Renal And Extra Renal Manifestations in Adult Zebra Fish Model Of Cystinosis
Mar 16, 2022
for more information:ali.ma@wecistanche.com
Part: II Renal and Extra Renal Manifestations in Adult Zebra fish Model of Cystinosis
2.3.5. Ctns−/− Zebrafish Present Increased Thickness of THE Cornea
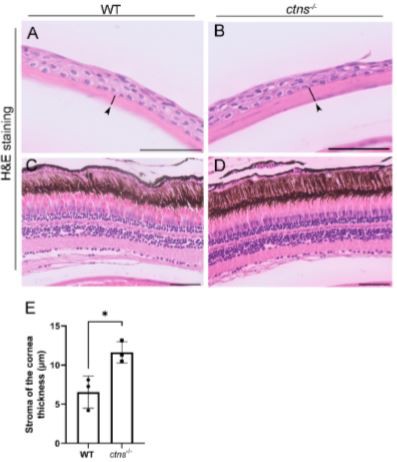
Cystinosis affects the eyes resulting in cystine accumulation in several tissues, such as the iris, conjunctiva, and retinal epithelium, however, the cornea is the most affected part due to cystine accumulation [19] and presents increased thickness [20]. Using H&E staining, we noted that ctns−/−zebrafish showed increased thickness of the stromal layer of the cornea compared with wild-type(Figure10A, B, E)zebrafish. However, we did not reveal apparent abnormalities in the retinal epithelium of ctns−/−zebrafish (Figure 10C, D).
Figure 9. Average swimming speed in wild-type and ctns−/−zebrafish. The average speed is decreased in ctns−/−zebrafish compared with wild-type in both genders. The average swimming speed is measured in pixel/second. Each dot represents one zebrafish, for a total of n = 6 wild-type and n = 6 ctns−/−18-month-old zebrafish. Two-way ANOVA with Fisher’s least significant difference (PLSD) test: ** p < 0.01.

Figure 10. Eye histology in wild-type and ctns−/− zebrafish. (A, B) Representative images of the cornea of wild-type (A) and ctns−/− (B) 18-month-old zebrafish. The stromal layer of the cornea (black arrowheads and black lines). H&E staining. The scale bars represent 50 µm. (C,D) Representative images of the retina of wild-type (A) and ctns−/− (B) zebrafish. H&E staining. The scale bars represent 50 µm. (E) Relative quantification of the thickness of the stromal layer of the cornea. Each dot represents one fication of the thickness of the stromal layer of the cornea. Unpaired t-test with Welch’s correction, two-tailed: * p < 0.05.
2.3.6. Additional Ctns−/− Zebrafish Phenotypes
In addition to the phenotypes mentioned above, we performed histological analysis ofthebrainandovarywithoutfindingdifferencesbetweenctns−/−and wild-typezebrafish. Moreover, we evaluated body length and weight of ctns−/−and wild-type zebrafish at 18monthsoldandwefoundanincreasedbodyweightinctns−/−zebrafishwhilewefound an increased body length in ctns−/−zebrafish of both genders compared with wild-type (Supplementary Figure S1).
3. Discussion
During the last decades, cystinosis has transformed from a fatal disease of childhood into a treatable metabolic disorder with which patients can survive into adulthood and reach advanced age [21,22]. Longer survival of the patients revealed novel disease phenotypes and raised new questions regarding the pathogenesis and long-term effects of therapies. In this regard, studying adult animal models of cystinosis becomes more and more relevant.
In the current study, we investigated the renal and extrarenal manifestations of cystinosisinanadultzebrafishmodelof18monthsold, which corresponds to the human age of 40–50 years [23,24]. First, we validated that the adult ctns−/− zebrafish model presents the whole-body and various organs cystine accumulation. In the kidney, at the histological level, we observed signs of PTEC damage reflected by the presence of numerous hyaline-like eosinophilic droplets, cytoplasmic vacuoles, and partial loss of brush borders. Furthermore, the ctns−/−male zebrafish developed glomerular hypertrophy. Lastly, we showed that cleaved-caspase 3 expression was increased in the renal PTEC of ctns−/−zebrafish, indicating that apoptosis is involved in the pathogenesis of nephropathic cystinosis in zebrafish. Since cystinosis patients present also extrarenal manifestations, we investigated the effect of ctns−/− mutation in other organs in our zebrafish model. Interestingly, we found that ctns−/−mutation caused an impairment in the skin anatomy and affect fertility, locomotor activity, and eyes.
Lysosomes are the sites of intracellular digestion and are considered to be the vital coordinators of cell metabolism [25]. Being a lysosomal storage disease, the key feature of cystinosis is lysosomal cystine accumulation [26,27]. Interestingly, we found a 54-fold increase of cystine in the whole-bodyofthe18-monthscystinosiszebrafishwhencompared with wild-type, while, in our previous study [10], cystinosis zebrafish larvae at 6 days post fertilization presented a 13-fold increase. Overall, these data suggest that cystine accumulates during the lifetime in cystinosis zebrafish. Liquid chromatography and mass spectrometryanalysishaveconfirmedthat, among all organs, kidney sare the preferred site for cystine accumulation. Lysosomal swelling and hyaline-like eosinophilic droplets inside the lysosomes have been reported in cystine-loaded PTEC [28,29] and, in human biopsies, cystine crystals appear as polymorphous shape spaces within the interstitial macrophages and the PTEC cytoplasm [27]. In human tissue, cystine accumulation and crystallization is a cumulative process occurring during the lifetime. Inline, cystine crystals were not detected in the ctns−/−zebrafish at the larval stage [10] while, in our pilot experiments, we found that the polymorphous shape of cystine crystals appear at the age of 3 and 6 months and are clearly visible at the age of 18-months, suggesting a progressive worsening of the effects of cystinosis in the kidney.

Click to Cistanche effects for kidney disease
To assess the histological changes of the glomeruli, we measured the surface areas of Bowman’s capsule, the glomerular tuft, and Bowman’s space of ctns−/−zebrafish on PAS-stained sections. We found that ctns−/−zebrafish developed glomerular hypertrophy, suggestingahyperfiltration. Themildglomerulardamageobservedinthectns−/−zebrafish might be attributed to the regenerative ability of the zebrafish kidney, being able to add new nephrons, as well as repairing existing nephrons [30].
In addition to the histological damage, cystine accumulation has been shown to activateproteinkinaseCtriggeringapoptosisinPTEC[12]. Thus, we evaluated the cleaved caspase-3 expression in PTEC and we found that the ctns−/−zebrafish presented increased caspase-3 and nuclear fragmentation. The presence of cleaved caspase-3 was mainly found in cells that accumulated the cytoplasmic vacuoles, suggesting that apoptosis takes place in the injured PTEC. One additional mechanism that can explain the increased apoptosis might be the presence of reactive oxygen species due to reduced glutathione associated with cystine accumulation [31]. However, further studies are necessary to evaluate if this mechanism occurs also in ctns−/−zebrafish.
Although kidneys are the first and most severely affected organs, cystinosis is a multi-systemic disease in which various organs, such as skin, gonads, eyes and muscles are involved. From the literature, it is known that cystinosis patients frequently display premature skin aging, blond hair, and fair skin color [13]. The latter phenotype is due to theinvolvementofcystinosintransporterintheregulationofmelaninsynthesis[32]andin the maintenance of darker pigmentation through the newly discovered cysteine transporter MFSD12 [33]. Excitingly, our adult ctns−/−zebrafish model presented a hypopigmented and spotted skin pattern and showed an impaired melanin distribution which was not observed at the larval stage [10].
In addition, cystinosis causes infertility in males, while females are known to be fertile [14]. Recent data suggest that infertility could be due to progressive testicular degeneration, leading to altered semen quality, or due to an obstruction in an undefined part of the male excretory system. The obstructed structures might affect the quality of the semen, especially in the regions where ducts have a very small diameter, for instance, the retetestis[16]. Inzebrafish, the sperm to genicprocessis are very similar to mammals; however, one of the main differences is that spermatogenesis occurs in the cysts [34]. In our study, we found increased spermatozoa in the spermatogenic cyst of ctns−/−zebrafish, however, we did not find signs of testicular degeneration. Therefore, the described phenotype could suggest an obstruction at testicular levels that leads to less release of spermatozoa in the environment, however further studies are necessary to confirm this hypothesis. At the functional level, the decreased egg numbers and decreased fertilized eggs seem to reflect an impairment in the fertility of ctns−/−zebrafish.
Another clinical manifestation of cystinosis, generally affecting patients starting from the second decade of life, is myopathy, characterized by muscle wasting and weakness [1]. Interestingly, we found that ctns−/−adult zebrafish present decreased locomotor activity compared with control,suggesting animpaired muscular function. Of note, this phenotype was not present in larvae [10], suggesting that it affects ctns−/−zebrafish during the later stage of the disease. Despite this, no visible histological differences were detected in the muscles. The decreased locomotor activity might be due to mitochondrial dysfunction which could not be detected by routine histology studies [35–37]. Hence, a comprehensive characterization of mitochondria in cystinosis zebrafish needs further evaluation.
Inadditiontotheabove-mentioned phenotypes, several studies have shown that cystinosis patients also display ocular symptoms such as photophobia and blepharospasm [1], with cornea being one of the most affected parts of the eye resulting in cystine crystal accumulation and increased thickness [20]. However, little is known about the causes of the increased thickness of the cornea. For instance, it was suggested that the described phenotype is due to a subclinical dysfunction of epithelial and/or endothelial cells leading tostromaledema[20]. In our study, we found that ctns−/−adultzebrafishdisplayincreased is the thickness of the stromal layer of the cornea, indicating that our model shows signs of cornea dysfunction in cystinosis.

Cistanche for kidney function
One of the main gaps to fill for a comprehensive understanding of cystinosis is the lack of an animal model that fully recapitulates the human disease. Several mouse models have been developed, showing cystine accumulation but failing to reproduce the complete renal phenotype [8,26]. Specifically, the C57BL/6 mouse presented cystine accumulation in several organs, which increases with age [8]. In addition, the C57BL/6 mouse model shows proximal tubular lesions starting from the age of 6 months. Similarly, in our model, proximal tubular lesions worsen over time. However, at the glomerular level, the C57BL/6 mouse model presented a normal phenotype, while our adult zebrafish model shows glomerular hypertrophy. In addition, the C57BL/6 mouse model fails to mimic the impaired fertility observed in humans, while the ctns−/−zebrafish model presents spermatogenic cysts enriched in spermatozoa and decreased eggs production. At the ocular level,bothmouse[38]andthectns−/−zebrafishmodelpresentsocularabnormalities. On the contrary, at the skin level, this mouse model does not show impaired melatonin production [8], which appears in the adult zebrafish model as a hypopigmented skin pattern. Lastly, at the behavioral level, our ctns−/− zebrafish model presents a decreased locomotor activity, which may be because by an impaired mitochondrial function or it might be caused by a secondary effect of the increased weight in cystinosis zebrafish. Overall, it seems that our ctns−/− zebrafish model better recapitulates the human disease. Recently, Shimizu et al. established a novel congenic Ctnsugl mutation in a rat strain that presented renal lesions and cystine crystals in the lysosomes of the kidney cortex [7]. However, a more detailed characterization of the model is still missing. Therefore, there is an urgent need to develop and characterize new functional models that recapitulate human disease.
In conclusion, we demonstrated that our adult ctns−/− zebrafish model reproduces several human phenotypes of cystine sandpit may be useful for studying the pathogenesis of the disease in adults and for testing long-term effects of novel drugs for correcting renal and extra-renal manifestations.
4. Materials and Methods
4.1. Zebrafish Maintenance and Breeding
Zebrafish was handled and maintained in compliance with the KU Leuven animal welfare regulations (Ethical approval nr.142/2019). For this study, we included ctns−/−zebrafish at 18 months old and the wild-type control zebrafish in both genders. The details of generating the ctns−/−zebrafish were described in our previous study [10].
4.2. Cystine Measurement
Cystinelevelsweremeasuredinzebrafish(wild-typemalefish,n=3andctns−/−male fish, n = 3) and the amount was expressed as nmol/mg proteins. First, zebrafish were sacrificed with immersion in tricaine methanesulfonate MS-222 (300 mg/L), afterward, tissue samples were sonicated in the presence of 200 µL of 5 mM N-ethylmaleimide (NEM, St. Louis, MO, USA, Sigma-Aldrich E3876) in Dulbecco’s phosphate-buffered saline (PBS), for a maximum of 200 mg of tissue for each lysate. Subsequently, 100 µL of 12% sulfosalicylic acid (SSA) were added to each homogenate, and samples were centrifuged at 12,000 g for 10 min, 4◦C. Supernatants containing cystine were stored at−80◦C until time of analysis, while pellets were dissolved overnight at 4◦C in 300 µL of 0.1 M NaOH and kept at−80◦C until protein is measured using Pierce BCA Protein Assay Reagent Kit. Subsequently, the amount(nmol)of cystine was normalized to the quantity (mg) of protein from each sample. Next,50 µLofthesupernatantcontainingcystinewasspikedwith50 µL of the internal standard solution (Cystine d6) and vortexed for 5 s; then the mixture was extracted with 200 µL of acetonitrile, vortexed for 30 s, and then centrifuged at 16,000 g for 9 min. Liquid chromatography and mass spectrometry analysis were performed by a UHPLCAgilent1290InfinityII6470(Agilent technologies, Santa Clara, CA, USA)equipped with an ESI-JET-STREAM source operating in the positive ion (ESI+) mode. The software used for controlling this equipment and analyzing data was MassHunter (Work station Agilent Technologies, Barcelona, Spain). The separation column used was InfinityLab Poroshell 120 HILIC 1.9 µm 100×2.1 mm(Agilent Technologies, Santa Clara, CA, USA).
4.3. Hematoxylin and Eosin and Periodic Acid-Shiff Staining
Zebrafish (wild-type female fish, n = 3; wild-type male fish, n = 3; ctns−/−female fish, n = 3; ctns−/−male fish, n = 3) were sacrificed with immersion in tricaine methanesulfonate MS-222 (300 mg/L), afterward, the whole zebrafish was fixed in 4% buffered paraformaldehyde (4% PFA) at 4 ◦C for 1 week. After being washed with PBS twice, the zebrafish were transferred to EDTA solution (100 mM, pH = 8) for the decalcification for another 1 week, followed by embedding into paraffin. Paraffin-embedded zebrafish tissuewascut(4-µmthickness)onaLeicamicrotome(LeicaBiosystems,Wetzlar,Germany). Sections were stained with H&E and PAS according to the standard protocols.
Forhistologicalstudiesofzebrafisheyes(wild-typefemalefish,n=3andctns−/−female fish, n = 3), eyes were enucleated and fixed in 4% PFA at room temperature for 4 h. The following embedding into optimal cutting temperature medium(Tissue-Tek® VIP®; Sakura Finetek, Japan), 10-µm cryosections were made(Cryostar NX70, Thermofisher Scientific, Tokyo, Japan). Sections were stained with H&E according to the standard protocols. Images were made with a Leica brightfield microscope(Leica DM6, Wetzlar, Germany).
4.4. Immunohistochemistry (Cleaved Caspase-3 Staining)
Sections with the fresh-cut zebrafish tissue were deparaffinized and rehydrated. Sections were subjected to heat-induced antigen retrieval using 10 mM citrate buffer (pH = 6). After blocking with 5% Normal Goat Serum (NGS) in dPBS, the sections were incubated with rabbit anti-cleaved caspase-3 (Cell Signaling, Danvers, MA, USA, 1:200 dilution) followed by an anti-rabbit-Envision, HRP-labelled secondary antibody(Dako Products, Agilent, Santa Clara, CA, USA). As a negative control, a normal rabbit serum was used. Diaminobenzidine (DAB, DAKO, Glostrup, Denmark) was used as the chromogen. Subsequently, sections were counterstained with H&E, dehydrated, and mounted.

Treatment of Kidney disease: Cistanche
4.5. Toluidine Blue Staining and TEM
Zebrafish renal tissues (wild-type male zebrafish, n = 3 and ctns−/−male zebrafish, n = 3) were harvested and fixed in the EM fixation buffer (1.5% glutaraldehyde/1% paraformaldehyde) for 24 h. Subsequently, the renal tissues were post-fixed with 2.5% glutaraldehyde/1.2% acrolein in fixation buffer (0.1 mol/L cacodylates, 0.1 mol/L sucrose, pH 7.4) and 1% osmium tetroxide, and embedded into upon resin. Semi-thin sections (0.5-µm thickness) were stained with toluidine blue. The ultrathin sections were stained with uranyl acetate. The images were collected using a JEM-1200 EX transmission electron microscopy (JEOL Ltd, Tokyo, Japan) with different magnifications.
4.6. Digital Image Analysis
Stained slides were digitized using a Philips Ultra-Fast Scanner 1.6 RA (Philips, Eindhoven, The Netherlands)
4.6.1. Glomerular Hypertrophy Analysis
To analyze glomerular hypertrophy in zebrafish, the surface areas (µm2) of Bowman’s capsule, Bowman’s space, and glomerular tuft were measured on PAS-stained slides. All available glomeruli per section were included and measured using ImageJ software. The average of the measurements from each zebrafish was used for statistical analysis.
4.6.2. Cleaved Caspase-3 Expression Analysis
Twoobserversscoredthecleavedcaspase-3expressioninthetubulesofeachzebrafish. The semiquantitative score was conducted on the three randomized tubular fields in each zebrafish(20×magnification). Thepercentageofcaspase-3positivearearelativetothetotal area of tubules was scored as 1 (negative staining), 2 (1–10% positive staining), 3 (10%–25% positive staining), 4 (>25% positive staining). The mean of the score from each zebrafish was used for statistical analysis.
4.6.3. Thickness of the Corneal Stroma Analysis
The thickness of the corneal stroma of zebrafish (10×magnification) was measured at the same distances from the center of the eye by using ImageJ software on zebrafish. For each picture, n = 7 measurements at different locations in the eye section (central, middle, periphery) were taken along the cornea at intervals of 100 µM. The average of the measurements was used for statistical analysis.
4.7. Fertility Study
The fertility was assessed by mating female and male zebrafish (wild-type male fish, n = 4 and wild-type female fish, n = 6 and ctns−/−male fish, n = 4 and ctns−/−female fish, n = 6) in a spawning tray. In the next morning, one hour after the light, embryos were collected and the total number of eggs produced was recorded. Fertilized eggs were screened and counted from the unfertile ones using light microscopy. The breeding was performed n = 7 times once a week in a row. Two experiments resulting in no egg productions simultaneously in both wild-type and ctns−/−zebrafish were excluded.
4.8. Locomotory Activity
Zebrafish (wild-type female fish, n = 6; wild-type male fish, n = 6; ctns−/−female fish, n = 6; ctns−/−male fish, n = 6) were placed in the regular housing units and, after an adaptation time of 5 min, were filmed for 5 min. Video recording was executed using a GoPro® HERO 7 placed on top of the tank, thus offering a top view of the fish swimming. Resolution and frame rate were set respectively to 1080 p (1920×1080) and 30 fps. Videos wereanalyzedwithidTracker.aitrackingthetrajectoriesofindividualzebrafishthroughout the experiment. The trajectories were subsequently analyzed with custom python code to derive the average locomotor speed (pixel/sec) in each group [39].

Cistanche-renal
4.9. Body Weight and Length Measurements
Zebrafishwereeuthanizedwithimmersionin300mg/Loftricainemethanesulfonate MS-222. Aftertheverificationoftheabsenceofresponsetoexternalstimuli,the euthanized zebrafish were placed on a paper tissue and dried. The length of the euthanized zebrafish was evaluated using a caliper, measuring from the tip of the mouth to the caudal peduncle. The weight of the euthanized zebrafish was evaluated with an analytical scale.
4.10. Statistical Analysis
Statistical analysis was performed using SPSS (IBM, New York, NY, USA) and GraphpadPrism(GraphPadSoftware, LaJolla). Dataoftwogroupswereanalyzedusingunpaired t-test with Welch’s correction, two-tailed. Data of two categorical independent variables were analyzed with two-way ANOVA with Fisher’s least significant difference (PLSD) test. Differences with p < 0.05 were considered statistically significant.
4.11. Fertility Study
For the comparison of the rates of egg production between wild-type and ctns−/−zebrafish, a Poisson regression analysis using Generalized Estimating Equations with robust standard errors to take account of the correlation of rates with experiments was performed. For the comparison of the odds offer utilized eggs between wild-type and ctns−/−zebrafishAlogistic regression analysis using Generalized Estimating Equations with robust standard errors to account for the correlation of the odds within experiments were performed.
Supplementary Materials: The following are available online at https://www.mdpi.com/article/ 10.3390/ijms22179398/s1.
Author Contributions: Conceptualization, S.P.B., J.H., L.v.d.H., H.J.B., E.L.; methodology, S.P.B., J.H., L.D.G., H.T., T.N., P.B., S.C., B.G.; software S.P.B., J.H., L.D.G., H.T., T.N.; validation, S.P.B., J.H., L.D.G., P.B., L.v.d.H., H.J.B., and E.L.; formal analysis, S.P.B., J.H, L.D.G., H.T., T.N.; investigation, L.v.d.H., H.J.B., E.L.; resources, H.J.B. and E.L.; data curation, S.P.B., J.H., L.D.G., L.v.d.H., H.J.B., E.L.; writing—original draft preparation S.P.B., J.H.; writing—review and editing, L.v.d.H., H.J.B., E.L.; visualization, L.v.d.H., H.J.B., E.L.; supervision, P.d.W., L.v.d.H., H.J.B., E.L.; project administration, L.v.d.H., H.J.B., E.L.; funding acquisition, H.J.B., E.L. All authors have read and agreed to the published version of the manuscript.
Funding: TheauthorswouldliketoacknowledgetheinternalfundfromKULeuvenC1GranttoE.L. (C14/17/113), The China Scholarship Council Grant to J.H. (CSC n◦ 201508500109), and the FWO postdoctoral fellowship to L.D.G. (12I3820N).
Institutional Review Board Statement: The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Ethics Committee of KU Leuven n.142/2019. Acknowledgments: We would like to thank: Didier Cagnini for evaluating the histology of the brain in both ctns−/−and wild-typezebrafish, KathleeenLambaerts, FrédéricHendrickxandKimvanKelst for their constant support and assistance in the KU Leuven aquatic facility, Peter Neeskens for the technical support of the TEM, and Ron Wolterbeek for statistical analysis.
ConflictsofInterest: Theauthorsdeclarenoconflictofinterest. Thefundershadnoroleinthedesign of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.

References
1. Elmonem, M.A.; Veys, K.R.; Soliman, N.A.; van Dyck, M.; van den Heuvel, L.P.; Levtchenko, E. Cystinosis: A review. Orphanet J. Rare Dis. 2016, 11, 47. [CrossRef]
2. Jezegou, A.; Llinares, E.; Anne, C.; Kieffer-Jaquinod, S.; O’Regan, S.; Aupetit, J.; Chablis, A.; Sagne, C.; Debacker, C.; ChadefauxVekemans, B.; et al. Heptahelical protein PQLC2 is a lysosomal cationic amino acid exporter underlying the action of cysteamine in cystinosis therapy. Proc. Natl. Acad. Sci. USA 2012, 109, E3434–E3443. [CrossRef] [PubMed]
3. Besouw, M.T.; Levtchenko, E.N. Improving the prognosis of nephropathic cystinosis. Int. J. Nephrol. Renova. Dis. 2014, 7, 297–302. [CrossRef]
4. Brodin-Sartorius, A.; Tete, M.J.; Niaudet, P.; Antignac, C.; Guest, G.; Ottolenghi, C.; Charbit, M.; Moyse, D.; Legendre, C.; Lesavre, P.; et al. Cysteamine therapy delays the progression of nephropathic cystinosis in late adolescents and adults. KidneyInt. 2012, 81, 179–189. [CrossRef]
5. Cherqui, S. Cysteamine therapy: A treatment for cystinosis, not a cure. Kidney Int. 2012, 81, 127–129. [CrossRef]
6. David, D.; Princiero Berlingerio, S.; Elmonem, M.A.; Oliveira Arcolino, F.; Soliman, N.; van den Heuvel, B.; Gijsbers, R.; Levtchenko,E.MolecularBasisofCystinosis: GeographicDistribution,FunctionalConsequencesofMutationsintheCTNSGene, and Potential for Repair. Nephron 2019, 141, 133–146. [CrossRef] [PubMed]
7. Shimizu, Y.; Yanobu-Takanashi, R.; Nakano, K.; Hamase, K.; Shimizu, T.; Okamura, T. A deletion in the Ctns gene causes renal tubular dysfunction and cystine accumulation in LEA/Tohm rats. Mamm. Genome 2019, 30, 23–33. [CrossRef]
8. Nevo, N.; Chol, M.; Bailleux, A.; Kalatzis, V.; Morisset, L.; Devuyst, O.; Gubler, M.C.; Antignac, C. Renal phenotype of the cystinosis mouse model is dependent upon genetic background. Nephrol. Dial. Transplant 2010, 25, 1059–1066. [CrossRef] [PubMed]
9. Elmonem, M.A.; Berlingerio, S.P.; van den Heuvel, L.P.; de Witte, P.A.; Lowe, M.; Levtchenko, E.N. Genetic Renal Diseases: The Emerging Role of Zebrafish Models. Cells 2018, 7, 130. [CrossRef] [PubMed]
10. Elmonem, M.A.; Khalil, R.; Khodaparast, L.; Khodaparast, L.; Arcolino, F.O.; Morgan, J.; Pastore, A.; Tylzanowski, P.; Ny, A.; Lowe, M.; etal. Cystinosis(ctns)zebrafishmutantshowspronephricglomerularandtubulardysfunction. Sci. Rep. 2017,7, 42583. [CrossRef]
11. Lusco,M.A.;Najafian,B.;Alpers,C.E.;Fogo,A.B.AJKDAtlasofRenalPathology: Cystinosis. Am. J.KidneyDis. 2017,70,e23–e24. [CrossRef]
12. Park, M.A.; Pejovic, V.; Kerisit, K.G.; Junius, S.; Thoene, J.G. Increased apoptosis in cystinotic fibroblasts and renal proximal tubule epithelial cells results from cysteinylation of protein kinase C delta. J. Am. Soc. Nephrol. 2006, 17, 3167–3175. [CrossRef]
13. Veys, K.R.P.; Elmonem, M.A.; Dhaenens, F.; Van Dyck, M.; Janssen, M.; Cornelissen, E.A.M.; Hohenfellner, K.; Reda, A.; Quatresooz, P.; van den Heuvel, B.; et al. Enhanced Intrinsic Skin Aging in Nephropathic Cystinosis Assessed by High-Definition Optical Coherence Tomography. J. Invest. Dermatol. 2019, 139, 2242–2245. [CrossRef]
14. Besouw, M.T.; Kremer, J.A.; Janssen, M.C.; Levtchenko, E.N. Fertility status in male cystinosis patients treated with cysteamine. Fertil. Steril. 2010, 93, 1880–1883. [CrossRef]
15. Veys, K.R.; D’Hauwers, K.W.; van Dongen, A.; Janssen, M.C.; Besouw, M.T.P.; Goossens, E.; van den Heuvel, L.P.; Wetzels, A.; Levtchenko, E.N. First Successful Conception Induced by a Male Cystinosis Patient. JIMD Rep. 2018, 38, 1–6. [CrossRef] [PubMed]
16. Rohayem, J.; Haffner, D.; Cremers, J.F.; Huss, S.; Wistuba, J.; Weitzel, D.; Kliesch, S.; Hohenfellner, K. Testicular function in males with infantile nephropathic cystinosis. Hum. Reprod. 2021, 36, 1191–1204. [CrossRef] [PubMed]
17. Blakey, H.; Proudfoot-Jones, J.; Knox, E.; Lipkin, G. Pregnancy in women with cystinosis. Clin. Kidney J. 2019, 12, 855–858. [CrossRef]
18. Dogulu, C.F.; Tsilou, E.; Rubin, B.; Fitzgibbon, E.J.; Kaiser-Kupper, M.I.; Rennert, O.M.; Gahl, W.A. Idiopathic intracranial hypertension in cystinosis. J. Pediatr. 2004, 145, 673–678. [CrossRef] [PubMed]
19. Dixon, P.; Christopher, K.; Chauhan, A. Potential role of stromal collagen in cystine crystallization in cystinosis patients. Int. J. Pharm. 2018, 551, 232–240. [CrossRef]
20. Katz, B.; Melles, R.B.; Schneider, J.A.; Rao, N.A. Corneal thickness in nephropathic cystinosis. Br. J. Ophthalmol. 1989, 73, 665–668. [CrossRef] [PubMed]
21. Nesterova, G.; Gahl, W.A. Cystinosis: The evolution of a treatable disease. Pediatr. Nephrol. 2013, 28, 51–59. [CrossRef] [PubMed]
22. Ariceta, G.; Giordano, V.; Santos, F. Effects of long-term cysteamine treatment in patients with cystinosis. Pediatr. Nephrol. 2019, 34, 571–578. [CrossRef] [PubMed]
23. Gilbert, M.J.; Zerulla, T.C.; Tierney, K.B. Zebrafish (Danio rerio) as a model for the study of aging and exercise: Physical ability and trainability decrease with age. Exp. Gerontol. 2014, 50, 106–113. [CrossRef] [PubMed]
24. Gerhard, G.S.; Kauffman, E.J.; Wang, X.; Stewart, R.; Moore, J.L.; Kasales, C.J.; Demidenko, E.; Cheng, K.C. Life spans and senescent phenotypes in two strains of Zebrafish (Danio rerio). Exp. Gerontol. 2002, 37, 1055–1068. [CrossRef]
25. Saftig, P.; Klumperman, J. Lysosome biogenesis and lysosomal membrane proteins: Trafficking meets function. Nat. Rev. Mol. Cell Biol. 2009, 10, 623–635. [CrossRef]
26. Cherqui, S.; Sevin, C.; Hamard, G.; Kalatzis, V.; Sich, M.; Pequignot, M.O.; Gogat, K.; Abitbol, M.; Broyer, M.; Gubler, M.C.; et al. Intralysosomal cystine accumulation in mice lacking cystinosis, the protein defective in cystinosis. Mol. Cell. Biol. 2002, 22, 7622–7632. [CrossRef]
27. Raggi, C.; Luciani, A.; Nevo, N.; Antignac, C.; Terryn, S.; Devuyst, O. Dedifferentiation, and aberrations of the endolysosomal compartment characterize the early stage of nephropathic cystinosis. Hum. Mol. Genet. 2014, 23, 2266–2278. [CrossRef]
28. Hard, G.C. Some aids to histological recognition of hyaline droplet nephropathy in ninety-day toxicity studies. Toxicol. Pathol. 2008, 36, 1014–1017. [CrossRef]
29. Sakarcan, A.; Timmons, C.; Baum, M. Intracellular distribution of cystine in cystine-loaded proximal tubules. Pediatr. Res. 1994, 35, 447–450. [CrossRef]
30. Kroeger, P.T., Jr.; Wingert, R.A. Using zebrafish to study podocyte genesis during kidney development and regeneration. Genesis. 2014, 52, 771–792. [CrossRef]
31. Sumayao, R.; McEvoy, B.; Newsholme, P.; McMorrow, T. Lysosomal cystine accumulation promotes mitochondrial depolarization and induction of redox-sensitive genes in human kidney proximal tubular cells. J. Physiol. 2016, 594, 3353–3370. [CrossRef]
32. Chiaverini, C.; Sillard, L.; Flori, E.; Ito, S.; Briganti, S.; Wakamatsu, K.; Fontas, E.; Berard, E.; Cailliez, M.; Chat, P.; et al. Cystinosin is a melanosomal protein that regulates melanin synthesis. FASEB J. 2012, 26, 3779–3789. [CrossRef]
33. Adelmann,C.H.;Traunbauer,A.K.;Chen,B.;Condon,K.J.;Chan,S.H.;Kunchok,T.;Lewis,C.A.;Sabatini,D.M.MFSD12mediates the import of cysteine into melanosomes and lysosomes. Nature 2020, 588, 699–704. [CrossRef]
34. Leal, M.C.; Cardoso, E.R.; Nobrega, R.H.; Batlouni, S.R.; Bogerd, J.; Franca, L.R.; Schulz, R.W. Histological and stereological evaluation of zebrafish (Danio rerio) spermatogenesis with an emphasis on spermatogonial generations. Biol. Reprod. 2009, 81, 177–187. [CrossRef] [PubMed]
35. Sansanwal, P.; Sarwal, M.M. Abnormal mitochondrial autophagy in nephropathic cystinosis. Autophagy 2010, 6, 971–973. [CrossRef] [PubMed]
36. Bellomo, F.; Signorile, A.; Tamma, G.; Ranieri, M.; Emma, F.; De Rasmo, D. Impact of atypical mitochondrial cyclic-AMP level in nephropathic cystinosis. Cell. Mol. Life Sci. 2018, 75, 3411–3422. [CrossRef] [PubMed]
37. De Rasmo, D.; Signorile, A.; De Leo, E.; Polishchuk, E.V.; Ferretti, A.; Raso, R.; Russo, S.; Polishchuk, R.; Emma, F.; Bellomo, F. MitochondrialDynamicsofProximalTubularEpithelialCellsinNephropathicCystinosis. Int. J.Mol. Sci. 2019,21,192. [CrossRef]
38. Kalatzis, V.; Serratrice, N.; Hippert, C.; Payet, O.; Arndt, C.; Cazevieille, C.; Maurice, T.; Hamel, C.; Malecaze, F.; Antignac, C.; et al. The ocular anomalies in a cystinosis animal model mimic disease pathogenesis. Pediatr. Res. 2007, 62, 156–162. [CrossRef]
39. Romero-Ferrero, F.; Bergomi, M.G.; Hinz, R.C.; Heras, F.J.H.; de Polavieja, G.G. idtracker.ai: Tracking all individuals in small or large collectives of unmarked animals. Nat. Methods 2019, 16, 179–182. [CrossRef]






