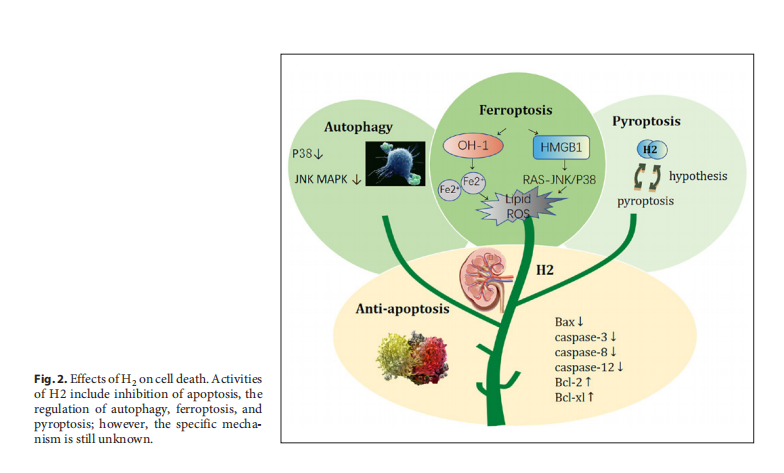
To fully explain the preventive and therapeutic effects of H2, Figures 1 and 2 show the biological effects of H2 and its impact on cell death.
Biological Effects of H2
1. Anti-Oxidation
H2, a potent oxidant, can react with nucleic acids without discrimination. The process of decomposing substances and releasing energy within or outside the body is known as oxidation. The intense oxidation of intracellular ROS or free radicals causes oxidative stress (OS), a state of imbalance between oxidation and antioxidant effects in the body which is considered to be a major contributor to aging and disease onset and progression. Acute OS induced by I/R or inflammation causes serious damage to tissues, and chronic OS is recognized as a causal agent of many kidney diseases. Hydrogen can scavenge free radicals. Hydrogen selectively reduces hydroxyl radicals (•OH) and peroxynitrite (ONOO−), which are strong oxidants that react indiscriminately with nucleic acids, lipids, and proteins, resulting in DNA fragmentation, lipid peroxidation, and protein inactivation [2]. The potential therapeutic benefits of H2 were first described in 2007. Ohsawa et al. [2] found that H2 selectively reduced •OH and ONOO− in cultured cells and examined whether H2 reduced the oxidized forms of biomolecules involved in metabolic oxidation-reduction reactions in cell-free assays. At room temperature and neutral pH, solutions saturated with H2 did not reduce the oxidized forms of nicotinamide adenine dinucleotide, the oxidized form of flavin adenine dinucleotide, or the oxidized form of cytochrome C. Thus, H2 did not affect oxidation-reduction metabolism or the levels of O2 −, H2O2, and NO, all of which play important physiological roles at low concentrations. As regulatory signaling molecules, they participate in many signal transduction cascades and regulate biological processes such as apoptosis, cell proliferation, and differentiation. Thus, H2 has selective antioxidant properties that protect the brain against I/R injury by specifically neutralizing •OH and ONOO− but not O2 −, H2O2, and NO [2]. Unlike many strong reducing antioxidants, it does not affect essential physiological processes of the body [3]. In addition to directly neutralizing free radicals, hydrogen can activate the Nrf2/ARE pathway in vivo and in vitro, increasing the transcription of downstream antioxidant enzyme genes, such as heme oxygenase 1 (HO-1), and contributing to the antioxidant activity [4]. Hydrogen can also increase the expression of endogenous superoxide dismutase, catalase [5], and reduced glutathione (GSH) [6], all of which help to reduce oxidative damage. Hydrogen plays various roles in the selective scavenging of ROS, downstream cascade signal transduction, and regulation of antioxidant enzyme activity. Therefore, studies on the antioxidant mechanisms of hydrogen and its influence in reducing damage due to OS in the kidneys provide a new direction for the future treatment of kidney diseases.
2. Immune System
Basic and clinical studies have shown that H2 is an important regulator with antioxidant, anti-inflammatory, and antiapoptotic effects [7]. H2 showed to have an anti-inflammatory effect in LPS-activated macrophages, according to Hong et al. [8], by inhibiting the release of proinflammatory cytokines and increasing the release of the anti-inflammatory cytokine, which could be mediated by HO-1. Subsequently, some researchers found that resuscitation with hydrogen-enriched saline could significantly improve tissue and organ damage, potentially lowering the inflammatory responses and OS by inhibiting the expression and activation of NF-κB [9]. H2 was also able to inhibit the expression of pro-inflammatory cytokines during inflammation and reduce the early overexpression of pro-inflammatory cytokines, such as interleukin (IL)- 1β, IL-6, IL-8, IL-10, and tumor necrosis factor-alpha (TNF-α) in many animal models [10]. The network of inflammatory mediators and inflammatory effector cells and the imbalance between pro-inflammatory cytokines and anti-inflammatory cytokines play an important role in the occurrence and progression of kidney diseases. Thus, the treatment of inflammatory kidney diseases from the perspective of pro-inflammatory cytokines and anti-inflammatory cytokines may be an interesting route for further investigations of the role of hydrogen in regulating and maintaining homeostasis in inflammatory kidney disease.
Diabetic nephropathy (DN) is a serious complication of diabetes mellitus. Metabolic disorders can occur in DN and manifest as local inflammation of the kidney that can lead to fibrosis and structural remodeling of the organ. Therefore, tackling immune-mediated inflammation is very significant for the treatment of DN [11]. In AKIs, such as renal artery infarction or toxin-mediated kidney injury, immune cells in the kidney are activated. Damaged renal epithelial cells activate the stress response pathways, leading to the secretion of cytokines and vasoactive factors, resulting in immunopathological damage [12]. Hydrogen, on the other hand, can suppress the production of immunoreactive substances [13]. We speculate that hydrogen has a future in immunological kidney disease as it is closely linked to significant antioxidant effects. However, there is currently no clinical data to confirm this. Further studies are required to confirm this theory.

Cistanche extract and Cistanche powder
3. Regulation of ER Stress
Endoplasmic reticulum (ER) stress occurs when pathological stress induces an accumulation of unfolded proteins in the ER. Zhao et al. [14] observed that the inhaled hydrogen significantly reduced the ER stress-related protein levels and alleviated tissue damage in myocardial IRI. Later, it was found that the mixture of H2 and O2 could inhibit ER stress via the PKR-like ER-localized eIF2α kinase-eukaryotic initiation factor 2 alpha-activating transcription factor 4 (PERK-eIF2α-ATF 4), inositol-requiring enzyme 1-X-box binding protein 1 (IRE 1-XBP1), and ATF 6 pathways. A study on the relationship between H2 and ER stress in rats with IRI found that H2 reduced the expression of GRP78 and TNF receptor-associated factor 2 [15], indicating that the protective effects of H2 on myocardial IRI are related to a decrease in ER stress. In terms of kidney disease, studies focusing on the influence of hydrogen as a down-regulator of ER stress are scarce; however, we consider this to be an interesting future line of research.
4. Mitochondria Quality Control
The urate-induced inflammasome pathway in urate nephropathy involves the entry of urate crystals into intracellular lysosomes, which are decomposed to produce mitochondrial ROS, activating NLRP3 inflammasomes [16]. Excessive ROS production is due to the release of calcium in ER, leading to mitochondrial depolarization, and mitochondrial membrane potential loss. Mitochondrial depolarization leads to the release of more ROS in the mitochondria. The negative regulation of ROS by hydrogen may help to maintain mitochondrial function [13, 17]. Currently, the regulation of the mitochondrial function using hydrogen has not been yet reported in the treatment of kidney diseases, but this could be a promising research area.

Herba Cistanche
Effects of H2 on Cell Death
1. Anti-apoptosis
Apoptosis is a form of programmed cell death characterized by cell shrinkage, apoptotic body formation, karyorrhexis, and chromatin condensation. Apoptosis can be induced by both endogenous and exogenous pathways. H2 plays an antiapoptotic role by upregulating or downregulating apoptotic-related factors. H2 also inhibits the expression of the proapoptotic factors B-cell lymphoma- 2-associated X-protein (Bax), caspase-3, -8, and -12, and upregulates the antiapoptotic factors B-cell lymphoma-2 (Bcl-2) and B-cell lymphoma-extra-large (Bcl-xl) [18]. It was found that the intraperitoneal injection of a hydrogen-rich solution 10 min before skeletal muscle in I/R could reduce the expression of apoptotic protein Bax and cytochrome C (which can activate the expression of caspase-induced caspase cascade reaction), and increase the expression of antiapoptotic protein Bcl-2, thus alleviating skeletal muscle injury after reperfusion [19]. Other scholars have found that intraperitoneal injection of a hydrogen-rich solution can significantly increase the survival rate of skin flaps, which is related to the regulation of the ASK-1/JNK pathway and Bax/Bcl-2 ratio [20]. However, the exact mechanism of the antiapoptotic effect of hydrogen in kidney disease is not completely clear.
2. Autophagy
Autophagy is an important mechanism for maintaining cell homeostasis and promotes energy utilization [21]. Autophagy has a significant impact on renal function and homeostasis. In kidney studies using adult animals, autophagy has been found to affect different types of renal cells to help maintain kidney pathology and homeostasis [22]. Excessive autophagy or autophagy-related stress can aggravate the inflammatory damage of tissues and organs. When protein aggregates become toxic, autophagy is activated, and once excessive autophagy causes tissue damage, autophagy is blocked. H2 appears to play a regulatory role [7]. Guan et al. [23] revealed that H2 was able to ameliorate chronic intermittent hypoxia (CIH)-induced kidney injury by decreasing ER stress and activating autophagy by inhibiting OS-dependent p38 and JNK MAPK activation. However, additional animal and clinical trials are needed, to further explore the underlying mechanisms.

the effects of Cistanche
3. Pyroptosis
The definition of pyroptosis was first proposed in 2012. It is accompanied by the accumulation of reactive lipid oxygen catalyzed by iron, cell apoptosis, necrosis, and autophagy [24]. In recent years, there has been evidence that pyroptosis plays an important role in the occurrence and development of AKI [25, 26], renal fibrosis [27], polycystic kidney disease [28], and renal cell carcinoma [24, 29]. Since there is considerable overlap between hydrogen regulation and pyroptosis pathways, we hypothesized that hydrogen could play a role in the treatment of renal diseases by regulating pyroptosis pathways. There is very little research on this matter, so additional research may be needed to confirm this hypothesis.
4. Ferroptosis
Ferroptosis is morphologically, biochemically, and genetically distinct from apoptosis, autophagy, and various forms of necrosis. It is characterized by the iron-dependent accumulation of ROS and lipid peroxidation, and it can be suppressed by iron chelators, lipophilic antioxidants, and inhibitors of lipid peroxidation [24]. A recent study [30] showed that HMGB1 is a novel ferroptosis regulator via the RAS-JNK/p38 pathway. Yu et al. [31] demonstrated that the treatment with H2 in the form of gas reduced the levels of HMGB1. Therefore, we speculate that H2 may be a potential drug agent for kidney diseases. Ferroptosis is an iron-dependent form of regulation of nonapoptotic cell death, which contributes to damage in models of AKI. HO-1, a potential source of intracellular iron, is a cytoprotective enzyme induced by cellular stress.
Due to its antiapoptotic and anti-inflammatory properties [32], it has a protective effect on AKI. HO-1 activity can be increased by hydrogen. A recent study [32] demonstrated that HO-1-deficient renal epithelial cells were more sensitive to ferroptosis, indicating that free iron produced by HO-1 does not promote ferroptosis itself, and HO-1 has an anti-ferroptosis effect. Although the mechanisms behind hydrogen’s effect on ferroptosis have yet to be fully clarified, it may add new directions to the search for kidney disease treatments.
References
1 Dole M, Wilson FR, Fife WP. Hyperbaric hydrogen therapy: a possible cancer treatment. Science. 1975;190(4210):152–4.
2 Ohsawa I, Ishikawa M, Takahashi K, Watanabe M, Nishimaki K, Yamagata K, et al. Hydrogen acts as a therapeutic antioxidant by selectively reducing cytotoxic oxygen radicals. Nat Med. 2007;13(6):688–94.
3 Ohta S. Recent progress toward hydrogen medicine: the potential of molecular hydrogen for preventive and therapeutic applications. Curr Pharm Des. 2011;17(22):2241–52.
4 Xie Q, Li XX, Zhang P, Li JC, Cheng Y, Feng YL, et al. Hydrogen gas protects against serum and glucose deprivation-induced myocardial injury in H9c2 cells through activation of the NF-E2-related factor 2/heme oxygenase 1 signaling pathway. Mol Med Rep. 2014;10(2): 1143–9.
5 Wang F, Yu G, Liu SY, Li JB, Wang JF, Bo LL, et al. Hydrogen-rich saline protects against renal ischemia/reperfusion injury in rats. J Surg Res. 2011;167(2):e339–44.
6 Qian L, Cao F, Cui J, Huang Y, Zhou X, Liu S, et al. Radioprotective effect of hydrogen in cultured cells and mice. Free Radic Res. 2010; 44(3):275–82.
7 Huang CS, Kawamura T, Toyoda Y, Nakao A. Recent advances in hydrogen research as a therapeutic medical gas. Free Radic Res. 2010; 44(9):971–82.
8 Chen HG, Xie KL, Han HZ, Wang WN, Liu DQ, Wang GL, et al. Heme oxygenase-1 mediates the anti-inflammatory effect of molecular hydrogen in LPS-stimulated RAW 264.7 macrophages. Int J Surg. 2013;11(10):1060–6.
9 Wang X, Yu P, Yang Y, Liu X, Jiang J, Liu D, et al. Hydrogen-rich saline resuscitation alleviates inflammation induced by severe burns with delayed resuscitation. Burns. 2015;41(2): 379–85.
10 Liu W, Shan LP, Dong XS, Liu XW, Ma T, Liu Z. Combined early fluid resuscitation and hydrogen inhalation attenuates lung and intestine injury. World J Gastroenterol. 2013; 19(4):492–502.
11 Zheng Z, Zheng F. Immune cells and inflammation in diabetic nephropathy. J Diabetes Res. 2016;2016:1841690.
12 Yatim KM, Lakkis FG. A brief journey through the immune system. Clin J Am Soc Nephrol. 2015;10(7):1274–81.
13 Yang M, Dong Y, He Q, Zhu P, Zhuang Q, Shen J, et al. Hydrogen: a novel option in human disease treatment. Oxid Med Cell Longev. 2020;2020:8384742.
14 Zhang Y, Liu Y, Zhang J. Saturated hydrogen saline attenuates endotoxin-induced lung dysfunction. J Surg Res. 2015;198(1):41–9.
15 Huang T, Wang W, Tu C, Yang Z, Bramwell D, Sun X. Hydrogen-rich saline attenuates ischemia-reperfusion injury in skeletal muscle. J Surg Res. 2015;194(2):471–80.
16 Liu YQ, Liu YF, Ma XM, Xiao YD, Wang YB, Zhang MZ, et al. Hydrogen-rich saline attenuates skin ischemia/reperfusion-induced apoptosis via regulating Bax/Bcl-2 ratio and ASK- 1/JNK pathway. J Plast Reconstr Aesthet Surg. 2015;68(7):e147–56.
17 Zhong H, Song R, Pang Q, Liu Y, Zhuang J, Chen Y, et al. Propofol inhibits parthanatos via ROS-ER-calcium-mitochondria signal pathway in vivo and in vitro. Cell Death Dis. 2018;9(10):932.
18 Zhao YS, An JR, Yang S, Guan P, Yu FY, Li W, et al. Hydrogen and oxygen mixture to improve cardiac dysfunction and myocardial pathological changes induced by intermittent hypoxia in rats. Oxid Med Cell Longev. 2019; 2019:7415212.
19 Gao Y, Yang H, Chi J, Xu Q, Zhao L, Yang W, et al. Hydrogen gas attenuates myocardial ischemia-reperfusion injury independent of postconditioning in rats by attenuating endoplasmic reticulum stress-induced autophagy. Cell Physiol Biochem. 2017;43(4):1503–14.
20 Isaka Y, Takabatake Y, Takahashi A, Saitoh T, Yoshimori T. Hyperuricemia-induced inflammasome and kidney diseases. Nephrol Dial Transplant. 2016;31(6):890–6.
21 Parzych KR, Klionsky DJ. An overview of autophagy: morphology, mechanism, and regulation. Antioxid Redox Signal. 2014;20(3): 460–73.
22 He L, Livingston MJ, Dong Z. Autophagy in acute kidney injury and repair. Nephron Clin Pract. 2014;127(1–4):56–60.
23 Guan P, Sun ZM, Luo LF, Zhou J, Yang S, Zhao YS, et al. Hydrogen protects against chronic intermittent hypoxia-induced renal dysfunction by promoting autophagy and alleviating apoptosis. Life Sci. 2019;225:46–54.
24 Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149(5):1060– 72.
25 Friedmann Angeli JP, Schneider M, Proneth B, Tyurina YY, Tyurin VA, Hammond VJ, et al. Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice. Nat Cell Biol. 2014;16(12):1180–91.
26 Müller T, Dewitz C, Schmitz J, Schröder AS, Bräsen JH, Stockwell BR, et al. Necroptosis and ferroptosis are alternative cell death pathways that operate in acute kidney failure. Cell Mol Life Sci. 2017;74(19):3631–45.
27 Hou W, Xie Y, Song X, Sun X, Lotze MT, Zeh HJ 3rd, et al. Autophagy promotes ferroptosis by the degradation of ferritin. Autophagy. 2016; 12(8):1425–8.
28 Schreiber R, Buchholz B, Kraus A, Schley G, Scholz J, Ousingsawat J, et al. Lipid peroxidation drives renal cyst growth in vitro through the activation of TMEM16A. J Am Soc Nephrol. 2019;30(2):228–42.
29 Yang WS, SriRamaratnam R, Welsch ME, Shimada K, Skouta R, Viswanathan VS, et al. Regulation of ferroptosis cancer cell death by GPX4. Cell. 2014;156(1–2):317–31.
30 Ye F, Chai W, Xie M, Yang M, Yu Y, Cao L, et al. HMGB1 regulates elastin-induced ferroptosis via RAS-JNK/p38 signaling in HL-60/ NRAS(Q61L) cells. Am J Cancer Res. 2019; 9(4):730–9.
31 Yu Y, Yang Y, Yang M, Wang C, Xie K, Yu Y. Hydrogen gas reduces HMGB1 release in lung tissues of septic mice in an Nrf2/HO- 1-dependent pathway. Int Immunopharmacol. 2019;69:11–8.
32 Adedoyin O, Boddu R, Traylor A, Lever JM, Bolisetty S, George JF, et al. Heme oxygenase-1 mitigates ferroptosis in renal proximal tubule cells. Am J Physiol Renal Physiol. 2018; 314(5):F702–14.
Bo Wang a Zhuoshu Li b, c Longfei Maod Mingyi Zhaob Bingchang Yange Xiaowu Taoa Yuxiang Lia Guangming Yina
a Department of Urology, The Third Xiangya Hospital, Central South University, Changsha, China;
bDepartment of Pediatrics, The Third Xiangya Hospital, Central South University, Changsha, China;
c Xiangya School of Medicine, Central South University, Changsha, China;
d Bioinformatics Center, College of Biology, Hunan University, Changsha, China;
e Department of Critical Care Medicine, Central South University, Changsha, China






