PartⅠMetabolic Understanding Of The Genetic Dysregulation in The Tumor Microenvironment Of Kidney Renal Clear Cell Carcinoma
May 08, 2023
Abstract
Metabolic dysregulation is a hallmark of cancers including KIRC, specifically caused by alterations in metabolic genes. Currently, a lack of consensus exists between metabolic signatures in the tumor microenvironment. In this study, we observed the significant correlations of differentially expressed metabolic genes (DEmGs) between KIRC and the related normal samples. Briefly, we collected sets of metabolic genes through RNA-seq data of KIRC and normal tissues from TCGA, followed by identifying KIRC-related DEmGs. Next, patients were classified into three clusters, and using WGCNA, we identified metabolic genes involved in the survival among different clusters.
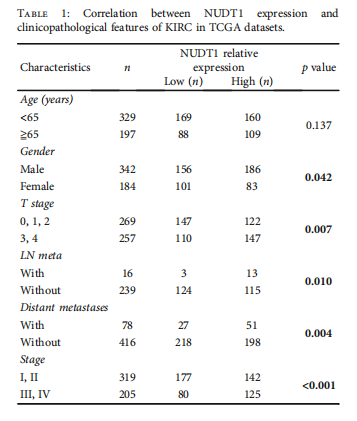
Furthermore, we investigated survival and clinical parameters along with immune infiltration in the clusters. At the same time, we constructed and validated a prediction model based on these DEmGs. These analyses revealed that the patients having high expression of DEmGs showed poor survival, while infiltration of less-immune cells was associated with the metastasis of KIRC. In the end, we identified NUDT1 as a hub gene as it showed significantly high expression in KIRC samples as well as associated with the survival and prognosis of the patients. Further analysis revealed the oncogenic role of NUDT1 in 786-O and ACHN cells. Thus, we conclude that NUDT1 could be a potential diagnostic and prognostic marker for KIRC.
Keywords
Metabolic dysregulation; Differentially expressed metabolic genes (DEmGs); Survival rate; NUDT1; Cistanche benefits.
Click here to know Cistanche's effects
Introduction
Kidney renal clear cell carcinoma (KIRC) is a type of renal cell carcinoma (RCC), considered one of the common cancers which accounts for 70–80% of cases [1], ranked as the 16th most common cause of cancer-related mortality worldwide [2]. Renal cell carcinoma (RCC) is the most common type of renal cancer and accounts for 90% of kidney cancer cases [3]. Recently, bioinformatics-based identification of the potential markers in cancers is being widely used but only a few reliable biomarkers of KIRC have been identified or most of the markers are so far not fully validated. Thus, bioinformatics analyses coupled with experimental validations are necessary to elucidate the potential mechanism of the biogenesis and progression of KIRC [4]. Deep exploration of the tumor microenvironment and the development of immunotherapy have made it possible to study the interaction between tumors and the immune system [5]. Yet, many of the identified biomarkers have not been studied for their effect on the cellular phenotype and respective underlying molecular mechanisms in the KIRC [6]. However, the prognosis of KIRC patients is poorly understood and the overall 5-year survival is less than 10% after first diagnosis [7, 8]. Generally, the normal tissues predominantly contain high levels of antiangiogenic factors. The disturbance in the level of these factors may activate the proangiogenic factors which further increase the division of cells at an abnormal rate leading to tumor formation [9]. In this process, tumor cells express a high level of proangiogenic growth factor and this effect supports the development of new blood vessels in the tumors [10]; thus, the development of the tumor begins. Hence, many other factors are involved in tumorigenesis, including infiltration of the immune cells and the expression of various metabolic genes. It has been well known that the presence of different types of immune cells in the tumor microenvironment largely affects tumor progression and immunotherapy; thus, alterations of immune-related genes also affect the proportion and function of immune cells in the tumor microenvironment [11]. For example, the infiltration of immune cells in the tumor microenvironment significantly affects the development of glioma [12]. Despite the lacking interest in metabolic genes that influence cancers, the current studies have renewed that awareness of cancer as a metabolic disorder [13, 14]. These studies provide the base for including metabolic reprogramming as a new hallmark of malignant transformation [15]. However, the expression of all metabolic genes or pathways among different tissues including tumors and normal tissues differs from each other and it is largely unexplored, especially in KIRC.
Overall, there is a smaller number of reliable biomarkers available for the prediction of prognosis and immunotherapeutic responses in the KIRC; thus, it is hard to have complete clinical success. Therefore, comprehensive bioinformatics studies coupled with experimental validations could elucidate the potential mechanisms of the biogenesis and progression of KIRC. Thus, the current study is aimed at comprehensively assessing the effects of the metabolic pathways on the metabolic transcriptional profiles in KIRC compared with their matched normal tissues. Here, we successfully not only segregated the different disease sites or different molecular subtypes of the same disease but also predicted the response to metabolism-targeted therapy. This represents a new way of identifying a mechanism by which metabolic pathways are disturbed in the malignancy and offers novel targets for clinical interventions.

Standardized Cistanche
Material and Methods
1. Data Acquisitions.
The RNA sequencing data (FPKM) relevant to KIRC were directly downloaded from The Cancer Genome Atlas (TCGA). Subsequently, the data were processed according to desired downstream applications. In addition, clinicopathologic data for the corresponding KIRC patients, including gender, race, age, tumor location, histology classification, differentiation grade, tumor stage, and survival information, were obtained from UCSC Cancer Browser. To gain insights into the metabolic heterogeneity of KIRC patients, the metabolic gene sets were taken from previous publications [16, 17]. The graphical abstract of this study is shown in Supplementary Figure 1, and the detailed workflow of data acquisition and downstream process study is shown in Supplementary Figure 2.

2. Identification of the Differentially Expressed Metabolic Genes (DEmGs).
The sets of metabolic genes were identified by intersections of two datasets and used for DEG analysis between tumor and normal samples. Later, we also identified DEmGs using WGCNA, and based on survival modes, patients classified them into three clusters. In short, the patients were divided into three clusters, namely, cluster C1, cluster C2, and cluster C3. To identify DEGs, linear models were used by employing the R package limma. A false discovery rate (FDR) adjusted p-value < 0.05 combined with a simultaneous absolute value of log2 fold change, logFC > 1, and logFC < −1 were set as the threshold for DEG identification. The volcano plots were made using ggplot2. The genes with logFC > 1 were considered upregulated, and those with logFC < −1 were assigned as downregulated genes.
3. Functional Enrichment Analyses and Protein-Protein Interaction.
The enrichment analyses by GO and KEGG terms were performed through the R package Cluster profile. The GO terms were divided into three categories including molecular functions (MF), cellular components (CC), and biological processes (BP). To determine the significant differences and correlations of DEmGs between tumor and normal tissues, we used computational software GSEA (gene set enrichment analysis). KEGG and GO (Gene Ontology) enrichment analyses of the DEGs were performed to identify potential pathways and functions. Furthermore, the cystoscope application was used to construct the protein-protein interaction (PPI) network of DEmGs. The initial PPI was obtained from the STRING database. The highest confidence limit was set to 0.9.
4. Survival Analysis.
The differentially expressed genes in renal cancer patients were identified through the limma package, and the differentially expressed genes were analyzed by using the WGCNA package to perform coexpression, to construct a coexpression network that identified a total of 6 modules. β = 5 and the network is a scale-free network. The prognostic significance of DEmGs identified by WGCNA in survival mode was determined by Kaplan-Meier (KM) plots with the log-rank test.

Cistanche pills
5. Immune Infiltration Analysis.
The immune and stromal cells were calculated using the R package, “xCELL; ” samples with p < 0:05 were selected for further analysis. Furthermore, the immune cells containing biological markers were analyzed by the R package GSVA. Moreover, to analyze the purity of the tumor, we used the ESTIMATE algorithm to estimate the stromal and immune scores of a series of KIRC tissues.
6. Construction of the Prediction Model.
The differentially expressed metabolic genes were obtained from RNA sequencing data of KIRC tumors with survival information, and the patients were randomly divided into training and testing cohorts using the R package “caret;” then, univariate survival analysis was performed on the DEmGs. Furthermore, LASSO analysis was performed by R package “glmnet;” and for optimization of the prediction model, a stepwise proportional hazards model was used.
7. Cell Culture.
The human renal cancer cell lines 786-O and ACHN were purchased from the American Type Culture Collection (ATCC) (Manassas, VA, USA). The 786-O cell lines were cultured in RPMI 1640 medium (cat. no. 11875093, Invitrogen, Carlsbad, CA, United States) supplemented with 10% fetal bovine serum (cat. no. 04-001- 1ACS, Biological Industries, Israel). The ACHN cell lines were cultured in MEM medium (cat. no. 41500034, Invitrogen, Carlsbad, CA, United States) supplemented with 10% fetal bovine serum. All cultures were incubated at 37° C and 5% CO2 in a humidified incubator.
8. Real-Time Quantitative PCR.
Total RNA from the KIRC cell lines and tissues was isolated by using TRIzol (cat. no. 9109; TaKaRa, Tokyo, Japan) according to the manufacturer’s protocols. RT-qPCR was performed using QuantiNova SYBR Green PCR Kit (cat. no. 208054, QIAGEN, Duesseldorf, Germany). Relative gene expression was calculated by the 2−ΔΔCt method. The primer sequences are listed in Supplementary Table 1.
9. Cell Viability Assay.
The cytotoxicity after NUDT1 siRNA transfection in 786-O and ACHN was determined using a Cell Counting Kit-8 (CCK-8) assay (cat. no. C0038, Beyotime Biotechnology Co. Ltd., China). The NUDT1 siRNA sequences are provided in Supplementary Table 1. They were synthesized from GenePharma (Shanghai, China). Briefly, both cell lines 786-O and ACHN in the logarithmic growth phase (24 h after siRNA transfection) were collected and dispensed into 96-well cell culture plates (1000 cells/well). The following day, 10 μl of CCK-8 was added to each well. After incubation at 37° C for a further 2 h, the optical density (OD) at 450 nm of cells was detected via a microplate reader (Bio-Rad Laboratories, Richmond, CA, USA).
10. Cell Migration, Invasion, and Apoptosis Assay.
KIRC cells 786-O and ACHN were transfected for siRNA-mediated knockdown of the NUDT1. After 24 h of the transfection, cells were seeded into the 6-well plates for wound healing assay. After the cells obtained 100% confluence, the wounds were generated in the cells as a monolayer using a plastic pipette tip. The cells were then rinsed with PBS and cultured for another 48 h. The distance of wound closure was visualized by a microscope and photographed for measuring the effect of NUDT1 knockdown on KIRC 786-O and ACHN cells. Cell migration & invasion were performed using transwell 24-well plates (Corning, New York, NY). In short, an equal number of the cells were cultured in the FBS-free media in the upper chamber, and migration of the cells was observed in the lower chamber of the transwell where media was supplemented with 10% FBS. Transwell chambers were carefully washed and stained with 0.2% crystal violet, visualized, and photographed; later on, differences between migrated cells between negative control and NUDT1 knockdown were determined by simply cell counting. Apoptosis assay was performed after 72 h of the transfection, cells were harvested, washed, and resuspended in ice-cold PBS. Cells were then detected by the Apoptosis Detection Kit (cat. no. KGA1013, KeyGen BioTech, Nanjing, China) according to the manufacturer’s instructions and examined by flow cytometry (FACScan; BD Biosciences). All experiments were performed in triplicate.
11. Statistical Analysis.
All the statistical analyses were performed at GraphPad prism (V7.0) and R (3.6.4), the Wilcox test was used to compare the infiltration of immune cells in normal and tumor tissues, and the chi-square test was performed for studying the correlation between immune cell infiltration and pathological parameters. Analysis of the variance was used to compare the immune score, stromal score, and tumor purity among the three clusters. For the survival analysis, the p-value was calculated using the log-rank test. A p-value of <0.05 was considered statistically significant.

Cistanche supplements
References
[1] V. Nerich, M. Hugues, T. Nai, et al., “Clinical impact of targeted therapies in patients with metastatic clear-cell renal cell carcinoma,” Oncotargets and Therapy, vol. 7, pp. 365–374, 2014.
[2] A. Znaor, J. Lortet-Tieulent, M. Laversanne, A. Jemal, and F. Bray, “International variations and trends in renal cell carcinoma incidence and mortality,” European Urology, vol. 67, no. 3, pp. 519–530, 2015.
[3] B. Ljungberg, L. Albiges, Y. Abu-Ghanem, et al., “European Association of Urology Guidelines on Renal Cell Carcinoma: The 2019 Update,” European Urology, vol. 75, no. 5, pp. 799– 810, 2019.
[4] W. Zheng, S. Zhang, H. Guo, et al., “Multi-omics analysis of tumor angiogenesis characteristics and potential epigenetic regulation mechanisms in renal clear cell carcinoma,” Cell Communication and Signaling: CCS, vol. 19, no. 1, p. 39, 2021.
[5] J. Galon and D. Bruni, “Approaches to treating immune hot, altered and cold tumors with combination immunotherapies,” Nature Reviews Drug Discovery, vol. 18, no. 3, pp. 197– 218, 2019.
[6] W. du, L. Zhang, A. Brett-Morris et al., “HIF drives lipid deposition and cancer in ccRCC via repression of fatty acid metabolism,” Nature Communications, vol. 8, no. 1, p. 1769, 2017.
[7] A. Jemal, R. Siegel, E. Ward, et al., “Cancer statistics, 2008,” CA: A Cancer Journal for Clinicians, vol. 58, no. 2, pp. 71–96, 2008.
[8] I. Lucca, T. Klatte, H. Fajkovic, M. de Martino, and S. F. Shariat, “Erratum: Gender differences in incidence and outcomes of urothelial and kidney cancer,” Nature Reviews Urology, vol. 12, no. 12, p. 653, 2015.
[9] I. J. Fidler, “Angiogenesis and cancer metastasis,” Cancer Journal, vol. 6, Supplement 2, pp. S134–S141, 2000.
[10] C. Kut, F. Mac Gabhann, and A. S. Popel, “Where is VEGF in the body? A meta-analysis of VEGF distribution in cancer,” British Journal of Cancer, vol. 97, no. 7, pp. 978–985, 2007.
[11] F. Ren, Q. Zhao, M. Zhao, et al., “Immune infiltration profiling in gastric cancer and their clinical implications,” Cancer Science, vol. 112, no. 9, pp. 3569–3584, 2021.
[12] X. Ge, Z. Wang, R. Jiang et al., “SCAMP4 is a novel prognostic marker and correlated with the tumor progression and immune infiltration in glioma,” The International Journal of Biochemistry & Cell Biology, vol. 139, 2021.
[13] M. G. Vander Heiden and R. J. DeBerardinis, “Understanding the intersections between metabolism and cancer biology,” Cell, vol. 168, no. 4, pp. 657–669, 2017.
[14] D. S. Wishart, “Is cancer a genetic disease or a metabolic disease?” biomedicine, vol. 2, no. 6, pp. 478-479, 2015.
[15] D. Hanahan and R. A. Weinberg, “Hallmarks of cancer: the next generation,” Cell, vol. 144, no. 5, pp. 646–674, 2011.
[16] X. Peng, Z. Chen, F. Farshidfar, et al., “Molecular characterization and clinical relevance of metabolic expression subtypes in human cancers,” Cell Reports, vol. 23, no. 1, pp. 255–269.e4, 2018.
[17] S. Haider, A. McIntyre, R. G. van Stiphout, et al., “Genomic alterations underlie a pan-cancer metabolic shift associated with tumor hypoxia,” Genome Biology, vol. 17, no. 1, p. 140, 2016.
Junwei Xie,1,2,3,4,5 Lingang Cui,6 Shaokang Pan,1,2,3,4,5 Dongwei Liu,1,2,3,4,5 Fengxun Liu,1,2,3,4,5 and Zhangsuo Liu 1,2,3,4,5
1 Department of Nephrology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, China
2 Research Institute of Nephrology, Zhengzhou University, Zhengzhou 450052, China
3 Research Center for Kidney Disease, Zhengzhou, 450052 Henan, China
4 Key Laboratory of Precision Diagnosis and Treatment for Chronic Kidney Disease in Henan Province, Zhengzhou 450052, China
5 Core Unit of National Clinical Medical Research Center of Kidney Disease, Zhengzhou 450052, China
6 Department of Urology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, China






