Part Ⅰ Smad3 Deficiency Improves Islet-based Therapy For Diabetes And Diabetic Kidney Injury By Promoting β Cell Proliferation Via The E2F3-dependent Mechanism
May 25, 2023
Abstract
1. Rationale
Poor β cell proliferation is one of the detrimental factors hindering islet cell replacement therapy for patients with diabetes. Smad3 is an important transcriptional factor of TGF-β signaling and has been shown to promote diabetes by inhibiting β cell proliferation. Therefore, we hypothesize that Smad3-deficient islets may be a novel cell replacement therapy for diabetes.
2. Methods
We examined this hypothesis in streptozocin-induced type-1 diabetic mice and type-2 diabetic db/db mice by transplanting Smad3 knockout (KO) and wild-type (WT) islets under the renal capsule, respectively. The effects of Smad3KO versus WT islet replacement therapy on diabetes and diabetic kidney injury were examined. In addition, RNA-seq was applied to identify the downstream target gene underlying Smad3-regulated β cell proliferation in Smad3KO-db/db versus Smad3WT-db/db mouse islets.
3. Results
Compared to Smad3WT islet therapy, treatment with Smad3KO islets produced a much better therapeutic effect on both type-1 and type-2 diabetes by significantly lowering serum levels of blood glucose and HbA1c and protecting against diabetic kidney injuries by preventing an increase in serum creatinine and the development of proteinuria, mesangial matrix expansion, and fibrosis. These were associated with a significant increase in grafted β cell proliferation and blood insulin levels, resulting in improved glucose intolerance. Mechanistically, RNA-seq revealed that compared with Smad3WT-db/db mouse islets, deletion of Smad3 from db/db mouse islets markedly upregulated E2F3, a pivotal regulator of cell cycle G1/S entry. Further studies found that Smad3 could bind to the promoter of E2F3, and thus inhibit β cell proliferation via an E2F3-dependent mechanism as silencing E2F3 abrogated the proliferative effect on Smad3KO β cells.
4. Conclusion
Smad3-deficient islet replacement therapy can significantly improve both type-1 and type-2 diabetes and protect against diabetic kidney injury, which is mediated by a novel mechanism of E2F3-dependent β cell proliferation.
Keywords
Smad3, islet transplantation, diabetes, E2F3, cell cycle.

Click here to get Cistanche benefits
Introduction
type-1 and 2 diabetes mellitus (T1DM and T2DM). β cell or islet transplantation is an attractive therapy and has been successfully applied in patients with T1DM. The islets or β cells used for transplantation are usually from the cadaveric donor or induced pluripotent stem cells (iPSC) [1, 2]. More than 1500 cases of clinical islet transplantation have been completed in patients with T1DM following the well-established Edmonton Protocol [1]. Although about 50% of patients hold insulin independence for 5 years after transplantation, engraftment failure is a common concern clinically. One of the factors that hinder islet transplantation is the extremely low proliferative rate of the grafted islet β cells [3]. In addition, the hazardous stimuli from the graft microenvironment such as inflammation, immune allograft rejection, and metabolic stress may cause transplanted β cells to undergo apoptosis and necrosis, resulting in progressive graft loss [1]. Therefore, improvement of the transplanted β cell proliferation is pivotal for the success of islet transplantation-based treatment for diabetes.
Transforming growth factor-beta (TGF-β) signaling plays pleiotropic roles in many biological processes. The signal transduction initiates by the binding of the TGF-β ligand to its type 2 receptor TGFBR2. The latter recruits and activates type 1 receptor TGFBR1 by phosphorylation. Activated TGFBR1 further phosphorylates the receptor-regulated Smad proteins (R-Smads), Smad2, and Smad3. The R-Smads form a complex with Smad4, which translocates into the nucleus to transcriptionally regulate target genes [4, 5]. It is reported that the pancreatic islet maintains universally activated TGF-β signaling as revealed by a relatively high level of phosphorylated Smad2/3 in the physiological conditions. However, in response to insulin demand, the TGF-β signaling activity is decreased, accompanied by increased β cell proliferation [6]. In addition, pharmacological inhibition of TGF-β signaling promotes β cell proliferation in vitro and in vivo [7, 8]. More importantly, the deletion of Smad3 results in more robust β cell proliferation compared with the deletion of Smad2 in mice with partial pancreatectomy, indicating that Smad3 but not Smad2 is the primary effector downstream of TGF-β signaling to regulate β cell proliferation [6]. Later studies show that Smad3 suppresses β cell proliferation by inducing the expression of cyclin-dependent kinase inhibitors (CDKIs), including p16, p21, and p57 [7, 8].
Our recent work also found that the deletion of Smad3 in db/db mice (Smad3KO-db/db) prevents the onset of overt diabetes without obesity, hyperglycemia, insulin resistance, and glucose intolerance. Interestingly, Smad3KO-db/db mice show islet hyperplasia with significantly increased β cell proliferation and persistent hyperinsulinemia [9]. This implies that Smad3 is pathogenic in diabetes and targeting Smad3 in β cells may represent a novel β cell or islet replacement therapy for diabetes. This was examined in the present study by using Smad3 knockout (Smad3KO) islet transplantation in streptozocin (STZ)-induced type-1 diabetic mice and in type-2 diabetic db/db mice.

Cistanche supplements
Results
1. Smad3KO islet transplantation produced a better therapeutic effect on STZ-induced type-1 diabetes
To compare the therapeutic efficacy of Smad3KO versus Smad3WT islet on diabetes, we first performed syngeneic islet transplantation under the renal capsule of STZ-induced diabetic mice where hyperglycemia is caused by β cell deficiency. Diabetic mice on day 7 after STZ injection were transplanted with 3 doses of islets (50, 100, and 200 islets per mouse) that were freshly isolated from Smad3WT or Smad3KO mice. Both random blood glucose (RBG) and fasting blood glucose (FBG) levels were monitored weekly for 16 weeks. The transplantation doses were selected based on the previous findings that transplantation of 300 islets is sufficient to restore euglycemia in β cell-depleted mice rapidly [10]. As shown in Figures 1A to D, the diabetic mice with sham operation developed persistent hyperglycemia over 16 weeks. Transplantation with a low dose (50 islets) of Smad3KO or WT islets failed to reduce both RBG and FBG levels. However, diabetic mice receiving 100 Smad3KO islets, but not the same dose of Smad3WT islets, gradually recovered the normal RBG and FBG similar to those receiving 200 Smad3KO or WT islets. We also found that the blood-glucose-lowering effect is solely attributed to the transplanted islets as hyperglycemia recurred immediately once the islet-bearing kidney was removed from 16 weeks onwards (arrow, Figure 1A).

Impaired glucose tolerance was also developed in the sham-operated diabetic mice, which was significantly relieved by transplanting the high dose (200) of Smad3KO or WT islets and 100 Smad3KO islets, but not by the low dose of 50 Smad3KO or WT islets and 100 Smad3WT islets (Figure 1E and G). As expected, STZ-induced diabetic mice didn’t develop insulin resistance.
In contrast, prolonged response to insulin was observed in hyperglycemic mice with the sham operation or ineffective therapy with 50 Smad3WT or KO islets and 100 Smad3WT islets (Figure 1F and H). In line with the recovered euglycemia and improved glucose intolerance, diabetic mice given 200 Smad3KO or WT islets and 100 Smad3KO islets, but not 50 Smad3KO or WT islets and 100 Smad3WT islets, showed decreased blood HbA1c level accompanied by increased insulin level (Figure 1 I and J). Furthermore, STZ-induced diabetes caused growth retardation, which was partially recovered in mice transplanted with 200 Smad3KO or WT islets and 100 Smad3KO islets (Figure S1). In addition, STZ-induced diabetic mice also developed diabetic nephropathy at 16 weeks as demonstrated by mesangial expansion, interstitial fibrosis, proteinuria, and increased serum creatinine (Figure 2). Intriguingly, treatment with 100 Smad3KO islets, but not 100 Smad3WT islets, resulted in protection against the development of diabetic kidney disease, although this was also notable in diabetic mice treated with the high dose of 200 Smad3KO or WT islets (Figure 2). These findings demonstrate a better therapeutic efficacy of Smad3KO islets on STZ-induced diabetes and suggest about a 50% reduction in the islets needed to achieve competent glycemic control and protection against diabetic kidney injury by using Smad3-deficient islet replacement therapy for diabetes.

2. Smad3KO islet transplantation produced a better therapeutic effect on type-2 diabetes in db/db mice
It has been reported that islet transplantation improves T2DM in a high-fat diet-fed mouse model challenged with a low dose of STZ [11]. We next investigated whether transplantation of Smad3KO islets also produces a better therapeutic effect on T2DM by treating db/db mice with 250 Smad3KO or WT islets from the pre-diabetic age of week 4 to week 12. We found that transplantation with either Smad3KO or WT islets did not influence the growth of recipient db/db mice (Figure S2). Although db/db mice receiving Smad3WT islet transplantation showed a trend decrease of RBG and FBG compared with the sham-operated db/db mice, db/db mice transplanted with the equivalent dose of Smad3KO islets showed a significant decrease in RBG and FBG (Figure 3A and B). Glucose intolerance developed in db/db mice was significantly improved by Smad3KO but not Smad3WT islet transplantation, although insulin resistance was not altered (Figure 3C-F). Consistently, compared with Smad3WT islet transplantation or sham operation, db/db mice treated with Smad3KO islet transplantation also presented a significant decrease in blood HbA1c levels, which was accompanied by a notable increase in levels of blood insulin (Figure 3G and H). Furthermore, treatment with 250 Smad3KO islets significantly attenuated glomerular mesangial expansion (Figure 4). However, since no significant changes in serum creatinine, proteinuria, and renal fibrosis were evidenced in db/db mice at the age of 12 weeks, no obvious alterations in these parameters were found in db/db mice treated with Smad3KO or WT islets (Figure 4). Collectively, these data again indicate that transplantation of Smad3KO islets produces a better treatment over Smad3WT islets in type-2 diabetic db/db mice.

3. Smad3 deficiency promotes β cell proliferation in the grafted islets in both STZ-induced diabetic mice and db/db mice.
We next investigated the potential mechanisms whereby transplantation of Smad3KO islets produces a better therapeutic effect on both T1DM and T2DM. We first examined the β cell mass in the grafted islets by immunostaining with insulin. In STZ-induced diabetes, we failed to detect the existence of insulin-positive cells under the renal capsule in mice engrafted with 50 Smad3KO or WT islets at week 16 after transplantation, indicating the loss of β cells. Unexpectedly, diabetic mice receiving 100 Smad3WT islets showed only a few dispersed insulin-producing β cell clusters, but this was largely increased in diabetic mice treated with 100 Smad3KO islets, resulting in nearly a 17-fold increase in the β cell mass (Figure 5A and B). Interestingly, diabetic mice given a high dose of 200 Smad3KO or WT islets exhibited a similar β cell mass (Figures 5A and B). These findings were consistent with the dose-dependent effect of islet transplantation on hyperglycemia as shown in Figures 1A and B.

We next investigated whether the increased β cell mass in Smad3KO islet grafts is associated with enhanced β cell proliferation. Two-color immunofluorescence showed that at week 16 post-islet transplantation, the mice treated with 100 Smad3WT islets showed undetectable or rare PCNA+ insulin+ cells, which was primarily increased in those receiving 100 Smad3KO islets or a high dose of 200 Smad3KO or WT islets (Figure 5C and D). We then investigated whether Smad3 controls the early β cell proliferation in the grafted islets under progressive diabetic conditions by BrdU labeling on day 7 post-islet transplantation when levels of blood glucose remained high. BrdU labeling revealed a 3-fold increase in BrdU+ insulin+ β cells in 100 Smad3KO islet grafts compared with the same dose of Smad3WT islet grafts (Figure 5E and F). This finding indicates that Smad3 deficiency largely promotes β cell proliferation through the S-phase cell cycle under hyperglycemic conditions, contributing to a 17-fold increase in the β cell mass as seen at week 16 post-islet transplantation (Figure 5A and B). Taken together, all these findings suggest that Smad3 deficiency largely promotes β cell proliferation, thereby restoring euglycemia in STZ-induced type-1 diabetes.

Similarly, high β cell proliferative activities were also detected in Smad3KO islet grafts in type-2 diabetic db/db mice as shown by a 5-fold increase in the β cell mass when compared to those with Smad3WT islet grafts at the age of week 12 after islet transplantation (Figure 6A and B), which was associated with a 2.5-fold increase in insulin+ PCNA+ cells (Figure 6C and D). By using BrdU-labelling, we also detected a 3-fold increase in insulin+ BrdU+ β cells in Smad3KO islet grafts on day 7 post-transplantation in 12-week-old db/db mice (Figure 6E and F). This finding again confirms that β cells lacking Smad3 are resistant to hyperglycemia and remain highly proliferative to maintain the β cell mass to improve T2DM.

4. Smad3 deficiency promotes β cell proliferation in vitro
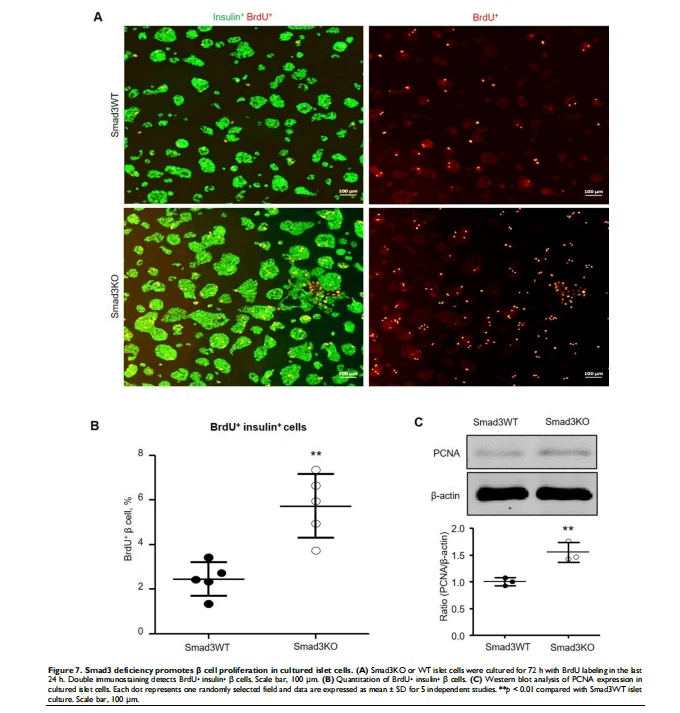
It has been reported that pharmacological inhibition of TGFBR1 promotes β cell proliferation in vitro [7, 8]. We, therefore, investigated whether the genetic deletion of Smad3 also promotes β cell proliferation by culturing the Smad3KO and WT islet cells followed by BrdU labeling. Double immunofluorescence showed that islet cells lacking Smad3 exhibited a significantly higher level of BrdU-positive β cells (5.7%) compared with Smad3WT islet cells (2.4%) (Figure 7A and B). The augmented proliferation of Smad3KO β cells was also demonstrated by increased expression of PCNA as revealed by Western blot analysis (Figure 7C), confirming the key regulatory role of Smad3 in β cell proliferation.
5. RNA-seq identifies E2F3 as a Smad3 target gene that regulates β cell proliferation
To uncover the downstream mechanism of Smad3 in regulating β cell proliferation, we performed RNA-seq in islets isolated from db/m or db/db mice with or without deletion of Smad3 gene including Smad3WT-db/db, Smad3KO-db/db, Smad3WT-db/m, and Smad3KO-db/m mice as previously described [9]. Pathway enrichment analysis was conducted among the differentially expressed genes (DEGs) between Smad3WT and Smad3KO islets in db/m or db/db background. In the genetic background of db/db, Smad3KO resulted in substantial changes in cell cycle-related genes or pathways as revealed by GO and KEGG analysis (Figure 8A and Figure S3). Given the individual genes, RNA-seq analysis revealed an increase in the expression of genes involving the cyclin, cyclin-dependent kinases (CDKs), and cell division cycle (CDC) protein family in Smad3KO-db/db islets compared with Smad3WT-db/db islets, which was further validated in cultured Smad3KO islet cells by RT-PCR (Figure S4 A and C). In contrast, in the genetic background of db/m, Smad3KO caused no remarkable influence on cell cycle-related genes or pathways (Figure S3B and C). It is reported that TGF-β/Smad3 signaling negatively regulates the cell cycle by inducing the cyclin-dependent kinase inhibitor genes (CDKIs) [7, 8, 12]. Surprisingly, none of these CDKI genes’ expression was suppressed in Smad3KO-db/db islets in vivo and cultured Smad3KO islet cells as revealed by RNA-seq and RT-PCR, respectively (Figure S4B and C). In contrast, the expression levels of several CDKI genes like p16, p18, and p57 were even upregulated in cultured Smad3KO islet cells (Figure S4C). This implies that an alternative mechanism exists in Smad3-regulated β cell proliferation.
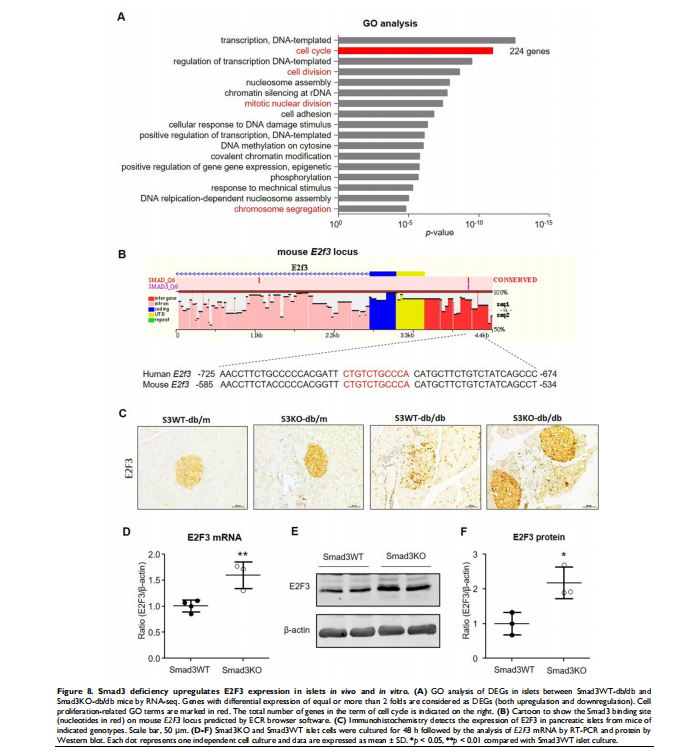
We thus analyzed the Smad3 binding potential for the promoter sequences of the 224 DEGs in the GO term of the cell cycle with the ECR browser software (Figure 8A). Among them, one gene named E2F3 gained our attention as it’s a key and terminal executor regulating G1/S entry of the cell cycle [13, 14]. We found a potential Smad3 binding site in the promoter of the mouse E2F3 locus which is also conserved in humans (Figure 8B).

Cistanche tubulosa
To investigate whether E2F3 is essential for Smad3-regulated β cell proliferation, we examined the association between E2F3 and Smad3 in islets. Consistent with the RNA-seq, immunohistochemistry detected that the expression of E2F3 was largely reduced in the islets of Smad3WT-db/db mice but greatly increased in Smad3KO-db/db mice (Figure 8C). Furthermore, increased E2F3 expression was also observed in cultured Smad3KO islet cells at both RNA and protein levels (Figure 8 D-F). In contrast, adenovirus-mediated silence of E2F3 abrogated the proliferative activity of β cells (insulin+ BrdU+) in Smad3KO islet cells (Figure 9), revealing an essential role for E2F3 in Smad3-regulated β cell proliferation.

To further confirm the predicted binding of Smad3 in the E2F3 promoter, we performed chromatin immunoprecipitation (ChIP) in isolated mouse islets (Figure 10A). The specificity of the Smad3 antibody used for ChIP was initially validated by immunoprecipitation in mouse islets (Figure 10B). ChIP PCR confirmed the binding of Smad3 to the E2F3 promoter as the anti-Smad3 antibody, but no non-specific IgG isotype successfully precipitated the chromatin fragment corresponding to the E2F3 promoter (Figure 10C). Quantitative RT-PCR revealed a 6-fold enrichment of the E2F3 promoter sequence in the ChIP assay with the Smad3 antibody compared with the IgG isotype (Figure 10D), demonstrating that Smad3 can bind to the promoter region of the E2F3 genomic locus.

To validate the functional significance of Smad3 binding on the E2F3 promoter, we constructed a luciferase reporter vector driven by a mouse E2F3 promoter harboring the Smad3 binding site or its mutant form (Figure 10E). Dual-luciferase assay in HEK293T cells revealed a robust transcription of luciferase driven by the cloned E2F3 promoter, which was significantly inhibited by overexpressing Smad3. However, mutation of the Smad3 binding site abrogated the inhibitory effect of Smad3 (Figure 10F). These findings provided direct evidence for the suppressive role of Smad3 in E2F3 transcription by binding to its promoter.
In this study, we demonstrated that transplantation of Smad3-deficient islets produced a better therapeutic effect on diabetes and diabetic kidney injury in STZ-induced diabetic mice and db/db mice. In STZ-induced diabetes, transplantation of 100 Smad3KO but not 100 Smad3WT islets resulted in a 17-fold increase in β cell mass, which was associated with a significant increase in blood insulin levels and thus improved glucose intolerance and recovered euglycemia, resulting in protection against diabetic kidney injury.
Similar results were also found in T2DM in db/db mice in which transplantation with 250 Smad3KO islets but not the same dose of Smad3WT islets also increased a 5-fold β cell mass and produced a better glycemic control. These findings suggest that Smad3 deficiency is resistant to diabetic microenvironments and largely promotes the expansion of grafted islet β cells and insulin production to exhibit better glycemic control. Interestingly, we also found that treatment with 100 Smad3KO islets could produce an equal glycemic control as the use of 200 Smad3WT or KO islets in STZ-induced diabetes. This observation suggests that Smad3 deficiency may be able to largely reduce (at least 50%) the number of islets required for the successful establishment of insulin independence in islet cell replacement therapy for T1DM. These findings keep in line with the previous report that pretreatment of the islets with SB-431542, an inhibitor of TGFBR1, promotes the proliferation and function of β cells after transplantation [15]. However, as Smad2 also plays an essential role in the glucose-stimulated release of insulin in β cells [16], targeting Smad3 but not upstream TGFBR1 in islets (β cells) may be more favorable for cell replacement treatment of diabetes. Thus, Smad3-deficient islet transplantation may represent a novel therapy for both T1DM and T2DM clinically. In addition, treatment with Smad3KO islets could also protect against diabetic kidney injury at 16 weeks of T1DM, although this is not profound in T2DM due to the minimal diabetic kidney injury occurring in db/db mice at the age of week 12.
Mechanistically, we found that Smad3 functions by suppressing E2F3 to inhibit β cell proliferation. This is supported by the evidence that the E2F3 promoter contains a functional Smad3 binding site as confirmed by ChIP and luciferase reporter assay. Islet β cells lacking Smad3 showed enhanced proliferation in an E2F3-dependent mechanism as the knockdown of E2F3 abolished this pro-proliferative activity. E2F3 belongs to the E2F family and is a critical component of the Rb-E2F machinery governing G1/S cell cycle entry [13]. E2Fs are the transcriptional factors and final executors to regulate genes orchestrating cell cycle progress [14]. Overexpression of E2F3 has been proven to promote β cell proliferation in islets from rodents and humans [17]. The findings that Smad3 transcriptionally targets E2F3 to repress the cell cycle in β cells provide a novel regulatory mechanism for β cell proliferation. Thus, disruption of Smad3 in islet β cells promotes E2F3-dependent β cell proliferation as demonstrated by a marked increase in insulin-producing PCNA+ or BrdU+ β cells, suggesting that treatment with a subtherapeutic dose of Smad3KO islets is sufficient to expand β cell mass and inhibits both T1DM in STZ-induced mice and T2DM in db/db mice.

Herba Cistanche and Cistanche extract
It is reported that TGF-β signaling plays a critical role in pancreatic islet β cell development and function, arrests the cell cycle, and causes β cell apoptosis by inducing CDKIs [7, 8, 18]. To our surprise, both in vivo and in vitro experiments revealed that the transcription of CDKIs was not changed or even upregulated in Smad3KO islets (Figure S4). This discrepancy may be attributed to the different experimental conditions. The results in the current study were derived from Smad3KO mice (islets). However, the previous studies are based on the pharmacological inhibition of TGFBR1, which shall influence the entire TGF-β signaling [7, 8]. Furthermore, we used islet samples from young adult mice (age of 8-10 weeks), while islets from relatively old animals or cadaveric donors of aged people were used in previous studies [7, 8]. As islet β cells show age-associated accumulation of CDKIs [19], CDKIs in aged β cells may be more sensitive to TGF-β signaling inhibition.
There were several limitations in the present study. First, the efficacy of islet transplantation is investigated in the STZ-induced diabetic mouse model without autoimmune reaction. Therefore, the advantage of Smad3KO over Smad3WT islet therapy for diabetes should also be verified in an authentic T1DM model in the future. Second, the findings from this study are based on the whole-islet but not β cell-specific Smad3KO cell therapy. It’s unclear whether cell types other than β cells in the islet would execute a Smad3-dependent role to modulate β cell proliferation by intercellular crosstalk pathways. Thus, further studies with β cell-specific Smad3KO are warranted. Furthermore, findings from this study need to be verified in human islet samples to prove their clinical significance.
References
1 Shapiro AM, Pokrywczynska M, Ricordi C. Clinical pancreatic islet transplantation. Nat Rev Endocrinol. 2017; 13: 268-77.
2. Jacobson EF, Tzanakakis ES. Human pluripotent stem cell differentiation to functional pancreatic cells for diabetes therapies: Innovations, challenges, and future directions. J Biol Eng. 2017; 11: 21.
3. Toso C, Isse K, Demetris AJ, Dinyari P, Koh A, Imes S, et al. Histologic graft assessment after clinical islet transplantation. Transplantation. 2009; 88: 1286-93.
4. Lan HY. Diverse roles of TGF-beta/Smads in renal fibrosis and inflammation. Int J Biol Sci. 2011; 7: 1056-67.
5. Meng XM, Tang PM, Li J, Lan HY. TGF-beta/Smad signaling in renal fibrosis. Front Physiol. 2015; 6: 82.
6. El-Gohary Y, Tulachan S, Wiersch J, Guo P, Welsh C, Prasadan K, et al. A smad signaling network regulates islet cell proliferation. Diabetes. 2014; 63: 224-36.
7. Dhawan S, Dirice E, Kulkarni RN, Bhushan A. Inhibition of TGF-beta Signaling Promotes Human Pancreatic beta-Cell Replication. Diabetes. 2016; 65: 1208-18.
8. Wang P, Karakose E, Liu H, Swartz E, Ackeifi C, Zlatanic V, et al. Combined Inhibition of DYRK1A, SMAD, and Trithorax Pathways Synergizes to Induce Robust Replication in Adult Human Beta Cells. Cell Metab. 2019; 29: 638-52 e5.
9. Sheng J-Y, Wang L, Tang PM-K, Wang H-L, Li J-C, Xu B-H, et al. Smad3 deficiency promotes beta cell proliferation and function in db/db mice via restoring Pax6 expression. Theranostics. 2021; 11: 2845-59.
10. Zmuda EJ, Powell CA, Hai T. A method for murine islet isolation and subcapsular kidney transplantation. J Vis Exp. 2011.
11. Choi MY, Lim SJ, Kim MJ, Wee YM, Kwon H, Jung CH, et al. Islet isograft transplantation improves insulin sensitivity in a murine model of type 2 diabetes. Endocrine. 2021; 72: 660-71.
12. Vijayachandra K, Higgins W, Lee J, Glick A. Induction of p16ink4a and p19ARF by TGFbeta1 contributes to growth arrest and senescence response in mouse keratinocytes. Mol Carcinog. 2009; 48: 181-6.
13. Dyer MA, Cepko CL. Regulating proliferation during retinal development. Nat Rev Neurosci. 2001; 2: 333-42.
14. Dyson N. The regulation of E2F by pRB-family proteins. Genes Dev. 1998; 12: 2245-62.
15. Xiao X, Fischbach S, Song Z, Gaffar I, Zimmerman R, Wiersch J, et al. Transient Suppression of TGFbeta Receptor Signaling Facilitates Human Islet Transplantation. Endocrinology. 2016; 157: 1348-56.
16. Nomura M, Zhu HL, Wang L, Morinaga H, Takayanagi R, Teramoto N. SMAD2 disruption in mouse pancreatic beta cells leads to islet hyperplasia and impaired insulin secretion due to the attenuation of ATP-sensitive K+ channel activity. Diabetologia. 2014; 57: 157-66.
17. Rady B, Chen Y, Vaca P, Wang Q, Wang Y, Salmon P, et al. Overexpression of E2F3 promotes the proliferation of functional human beta cells without induction of apoptosis. Cell Cycle. 2013; 12: 2691-702.
18. Zhang Y, Alexander PB, Wang XF. TGF-beta Family Signaling in the Control of Cell Proliferation and Survival. Cold Spring Harb Perspect Biol. 2017; 9.
19. Wang P, Fiaschi-Taesch NM, Vasavada RC, Scott DK, Garcia-Ocana A, Stewart AF. Diabetes mellitus--advances and challenges in human beta-cell proliferation. Nat Rev Endocrinol. 2015; 11: 201-12.
20. Yang X, Letterio JJ, Lechleider RJ, Chen L, Hayman R, Gu H, et al. Targeted disruption of SMAD3 results in impaired mucosal immunity and diminished T-cell responsiveness to TGF-beta. EMBO J. 1999; 18: 1280-91.
21. Li DS, Yuan YH, Tu HJ, Liang QL, Dai LJ. A protocol for islet isolation from mouse pancreas. Nat Protoc. 2009; 4: 1649-52.
22. Szot GL, Koudria P, Bluestone JA. Transplantation of pancreatic islets into the kidney capsule of diabetic mice. J Vis Exp. 2007: 404.
23. Xu BH, Sheng J, You YK, Huang XR, Ma RCW, Wang Q, et al. Deletion of Smad3 prevents renal fibrosis and inflammation in type 2 diabetic nephropathy. Metabolism. 2020; 103: 154013.
Hong-Lian Wang1,2, Biao Wei2, Hui-Jun He2, Xiao-Ru Huang2,3, Jing-Yi Sheng2,4, Xiao-Cui Chen2,5, Li Wang1, Rui-Zhi Tan1, Jian-Chun Li1, Jian Liu1, Si-Jin Yang6, Ronald CW Ma2, and Hui-Yao Lan2,7
1 Research Center for Integrated Medicine and Department of Nephrology, Affiliated Traditional Chinese Medicine Hospital of Southwest Medical University, Luzhou, Sichuan, 646000, China.
2. Department of Medicine and Therapeutics, and Li Ka Shing Institute of Health Sciences, the Chinese University of Hong Kong, Hong Kong, 999077, China.
3. Guangdong-Hong Kong Joint Laboratory on Immunological and Genetic Kidney Diseases, Guangdong Provincial People’s Hospital, Guangdong Academy of Medical Sciences, Guangzhou, Guangdong, 510080, China.
4. State Key Laboratory of Bioelectronics, Jiangsu Key Laboratory for Biomaterials and Devices, School of Biological Sciences & Medical Engineering, Southeast University, Nanjing, China.
5. Key Laboratory of Prevention and Management of Chronic Kidney Disease of Zhanjiang City, Institute of Nephrology, Affiliated Hospital of Guangdong Medical University, Zhanjiang, Guangdong, 524001, China.
6. National Traditional Chinese Medicine Clinical Research Base, Affiliated Traditional Chinese Medicine Hospital of Southwest Medical University, Luzhou, Sichuan, 646000, China.
7. CUHK-Guangdong Provincial People’s Hospital Joint Research Laboratory on Immunological and Genetic Kidney Diseases, the Chinese University of Hong Kong, Hong Kong, 999077, China.






