Renal And Extra Renal Manifestations in Adult Zebra fish Model Of Cystinosis
Mar 16, 2022
for more information:ali.ma@wecistanche.com
Sante Princiero Berlingerio 1,† , Junling He 2,†, Lies De Groef 3 , Harold Taeter 4, Tomas Norton 4 , Pieter Baatsen 5 , Sara Cairoli 6 , Bianca Goffredo 6, Peter de Witte 7 , Lambertus van den Heuvel 1,8, Hans J. Baelde 2 and Elena Levtchenko 1,*
Part I: Renal and Extra Renal Manifestations in Adult Zebra fish Model of Cystinosis
1 Laboratory of Pediatric Nephrology, KU Leuven, 3000 Leuven, Belgium; santeprinciero.berlingerio@kuleuven.be (S.P.B.); Bert.vandenHeuvel@radboudumc.nl (L.v.d.H.)
2 Department of Pathology, Leiden University Medical Center, 2300 RC Leiden, The Netherlands; J.He@lumc.nl (J.H.); j.j.baelde@lumc.nl (H.J.B.)
3 Neural Circuit Development and Regeneration Research Group, KU Leuven, 3000 Leuven, Belgium; lies.degroef@kuleuven.be
4 Group of M3-BIORES, Division of Animal and Human Health Engineering, KU Leuven, 3000 Leuven, Belgium; harold.taeter@kuleuven.be (H.T.); tomas.norton@kuleuven.be (T.N.)
5 Molecular Neurobiology, VIB-KU Leuven, 3000 Leuven, Belgium; pieter.baatsen@kuleuven.be
6 Department of Pediatric Medicine, Laboratory of Metabolic Biochemistry Unit, Bambino Gesù Children’s Hospital, IRCCS, 00146 Rome, Italy; sara.cairoli@opbg.net (S.C.); biancamaria.goffredo@opbg.net (B.G.)
7 Laboratory for Molecular Biodiscovery, KU Leuven, 3000 Leuven, Belgium; peter.dewitte@kuleuven.be
8 Department of Pediatric Nephrology, Radboud University Medical Center, 6525 GA Nijmegen, The Netherlands
* Correspondence: elena.levtchenko@uzleuven.be; Tel.: +32-16-34-38-22
† These authors contributed equally to this work.
Abstract: Cystinosis is a rare, incurable, autosomal recessive disease caused by mutations in the CTNS gene. This gene encodes the lysosomal cystine transporter cystinosis, leading to lysosomal cystine accumulation in all cells of the body, with kidneys being the first affected organs. The current treatment with cysteamine decreases cystine accumulation but does not reverse the proximal tubular dysfunction, glomerular injury, or loss of renal function. In our previous study, we have developed a zebrafish model of cystinosis through a nonsense mutation in the CTNS gene and have shown that zebrafish larvae recapitulate the kidney phenotype described in humans. In the current study, we characterized the adult cystinosis zebrafish model and evaluated the long-term effects of the disease on kidney and extrarenal organs through biochemical, histological, fertility, and locomotor activity studies. We found that the adult cystinosis zebra fish presents cystine accumulation in various organs, altered kidney morphology, impaired skin pigmentation, decreased fertility, altered locomotor activity, and ocular anomalies. Overall, our data indicate that the adult cystinosis zebrafish model reproduces several human phenotypes of cystinosis and may be useful for studying pathophysiology and long-term effects of novel therapies.
Keywords: kidney disease; cystinosis; zebrafish model; renal and extrarenal manifestation; adult phenotypic features
1. Introduction
Cystinosis is an autosomal recessive storage disorder caused by mutations in the CTNS gene which encodes the lysosomal cystine proton co-transporter cystinosis, carrying cystine from the lysosomal lumen to the cytosol. Several organs are involved in the disease, such as muscles, testis, eyes, and brain, and among all, kidneys are the first and most severely affected organs [1]. The most frequent and severe clinical variant is the infantile nephropathic cystinosis, in which the first clinical manifestation is the renal Fanconi syndrome, characterized by an impaired proximal tubular epithelial cell
(PTE) reabsorption resulting in the urinary loss of amino acids, glucose, low molecular to intermediate molecular weight proteins, and other metabolites. The PTEC damage is followed by progressive glomerular dysfunction and, when left untreated, leads to endstage kidney disease [1]. The current therapeutic approach consists of cysteamine, which is a cystine-depleting amino thiol that breaks the disulfide bridge of cysteine, resulting in the formation of amino acid cysteine and a cysteamine-cysteine mixed disulfide. This latter moleculecanexitlysosomesviatheaminoacidtransporterPQLC2[2]. However, treatment with cysteamine, although decreasing lysosomal cystine accumulation, only delays the progression of the disease [3–5]. Therefore, further understanding of the pathogenesis of cystinosis and developing new the rapeuticoption are still on the research agenda. Several animal models have been developed in order to recapitulate the cystinosis phenotype [6,7]. One of the most used models is the Ctns−/− C57BL/6 mouse, which develops cystine accumulation and mild proximal tubulopathy. However, it does not show glomerular damage and the phenotype has been found dependent on the genetic background [8].
To date, zebrafish models have become an attractive tool for investigating human diseases due to several advantages such as their rapid development, high fecundity, lower maintenance cost, and the availability of easily applicable gene-editing technologies [9]. In a previous study, we presented a congenic zebrafish larvae model with a homozygous nonsense mutation in the exon 8 of the ctns−/− gene, resulting in a functional loss of cystinosis [10], leading to cystine accumulation in the entire body, increased larvae deformity, delayed development, and kidney damage. However, long-term disease consequences in adult zebrafish have not been studied so far. In the current study, we aimed to characterize the renal and extrarenal manifestations in the adult zebrafish model of cystinosis.

Click to Organic Cistanche for renal Disease
2. Results
2.1. Cystine Accumulates in Ctns−/− Zebrafish
Cystinosis is considered a multisystemic disorder resulting in cystine accumulation in all cells of the body. In our previous work, we found that cystinosis (ctns−/−) zebrafish display cystine accumulation starting from the larval stage of development and in several organs up to the age of 8 months [9]. In order to evaluate if our model maintains cystine accumulation at the later stages of the disease, we measured cystine levels in the whole body, kidney, testis, eyes, muscle, and brain in 18-month-old ctns−/− zebrafish compared with age-matched wild-type male zebrafish. We found a 54-fold increase in cystine content in the whole body (Figure 1A) and a 146-fold increase in the kidney (Figure 1B) of ctns−/− zebrafish compared with wild-type zebrafish. Moreover, we observed extensive cystine accumulation in the testis, eyes, muscle, and brain (Figure 1C–F) in ctns−/− zebrafish.

2.2. Renal Manifestation
2.2.1. Ctns−/− Zebrafish Present PTEC Damage
Since the kidney is the most severely affected organ, we examined the histological characteristics of the kidney using Hematoxylin and Eosin(H&E)and Periodic Acid-Shiff(PAS) staining. We noted the presence of cloudy swelling, hyaline-like eosinophilic droplets, and cytoplasmic vacuoles in the PTEC of ctns−/− zebrafish (Figure 2B, D) but not in wild-type zebrafish (Figure 2A, C). Of note, no distal tubular damage was found. Cystine crystals were previously observed in PTEC as cytoplasmic vacuoles with rectangular or polymorphous shapes [11]. Therefore, we investigated if these structures were also present in tubules of zebrafish. We found numerous cytoplasmic vacuoles in the PTEC of ctns−/− zebrafish through toluidine blue staining (Figure 3B, D, indicated with asterisks). In addition, we found vacuolar space with rectangular or polymorphous shapes (Figure 3B, D, indicated with arrowheads). We confirmed this finding by transmission electron microscopy (TEM) (Figure 3F). These structures were not present in the PTEC of wild-type zebrafish (Figure 3A, C, E). Interestingly, in addition to the presence of polymorphous-shaped vacuoles, we observed a partial loss of the brush border in PTEC ultrastructure (Figure 3F).
Figure 1. Cystine content in homogenates of wild-type and ctns−/− zebrafish. (A–F) Cystine content in whole-body (A), kidney (B), testis (C), eyes (D), muscle (E), and brain (F) in wild-type and ctns−/− 18-month-old male zebrafish. Cystine concentration is expressed as nmol/mg protein. Each dot represents one zebrafish, for a total of n = 3 wild-type and n = 3 cents−/− 18-month-old male zebrafish; unpaired t-test with Welch’s correction, two-tailed: * p < 0.05; ** p <0.01; ns: not significant.
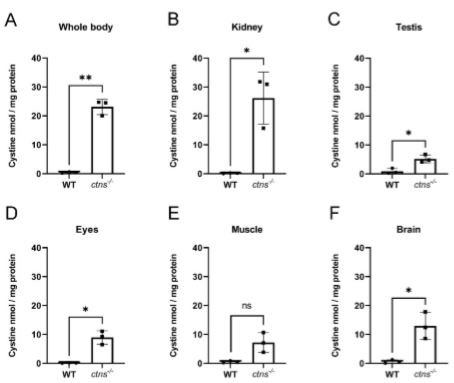
Figure 2. Kidney histology in wild-type and ctns−/− zebrafish. (A, B) Representative images of renal tubules of wild-type (A) and ctns−/− (B) 18-month-old zebrafish. Details of proximal tubules with hyaline-like eosinophilic droplets (black arrowheads) and cytoplasmic vacuoles (*). H&E staining. The scale bars represent 50 µm. (C,D) Representative images of the PAS staining of renal tubules of wild-type (C) and ctns−/− (D) 18-month-old zebrafish. Details of proximal tubules with hyaline-like droplets (black arrowheads) and cytoplasmic vacuoles (*). PAS staining. The scale bars represent 50 µm. PT: proximal tubule; DT: distal tubule.

Figure 3. Kidney cystine crystals in wild-type and ctns−/− zebrafish PTECs. (A,B) Representative toluidine blue-stained images of renal proximal tubules in wild-type (A) and ctns−/− (B) 18-month old zebrafish. The scale bars represent 50 µm. (C,D) Figures 3C, D show a higher magnification of Figure 3A,B. Details of renal proximal tubules with cytoplasmic vacuoles ((D); *), rectangular and polymorphous vacuolar spaces ((D); black arrowheads). (E, F) Representative TEM images of the renal proximal tubule of wild-type (E) and ctns−/− 18-month-old zebrafish (F). (F) Partial loss of proximaltubulebrushborders(PB)andaccumulationoftherectangularandpolymorphousvacuoles (*) were observed in the renal proximal tubule of ctns−/− zebrafish. The high-magnification view of the rectangle in the bottom right corner shows the polymorphous vacuoles and a large vacuole with straight membrane border segments (a straight line). The scale bars represent 5 µm.

2.2.2. Ctns−/− Zebrafish Show Glomerular Hypertrophy
Since glomerular damage usually follows PTEC lesions, we evaluated the glomerular histologyofthectns−/− zebrafish,andweobservedthatctns−/− malezebrafishshowedsignificant enlargement of Bowman’s capsule (Figure 4B, E) and glomerular tuft (Figure 4B, F), but not Bowman’s space (Figure 4B, G) when compared with wild-type male zebrafish (Figure 4A, E–G). Thesamewasconfirmedfortheglomerulartuftinfemalezebrafishcarrying the ctns−/− mutation (Figure 4F), and a similar trend was observed upon measurements oftheBowman’sspaceandtheBowman’scapsule(Figure4E, G). Noproliferationofmesangial cells or glomerulosclerosis were observed. On the TEM images, we did not observe alterations on the glomerular basement membrane, podocyte foot process effacement, or abnormally fenestrated endothelial cells in the glomeruli of ctns−/− zebrafish (Figure 4D) compared with wild-type zebrafish (Figure 4C).

Figure 4. Glomerular morphology in wild-type and in ctns−/− zebrafish. (A, B) Representative images of the glomerulus of wild-type (A) and ctns−/− (B) 18-month-old zebrafish. PAS staining. The scale bars represent 50 µm. (C,D) Representative TEM images of the glomerulus of wild-type ( ctns−/− (D) zebrafish. The scale bars represent 1 µm. (E–G) The quantification of the surface areas of Bowman’s capsule (E), glomerular tuft (F), and Bowman’s space (G) of wild-type and zebrafish in both genders.

2.2.3. Ctns−/− Zebrafish Display Increased Apoptosis at The PTEC Level
Next, we investigated whether cystinosis triggered cell death in our zebrafish model [10,12]. We performed cleaved caspase-3 immunostaining in the renal tissues of wild-type and ctns−/− zebrafish and we observed a significant increase of cleaved caspase3 protein levels in PTEC of ctns−/− zebrafish (Figure 5B, D, G) compared with wild-type zebrafish (Figure 5A, C, G) in both genders. The expression of cleaved caspase-3 was mainly present in cells showing the polymorphous cytoplasmic vacuoles (Figure 5B, D). No expression of cleaved caspase-3 was present in glomeruli, both in wild-type and ctns−/− zebrafish (Figure 5E, F).

Figure 5. Increased apoptosis in PTECs with vacuoles in ctns−/− zebrafish. (A, B) Representative images of the cleaved caspase-3 immunostaining of tubules of wild-type (A) and ctns− old zebrafish. The scale bars represent 50 µm. (C,D) The bottom panels show a higher magnification of the boxed areas in the upper panels. Details of PTECs with cytoplasmic vacuoles (*). The scale bars represent 50 µm. (E, F) Representative images of the cleaved caspase-3 immunostaining of glomerulus of wild-type (E) and ctns−/− (F) 18-month-old zebrafish. The scale bars represent 50 µm. (G) The quantification of the tubular caspase-3 staining positive area in wild-type and zebrafish in both genders. Each dot represents one zebrafish, for a total of n = 3 wild-type and ctns−/− 18-month-old zebrafish. Two-way ANOVA with Fisher’s least significant difference (PLSD) test: ** p < 0.01; *** p < 0.001.

2.3. Extra Renal Manifestations
2.3.1. Ctns−/− Zebrafish Show a Characteristic Hypopigmented Spotted Skin Pattern
Cystinosis patients frequently show premature skin aging, hypopigmentation, and blond hair [1,13]. Interestingly, we observed a hypopigmented spotted pattern of the skin of ctns−/− zebrafish, while wild-type fish presented the typical black striped pattern. The phenotype is present in both genders during their entire life (Figure 6A, B). In order to investigate the difference in pigmentation, we performed PAS staining on the zebrafish skin and we found that ctns−/− zebrafish display altered distribution of melanin layer in the epidermis, which caused the light spotted pattern (Figure 6C, D).
Figure 6. Hypopigmented spotted skin pattern in ctns−/− zebrafish. (A, B) Representative pictures of wild-type (A) and ctns−/− (B) 18-month-old zebrafish. The scale bars represent 0.5cm. The black arrowheads indicate the different striped skin patterns. (C,D) PAS-stained pictures of the epidermis of wild-type (C) and ctns−/− (D) 18-month-old zebrafish. The black arrowheads highlight the melanin layer. The interruption in the melanin layer is present in ctns−/− (D) while it is absent in wild-type zebrafish (C). The scale bars represent 50 µ

2.3.2. Ctns−/− Zebrafish Show Spermatogenic Cysts Enriched in Spermatozoa
Cystinosis is known to affect testicular functions, resulting in azoospermia and infertility in male patients [14–16]. The origin of the azoospermia is still unclear, although spermatogenesis has shown to be intact at the testicular level. In contrast with male patients, females with cystinosis are fertile [17]. In our zebrafish model, both female and male ctns−/− zebrafish are fertile. In order to investigate whether ctns−/− mutation affects the anatomy of the testis, we performed PAS staining and we found increased accumulation of spermatozoa in the spermatogenic cysts in ctns−/− zebrafish
Figure 7. Spermatogenic cysts enriched in spermatozoa in ctns−/− zebrafish. (A, B) Representative H&E stained pictures of testis of wild-type (A) and ctns−/− (B) 18-month-old zebrafish. Dark blue and rounded staining represent the spermatozoa accumulated in the spermatogenic cysts (indicated with the asterisks). The scale bars represent 200 µm.

2.3.3. Ctns−/− Female Zebrafish Show Decreased Egg Production
In order to investigate the functionality of the reproductive system, we measured egg production and the percentage of fertilized eggs in both wild-type and ctns−/− zebrafish. We found that ctns−/−females show a decreased egg production (Figure 8A) and decreased percentage of fertilized eggs (Figure 8B) compared with the wild-type females.
Figure 8. Egg production and percentage of fertilized eggs in wild-type and ctns−/− zebrafish. (A) Egg production was measured as a total number of eggs in wild type and ctns−/− zebrafish 18-month-old zebrafish. The egg production rate is 1.941 high in wild-type zebrafish compared with ctns−/− 18month-old zebrafish (rate ratio = 1.941); 95% CI (lower bound = 1.165, higher bound = 3.234); Poisson regression analysis; p = 0.011. (B) The percentage of fertilized eggs was measured on the total number of eggs. The odds of the fertilized eggs are 3.504 high in wild-type zebrafish compared with ctns−/− 18-months-old zebrafish (odds ratio = 3.504); 95% CI (lower bound = 2.005, higher bound = 6.124); Logistic regression analysis; p < 0.001; n/a: not applicable.

2.3.4. Ctns−/− Zebrafish Display Decreased Locomotor Activity
Since muscle wasting and weakness have been diagnosed in cystinosis patients, generally from the second decade of life [1,18], we evaluated the locomotor activity in our zebrafish model. We discovered that ctns−/− zebrafish from both genders showed decreased locomotor activity compared with wild-type zebrafish, suggesting an impaired muscle function (Figure 9). However, histological analysis of skeletal muscle tissue did not reveal apparent abnormalities in ctns−/− zebrafish.





