Presence Of Depression And Anxiety With Distinct Patterns Of Pharmacological Treatments Before The Diagnosis Of Chronic Fatigue Syndrome: A Population-based Study in Taiwan Part 1
Sep 11, 2023
Abstract
Objective An increased prevalence of psychiatric comorbidities (including depression and anxiety disorder) has been observed among patients with chronic fatigue syndrome (CFS). However, few studies have examined the presence of depression and anxiety disorder before the diagnosis of CFS. This study aimed to clarify the preexisting comorbidities and treatments associated with patients with subsequent CFS diagnosis in a population-based cohort in Taiwan.
Cistanche can act as an anti-fatigue and stamina enhancer, and experimental studies have shown that the decoction of Cistanche tubulosa could effectively protect the liver hepatocytes and endothelial cells damaged in weight-bearing swimming mice, upregulate the expression of NOS3, and promote hepatic glycogen synthesis, thus exerting anti-fatigue efficacy. Phenylethanoid glycoside-rich Cistanche tubulosa extract could significantly reduce the serum creatine kinase, lactate dehydrogenase, and lactate levels, and increase the hemoglobin (HB) and glucose levels in ICR mice, and this could play an anti-fatigue role by decreasing the muscle damage and delaying the lactic acid enrichment for energy storage in mice. Compound Cistanche Tubulosa Tablets significantly prolonged the weight-bearing swimming time, increased the hepatic glycogen reserve, and decreased the serum urea level after exercise in mice, showing its anti-fatigue effect. The decoction of Cistanchis can improve endurance and accelerate the elimination of fatigue in exercising mice, and can also reduce the elevation of serum creatine kinase after load exercise and keep the ultrastructure of skeletal muscle of mice normal after exercise, which indicates that it has the effects of enhancing physical strength and anti-fatigue. Cistanchis also significantly prolonged the survival time of nitrite-poisoned mice and enhanced the tolerance against hypoxia and fatigue.

Click on mentally exhausted
【For more info:george.deng@wecistanche.com / WhatsApp:8613632399501】
Methods An analysis utilizing the National Health Insurance Research Database of Taiwan was conducted. Participants included were 6303 patients with CFS newly diagnosed between 2000 and 2010 and 6303 age-/sex-matched controls.
Results Compared with the control group, the CFS group had a higher prevalence of depression and anxiety disorder before the diagnosis of CFS. Sampled patients who took specific types of antidepressants, namely, selective serotonin reuptake inhibitors (adjusted odds ratio [aOR]=1.21, 95% confidence interval [CI] 1.04–1.39), serotonin antagonists and reuptake inhibitors (SARI; aOR=1.87, 95% CI 1.59–2.19), and tricyclic antidepressants (aOR=1.46, 95% CI 1.09–1.95), had an increased risk of CFS. CFS risk was also higher among participants taking benzodiazepine, muscle relaxants, and analgesic drugs. A sub-group analysis revealed that SARI use was related to an increased risk of CFS in the depression, anxiety disorder, male, and female groups. In the depression and anxiety disorder groups, analgesic drug use was associated with an increased CFS risk. Nonpharmacological treatment administration differed between men and women.
Conclusion This population-based retrospective cohort study revealed an increased risk of CFS among populations with preexisting depression and anxiety disorder, especially those taking SARI and analgesic drugs.
Keywords Chronic fatigue syndrome, Depression, Anxiety, Pharmacological treatment
Introduction
Patients with chronic fatigue syndrome (CFS) experience prolonged and disabling fatigue that cannot be explained by the existing state of medical knowledge. The prevalence of CFS differs widely depending on the diagnostic criteria, assessment method, and studied population, with its numbers ranging from 0.2% to 6.41% [1, 2]. A systematic review of 46 studies in 2020 estimated a CFS prevalence rate of 0.89% based on the commonly used Centers for Disease Control (CDC)-1994 definition of CFS [3, 4]. The aforementioned review also reported a sex difference, with female individuals having prevalence rates that were 1.5 to 2 times higher than those of male individuals.
In addition to fatigue, several accompanying symptoms were also frequently reported, specifically muscle pain, multiple joint pain, poor sleep, anxiety, and depression [5]. Musculoskeletal pain and insomnia were included in the CDC-1994 diagnostic criteria. Furthermore, mood and anxiety disorders were reported to be more prevalent in individuals with CFS relative to the general population [6]. CFS, which is also known as myalgic encephalomyelitis, was found to be potentially related to immune processes such as inflammation and infection [7]. Recent comparisons between the similarities of CFS and the potential COVID-19 long-term effects, including persistent fatigue, postexertional malaise, and pain, have underlined the critical role of the immune response in such conditions [8, 9]. On the other hand, systemic inflammation may be the mediator of CFS and its psychiatric comorbidities [10, 11]. Notably, the relationship between CFS and psychiatric comorbidities might be bidirectional as an abnormal immune response has also been demonstrated among patients with depression or anxiety disorder [12–14]. A study investigated patients with CFS and reported that the prevalence rates of concurrent anxiety and depression were 42.2% and 33.3%, respectively [15]. However, few large-scale epidemiological investigations of psychiatric comorbidities, especially those that focused on Asian populations, have been conducted.
With a focus on CFS, depression, and anxiety, this population-based retrospective cohort study investigated and analyzed the data from the Taiwan National Health Insurance Research Database (NHIRD). The treatments received by participants were also further analyzed by sex, age, and comorbidities.
Methods
Data resource
The dataset used in this study was derived from the National Health Insurance Research Database (NHIRD) in Taiwan. The National Health Insurance (NHI) program was launched on March 1, 1995, by Taiwan’s government. NHIRD has contained details concerning the demographic characteristics, dates of admission and discharge, prescriptions, surgical procedures, and diagnostic codes for approximately 99% of the entire population of the 23 million people residing in Taiwan. We used the 2000 Longitudinal Health Insurance Database (LHID) which was established by NHIRD. LHID 2000 was created and released to the public by NHIRD and includes all the original claim data and registration fees between 2000 and 2013 for one million individuals randomly sampled from the Registry for beneficiaries of the NHI program in 2000 in Taiwan. The diseases are defined according to the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM).

Sample participants
Cases of CFS were identified using two outpatient records or one admission record with a diagnosis of ICD-9-CM code 780.71. The date of the first diagnosed record of chronic fatigue syndrome was defined as the index date. For each chronic fatigue syndrome case, we used a frequency matching method and randomly selected one control without a chronic fatigue syndrome diagnosis. The dataset for the control population of 1 million samples was randomly selected from the LHI dataset, and individuals without a diagnosis of CFS were selected as the control population with the same sex, age, and index date. (Fig. 1.) We excluded the participants aged below 18 years or with missing information on sex. In the ICD-9-CM, the diagnosis of CFS is mainly based on the CDC-1994 diagnostic criteria noted in the ICD-9-CM Coordination and Maintenance Committee Meeting in 2011. The CDC-1994 diagnostic criteria specifically defined the patients receiving appropriate treatment for depression or anxiety, the diagnosis could still be made among patients with premorbid depression or anxiety [3].
Exposure assessment and comorbidities
For this study, we examined the exposure of pharmaceutical and non-pharmaceutical treatments. We accounted the exposure to pharmaceutical treatments of SSRI drugs (ATC code N06AB10, N06AB06, N06AB03, and N06AB08), SNRI drugs (ATC code N06AX21, and N06AX16), SARI drugs (ATC code N06AX05), norepinephrine and dopamine reuptake inhibitor (NDRI) drug (ATC code N06AX12), noradrenergic and specifc serotonergic antidepressants (NaSSA) drug (N06AX11), TCAs drugs (ATC code N06AA09 and N06CA01), BZD drugs (ATC code N03AE01, N05BA06, N05BA12, N05BA01, N05BA17, N05BA22, N05CD04, N05CD05, N05CD03, N05CD09, N05CD01, N05CD08), muscle relaxant (ATC code M03BX08), analgesic drugs which including acetaminophen, NSAIDs, pregabalin, gabapentin (ATC code M02AA, D11AX18, M01A, M01B, N03AX16, and N03AX12) and non-pharmaceutical of support psychotherapy, supportive group psychotherapy, deep psychotherapy, in-depth group psychotherapy, special psychotherapy, special group psychotherapy, behavioral therapy evaluation, behavioral therapy plan, supportive psychosocial consultation for family members/caregivers, stretching exercise, exercise therapy, breathing exercise, induced deep breathing exercise, rehabilitation exercise, multiple physical examinations of sleep, brainwave examination, sleep or wakefulness, and brainwave examination for sleep disorders. Study participants were categorized based on their pharmaceutical and non-pharmaceutical exposure status. Patients exposed to pharmaceutical or non-pharmaceutical were classified as users or non-users. We adjusted for the potentially confounding efects of other comorbidities, including depression (ICD-9-CM code 296.2, 296.3, 926.82, 300.4, 309.0, 309.1, and 311), anxiety disorder (ICD-9-CM code 300.0–300.3, 300.5–300.9, 309.2–309.4, 309.81, and 313.0), insomnia (ICD-9-CM code 307.41, 307.42, 780.50, and 780.52), suicide (ICD- 9-CM code E950-E959), Crohn’s disease (ICD-9-CM code 555), ulcerative colitis (ICD-9-CM code 555–556), renal disease (ICD-9-CM code 580–589), diabetes mellitus (ICD-9-CM code 250 and A181), obesity (ICD-9-CM code 278), gout (ICD-9-CM code 274), dyslipidemia (ICD-9-CM code 272), malignancy (ICD-9-CM code 140–208), HIV (ICD-9-CM code 042–044), rheumatoid arthritis (ICD-9-CM code 714), psoriasis (ICD-9-CM code 696.x), ankylosing spondylitis (ICD-9-CM code 720.0), lymphadenopathy (ICD-9-CM code 289.1–289.3, 686, and 785.6), Hashimoto's thyroiditis (ICD-9-CM code 245.2), Sjogren’s syndrome (ICD-9-CM code 710.2), irritable bowel syndrome (ICD-9-CM code 564.1), SLE (ICD-9-CM code 710.0), celiac disease (ICD-9-CM code 579.00, and herpes zoster (ICD-9-CM code 053) prior to the index date were evaluated as part of the analysis.

Statistical analysis
Descriptive statistics of CFS and controls are reported, including demographic characteristics, comorbid diseases, and exposure to potentially confounding treatments. The chi-square test was used to compare categorical variables, whereas the Student’s t-test was used to compare continuous variables between the chronic fatigue syndrome cohort and the comparison cohort as necessary. We used conditional logistic regression to assess the risk of CFS according to each category of pharmaceutical and non-pharmaceutical. The odds ratio (ORs) and 95% confidence intervals (CIs) for CFS were calculated as an unadjusted incidence rate, and then subsequently adjusted for covariates including age, sex, comorbidities, pharmaceutical and non-pharmaceutical. Bonferroni correction was performed for the correction of multiple comparisons. Analyses were performed using SAS software (version 9.4 for Windows; SAS Institute, Cary, NC, USA) for Windows 10. All statistical significance levels were set at p<0.05.
Results
This study included 6306 patients with CFS and 6306 patients without, all of whom were identified from the NHIRD between January 1, 2000, and December 31, 2013. The demographic and clinical characteristics of the study participants are presented in Table 1. Among the participants, 52.9 were female, and most were between 25 and 64 years old; the mean age of the participants was 50.6 years. Regarding the prevalence of comorbidities, participants with CFS had higher numbers of psychiatric disorders (depression, anxiety disorder, and insomnia), irritable bowel syndrome, inflammatory bowel diseases (Crohn’s disease and ulcerative colitis), autoimmune disorders (rheumatoid arthritis, and Sjogren’s syndrome), metabolic disorders (type 2 diabetes mellitus, gout, and dyslipidemia), and renal disease (all p<0.005).

Table 2 and Fig. 2 show the pharmacological and non-pharmacological treatment received before the diagnosis of CFS. Participants taking certain types of antidepressants, including SSRI, SARI, and TCA, had higher odds of CSF, with the adjusted odds ratio (ORs) of 1.21 (95% CI 1.04–1.39), 1.87 (95% CI1.59–2.19), and 1.46 (95% CI 1.09–1.95). Other drugs with increased aORs of CFS included BZDs (1.60, 95% CI 1.46–1.76), muscle relaxants (1.74, 95% CI 1.39–2.19), and analgesics (3.56, 95% CI 3.16–4). As for the non-pharmacological treatments and examinations received by the participants, undergoing brainwave examination had a significantly increased odds ratio (1.6, 95% CI 1.44–1.77) of CFS but an insignificant aOR after being adjusted with demographic data and comorbidities.
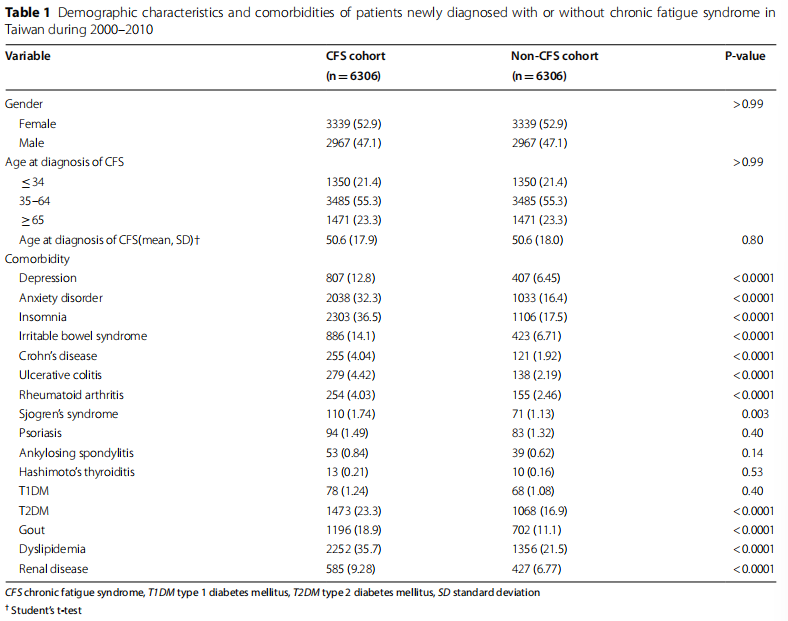



Table 3 and Fig. 3 present the treatment received before the diagnosis of chronic fatigue syndrome with comorbidity sub-classification by having depression or anxiety disorder. The aORs of SARI usages and analgesic drug usages increased in both groups with depression and anxiety disorders. Among the participants with depression who received supportive individual psychotherapy, the aORs of risk of CFS was 1.85 (95% CI 1.02– 3.35). As for the participants with anxiety disorder, the aORs of risk of CFS was 1.55 (95% CI 1.03–2.31) in those who also take muscle relaxants.
As presented in Table 4 and Fig. 4, the analysis with sub-classification by age also demonstrates different patterns of medications used across different ages. BZD, muscle relaxants, and analgesic drug usage were indicated on increased aORs of risks of CFS in all the age groups. In contrast, the usages of SSRI, SARI, and TCA among participants aged from 35 to 64 years old had as of 1.24 (95% CI 1.04–1.47), 1.90 (95% CI 1.56– 2.31), and 1.80 (95% CI 1.26–2.58), respectively. Among participants aged over 65 years old, the use of serotonin and norepinephrine reuptake inhibitor (SNRI) and SARI, with aORs being 2.15 (95% CI 1.22–3.81) and 1.93 (95% CI 1.46–2.57), respectively.

In Table 5 and Fig. 5, we present the therapeutic options received by the patients with CFS and controls with sex-specific sub-classifications. In female patients, the adjusted odds ratio of the risk of CFS were 1.22 (95% CI 1.01–1.48), 1.69 (95% CI 1.37–2.08), 1.72 (95% CI 1.17– 2.53), 1.66 (95% CI 1.45–1.9), 1.56 (95% CI 1.16–2.1), 3.23 (95% CI 2.72–3.84), 1.36 (95% CI 1.08–1.72), 1.38 (95% CI 1.09–1.76), and 1.26 (95% CI 1.02–1.54), folds with SSRI use, SARI use, TCA use, BZD use, muscle relaxant use, analgesic drug use, supportive individual psychotherapy, re-educative psychotherapy, and stretching exercise. In male patients, the adjusted odds ratio risk of CFS was 1.92 (95% CI 1.19–3.08), 2.20 (95% CI 1.70–2.84), 1.55 (95% CI 1.36–1.76), 2.07 (95% CI 1.45–2.97), and 3.90 (95% CI 3.31–4.59) folds with SNRI use, SARI use, BZD use, muscle relaxant use, and analgesic drug use.



【For more info:george.deng@wecistanche.com / WhatsApp:8613632399501】






