Schlafens Can Put Viruses To Sleep Part 2
Jun 25, 2023
5. SLFN5 as an Innate Immune Signal Modulator
Although type I IFNs play an important role in host defense against pathogen infection, their production must be properly regulated to avoid inordinately harmful immune responses. Thus, negative regulators are essential for cells to recover from IFN signaling, since IFN production dysregulation leads to autoimmune disorders. Some ISGs can regulate pathways that impact their expression, either positively or negatively.
For example, ISG56 is associated with the adapter protein STING, and disrupts STING interaction with downstream molecules VISA/MAVS or TBK1, inhibiting virus-induced IRF3 activation, IFN expression, and cellular antiviral responses. Another negative regulator is the ISG15 deconjugating protease ubiquitin-specific peptidase 18 (USP18). USP18 inhibits JAK-STAT signaling by interacting with IFNAR2 in a protease-independent manner [64].
Uncoupling protease (uPA) is an important protease that can degrade proteins in the extracellular matrix and participate in physiological and pathological processes such as cell migration, tumor metastasis, and angiogenesis. On the other hand, immunity is an important defense mechanism of the human body against pathogens and tumor cells. So, what is the relationship between uPA and immunity?
Several studies have shown that uPA can affect the function of the immune system through several mechanisms. First, the expression of uPA and its receptor uPAR can regulate the migration and infiltration of immune cells, thereby affecting the occurrence of inflammation and immune response. Second, uPA can activate other proteases on the surface of immune cells, such as MMPs, cathepsins, etc., thereby participating in biological processes such as signal transduction, proliferation, and differentiation. Most importantly, uPA can also affect the process of tumor immune surveillance, making the human immune system unable to recognize and attack cancer cells, thereby promoting tumor growth and metastasis.
Overall, the role of uPA in tumor growth and pathology has been extensively studied, while its effect on the immune system has yet to be further explored. Future research can start from the aspects of uPA in immune cell infiltration, immune regulation, and tumor immune escape, explore the relationship between uPA and the immune system, and provide new ideas and targets for the development of tumor treatment and immunotherapy. From this point of view, we need to pay attention to the improvement of our immunity. Cistanche can significantly improve immunity. Meat paste is rich in various antioxidant substances, such as vitamin C, vitamin C, carotenoids, etc. These ingredients can scavenge free radicals and reduce oxidative stress. Stimulate and improve the resistance of the immune system.

Click cistanche tubulosa benefits
Human SLFN5 has been reported to be a negative regulator of IFN-induced gene transcription [65]. It was found that STAT1 is present as a complex that binds the SLFN5 protein in a type I IFN-dependent manner and binds to the ISRE element in the promoter of ISGs. SLFN5 appears to serve as a repressor of STAT1-induced gene transcription through direct protein interaction. Consistent with this, it was demonstrated that SLFN5 is enriched on the promoters of type I IFN-inducible ISGs, where STAT1 binds.
Microarray experiments revealed that SLFN5 knockout cells expressed more ISGs than wild-type cells, suggesting a potential role for SLFN5 in regulating STAT1-mediated type I IFNinduced transcriptional activation of ISGs [65]. Similarly, in human foreskin fibroblasts and HeLa cells, basal level ISG15, a well-known antiviral protein, increased due to SLFN5 depletion; additionally, rapid induction of ISG15 protein expression by DNA viruses, such as human cytomegalovirus (HCMV), was observed [20]. Accordingly, SLFN5 appears to be a transcriptional repressor of IFN-gene transcription, as well as an IFN-stimulated response gene.
ZEB proteins are zinc-finger E homeobox-binding transcription factors best known for their role in driving epithelial-to-mesenchymal transition and metastasis in some cancers, including BRCA mutant cancer cells [66,67]. They are also widely expressed by immune cells, and regulate important transcriptional networks necessary for immune cell differentiation, maintenance, and function [68].
It has been found recently that human SLFN5 can inhibit ZEB1 transcription by directly binding to the SLFN5 binding motif on the ZEB1 promoter, thereby maintaining the epithelial cell morphology and inhibiting metastasis in BRCA mutant cancer cells [69,70]. SLFN5 increases PTEN by downregulating the transcription of ZEB1. Through the PTEN/PI3K/AKT/mTOR axis, an increase in PTEN inhibits lung adenocarcinoma growth and promotes apoptosis [47].
Although the SLFN5 interaction with the ZEB1 promoter in immune cells has not been validated, these reports suggest roles for SLFN5 as a multifunctional modulator of immune cells. Interestingly, SLFN12 inhibits ZEB1; however, unlike SLFN5, it is assumed to influence post-transcriptional regulation due to its cytoplasmic localization without the nuclear localization signal sequence. SLFN12 overexpression accelerated ZEB1 proteasome degradation and slowed ZEB1 translation in triple-negative breast cancer cells [9].

6. SLFN5, a Double-Edged Sword in IFN Therapy
Some malignancies can be treated with IFN therapy in combination with chemotherapy and radiation. Hematological malignancies and lymphomas can be treated with this therapeutic approach [71]. Recombinant IFNα2b is given to patients with recurrences of melanomas [72]. Hepatitis B and hepatitis C are treated with IFNα and other antiviral drugs, typically combined [73,74]. The anticancer effects of type I IFNs have become extensively recognized in recent decades, particularly their involvement in mediating interactions between tumors and the immune system.
In mouse malignant melanoma and renal cell carcinoma, IFNα promotes the expression of Slfn1, Slfn2, Slfn3, Slfn5, and Slfn8. The loss of Slfn2, Slfn4, or Slfn5 increased cell proliferation and anchorage-independent malignant growth, while decreasing the antiproliferative effect of IFN, implying crucial roles for Schlafens in tumorigenesis and neoplastic cell growth control [75].
All human Schlafen mRNA expression was induced in normal melanocytes by IFN therapy, while only SLFN5 was induced in malignant melanoma cells and renal cell carcinoma cells [8,40]. When melanoma cells are stimulated with IFN, SLFN5 expression is considerably increased, decreasing cancer cell proliferation. In contrast, the depletion of SLFN5 boosted the ability of melanomas to form colonies, even in the presence of IFN [40].
This suggests a potential role of SLFN5 in the anticancer effects of IFNα. However, SLFN5 also potentially reduces the anticancer effect of IFN in glioma cancer cells by transcriptionally co-repressing STAT1-mediated IFN responses, in contrast to its beneficial role in melanoma and renal cell carcinoma [65]. Decreasing SLFN5 leads to increased cellular susceptibility to IFN-induced antiproliferative responses in glioblastoma cells, implying that SLFN5 functions as a negative regulator of the IFN response in glioma cancer cells [65].
Thus, future therapeutic targeting of SLFN5 in malignancies may require precise analysis of other associated factors, and the design of therapeutic targeting of a particular tumor may be required for the selective targeting of SLFN5.
7. Functions of Viral Schlafen
The presence of intact v-Slfn ORFs in some OPVs suggests that it may be preserved for a critical function. Although there are few investigations into the function of v-Slfn, relatively detailed in vitro and in vivo studies on v-Slfn from CMLV have been reported. The expression of this gene was confirmed 2 h after CMLV infection and was expressed at the early stage of infection independent of viral DNA replication [35].
In contrast to mouse Slfn1, the expression of CMLV v-Slfn does not affect the proliferation of mouse fibroblasts. This is thought to be due to the lack of similarity between the first 27 amino acids of mouse Slfn1 and v-Slfn, a region that is essential for mouse Slfn1-mediated fibroblast cell growth inhibition. When the CMLV v-Slfn protein was expressed in VACV lacking intact v-Slfn, it did not affect recombinant virus replication or plaque morphology [35].
Additionally, intradermal infection of mice with this recombinant VACV did not affect skin lesion size [35]. However, in mice with intranasal infection, v-Slfn caused less weight loss and faster recovery compared to the control groups. At three days following in vivo infection, the viral titer was the same as in the control group, but by seven days v-Slfnmediated attenuation was observed.
This suggests that v-Slfn expression does not impede viral replication, but rather accelerates viral clearance by the immune system. This is consistent with the observation that the v-Slfn-bearing recombinant virus was delayed in spreading to the spleen and was more rapidly cleared from this organ. In addition, more extensive recruitment of lymphocytes into infected lung tissue was observed in the presence of v-Slfn expression, although these cells were less activated. Highly virulent viruses can quickly overwhelm their host, limiting viral transmission. The idea that v-Slfn can reduce the virulence of poxviruses, allowing the virus to spread appropriately in the host population, is compelling [35].
A novel feature of v-Slfn in poxviruses was recently discovered (Figure 1). Cyclic GMP-AMP synthase (cGAS) detects cytosolic DNA during virus infection and induces an antiviral state. cGAS activates the stimulator of interferon genes (STING) by synthesizing a second messenger, cyclic GMP-AMP (cGAMP) [76–78]. With the discovery of a viral cGAMP nuclease named Poxin (poxvirus immune nuclease), the immunomodulatory potential of poxviruses was given a new perspective [6].
Recent studies have demonstrated that Poxin, which is a domain of v-Slfns, can degrade cGAMP and is required to avoid cGAS-STING activation [79–81]. Poxin was discovered to be a product of the VACV gene B2R. This gene is also known as p26 in entomopoxviruses and baculoviruses [80]. Most orthopoxviruses include a v-Slfn protein composed of two domains that have evolved from different origins. According to amino acid sequence analysis, a domain resembling the baculovirus p26 sequence is fused to the N-terminus of a v-Slfn domain similar to the murine short form Schlafen [35]; this p26-like domain is Poxin, the cGAMP nuclease. VACV, in which Poxin activity was first reported, does not retain the intact v-Slfn. The loss of Poxin resulted in a considerable reduction in VACV replication in vivo [80].
The importance of self, which includes the Poxin domain, was studied extensively in ectromelia virus (ECTV), which causes mousepox. The Poxin domain, but not the Slfn-like domain, was sufficient to inhibit cGAS-STING signaling with cGAMP nuclease activity in a manner comparable to full-length Poxin–Schlafen-like domain fusion. This suggests that the ECTV Poxin domain preserves the full potential of v-Slfn to prevent the activation of DNA sensing via the cGASSTING axis [79].
In several mouse infection models, the replication of ECTV lacking v-Slfn was significantly attenuated, and mice displayed a robust IFN response [79]. The Poxin– Schlafen-like domain fusion of v-Slfn is highly conserved across orthopoxviruses, such as ECTV, CMLV, and the emerging zoonotic monkeypox virus, implying the importance of cGAMP nuclease activity.
The role of the Slfn-like domain in the activation of Poxin is unclear. Poxin retained its cGAMP nuclease activity in the absence of the Slfn-like domain. Nevertheless, it remains necessary to investigate why the Slfn-like domain is conserved in many OPVs. Given the aforementioned observation that the virulence of chimeric viruses was reduced by adding the Slfn-like domain of CMLV to VACV, it is a plausible hypothesis that regulating viral virulence may contribute to creating favorable conditions for virus propagation in nature.

8. Schlafens as Antiviral Restriction Factors
Antiviral restriction factors are host cellular proteins that operate as a first line of defense, preventing viral replication and spread. Restriction factors recognize pathogens and interfere with specific steps in the virus infectious cycle. The unique properties of restriction factors that serve to limit viruses at early stages include constitutive expression, self-sufficient activity, and immediate action [82]. Restriction factors are occasionally increased in response to IFNs. Although many cell types constitutively express restriction factors at low levels required by cells in the absence of pathogen invasion, the effective control of a pathogen frequently requires the induction of restriction factors in response to infection [83]. Since Schlafens belong to a group of ISGs whose expression is elevated in response to viral infection or stimulation with various pathogen-associated molecular patterns (PAMPs) [36–39], it has been postulated that they may have antiviral activity.
Along with the discovery of the cell biological functions of Schlafens throughout the last decade, interactions with viruses have also been uncovered. In this section, we describe the known antiviral functions of Schlafens, reviewing them in the chronological order in which they were reported (Figure 2). The immune evasion mechanisms by which viruses antagonize many restriction factors have been elucidated. Furthermore, consistent with the theme that viruses can antagonize restriction factors as part of immune evasion mechanisms, there are some recently reported examples of viral strategies to counteract the antiviral action of Schlafens.
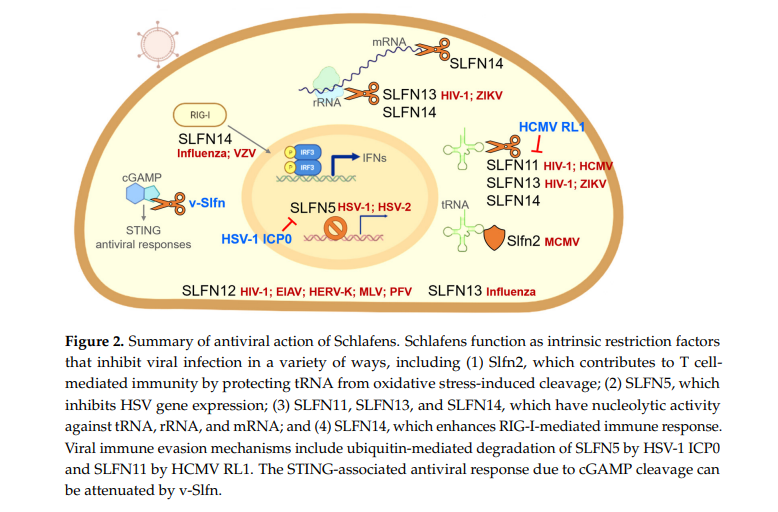
Schlafens belonging to different groups have been reported to have distinct roles, during infection, with many viruses. There is some evidence that the malfunction of group I mouse Slfn2 predisposes cells to virus infection in terms of acquired immunity [55]. Group II SLFN12 is an antiviral factor candidate against vesicular stomatitis virus and various retroviruses, including HIV-1, equine infectious anemia virus (EIAV), human endogenous retrovirus type K (HERK-V), murine leukemia virus (MLV), and primate foamy virus (PFV) [84,85]. However, studies on the interaction of these short or intermediate forms of Schlafens with viruses are lacking, and most studies so far have focused on the antiviral function of group III Schlafens. Therefore, it is crucial to investigate whether the C-terminal extended domain of Schlafens plays a significant role in their intrinsic restriction factor function.
8.1. Roles of SLFN11 during Virus Infection
Human SLFN11 was first reported in 2012 as a potent inhibitor of human immunodeficiency virus 1 (HIV-1) that interferes with viral protein production [18]. It was discovered by Li et al. that SLFN11 binds transfer RNAs (tRNAs) and suppresses protein production selectively dependent on codon usage [18]. Further research revealed that equine SLFN11 inhibits the formation of EIAV by a mechanism similar to that employed by human SLFN11 [23]. A systematic investigation of the HIV replication cycle demonstrated that SLFN11 does not affect reverse transcription, integration, or the generation and nuclear export of viral RNA, nor does it interfere with viral particle budding or release. Instead, it was found to induce the selective inhibition of viral protein synthesis.
By exploiting a particular viral codon bias on the A/T nucleotide, SLFN11 functions at the moment of viral protein production. Although the antiviral effect of SLFN11 was similar to that of other viruses with an uncommon codon bias, such as influenza, it was not effective against the adenoassociated virus or herpes simplex virus (HSV). These findings established that SLFN11 is a highly effective interferon-inducible restriction factor for retroviruses, such as HIV, that mediates antiviral effects via codon usage discrimination [18]. This intriguing finding may partially explain the previously observed IFN suppression of viral protein-specific synthesis in HIV-infected cells [18,86]. It also highlights how the immune system can exploit possible differences between self and non-self for host cells to target and eliminate viruses. There does not appear to be a preference for tRNA type in the binding of SLFN11 to tRNAs [18].
It will be necessary to conduct biochemical experiments to unveil how SLFN11 modulates tRNA function and influences virus-specific codon usage. SLFN11 is highly expressed, not only in CD4+ T cells but also in monocytes and moDCs [37,87]. CD4+ T cells are known to be the primary reservoir for latent HIV infection, and HIV latency can also be established in monocytes and macrophages [88]. Thus, high expression of SLFN11 in these cells is thought to have a role in HIV latent infection and may be a key component of the innate immune response to HIV.
It was recently discovered that mouse Slfn2 binds to tRNA and inhibits its degradation in an oxidative stress environment [89]. Although this study showed that Slfn2 inhibited murine cytomegalovirus (MCMV) infection, the result was due to T cell-mediated adaptive immunity [89]. Nonetheless, these observations merit a thorough examination of the interaction between tRNA modulation of Slfn2 and murine retroviruses, as well as the parallels and differences with human SLFN11. Since the N-terminal portion of SLFN11 is involved in tRNA binding, there may be evolutionary similarities in sequence with the short form Slfn2. Additionally, the discovery that SLFN13 and SLFN14 participate in tRNA modulation paves the path for future investigations to identify whether Schlafens share common functions in tRNA biology [24,90].
Since the incoming viral genome of positive-sense single-stranded RNA viruses requires immediate translation to allow replication, these viruses are particularly sensitive to the effects of SLFN11 on protein synthesis. This has been demonstrated in the Flavivirus genus, including the West Nile virus (WNV), dengue virus (DENV), and Zika virus (ZIKV) [21]. There are similarities and differences in the mechanism of action of the Schlafen proteins against flaviviruses and lentiviruses. The N-terminal portion of SLFN11 is essential and sufficient for antiviral activity, as it prevents virus-induced alterations in the tRNA repertoire of infected cells. In contrast to WNV infection, which affected only a subset of tRNAs in SLFN11-deficient cells [21], HIV-1 raised tRNA levels overall in the absence of SLFN11 [18].
The ability of SLFN11 to regulate the abundance of tRNA pools could be related to the sensitivity of cells to DNA-damaging agents. Several studies have found that cancer cells with higher SLFN11 expression are more vulnerable to DNA-damaging agents [12,33,91,92]. Higher SLFN11 levels may limit the number of particular tRNAs that influence the translation of DNA repair proteins encoded by codon-biased open reading frames, such as ATM and ATR [93]. In addition, SLFN11 irreversibly inhibits DNA replication at DNA damage sites in a C-terminal helicase domain-dependent manner [34,94]. It has been known that various viruses exploit proteins involved in the DNA damage response of host cells for their effective replication [95].
The involvement of DNA damage control proteins ATM and ATR in HIV infection has been studied extensively. ATM has a positive effect on late gene expression of HIV and the function of Rev, a viral post-transcriptional regulator [96]; meanwhile, ATR kinase activity is required to complete the viral DNA integration process and support the survival of transduced cells [97]. In ZIKV infection, the ATM signaling pathway increases viral replication [98]. These findings suggest that Schlafens should be further investigated in terms of host cell resistance to viruses that favorably exploit DNA damage responses to ensure efficient replication.
ZIKV has generated widespread concern in recent years because of its ability to induce birth abnormalities in infants and Guillain–Barré syndrome in adults. ZIKV can be transmitted sexually, survive in the male reproductive system [99], and, in females, pass the placenta to infect the fetus [100]. Limited information is available on the effects of ZIKV on reproductive health and fertility. Given that SLFN11 is not expressed in the placenta or testes [22], additional research is needed to discover whether it is also connected with prenatal and sexually transmitted infections.
The SLFN11 gene evolved under repeated positive selection in primates [22]. Furthermore, the antiviral efficiency of SLFN11 was highest in non-human primate species, such as gibbons and marmosets, but less effective in humans and in bonobo species that are evolutionarily close to humans, indicating that the effects of SLFN11 have become highly species-specific over time [22]. SLFN11 is functional in the absence of infection and reduces protein production from certain host transcripts [18,93]. This implies that SLFN11 may inhibit protein synthesis from non-codon-optimized transcripts in general, thereby pre-establishing an unfavorable cellular environment for viral protein synthesis.
Viruses have evolved ways that counteract host restriction factors. Although a decreasing trend in SLFN11 proteins in HCMV-infected cells was demonstrated [101], viral antagonists for SLFN11 have not been discovered for many years. However, the antiviral effect of SLFN11, and its viral antagonistic mechanism on HCMV, have recently been demonstrated [102]. The late-expressed protein RL1 of HCMV targets SLFN11 for proteasome degradation and is the first discovery of a viral antagonist to this restriction factor. In this study, it was revealed that the cellular CRL4 E3 ubiquitin ligase complex is additionally involved in the degradation of SLFN11 by RL1 [102].
Even though SLFN11 has a significant impact on HIV, WNV, and ZIKV replication, these viruses can still replicate in cells that express SLFN11. Compared to other flaviviruses or HIV, DENV replication is significantly reduced by SLFN11 expression [21]. This suggests that DENV is more susceptible than other viruses to the effects of SLFN11. Thus, it would be expected that DENV lacks an antagonistic mechanism for SLFN11, whereas WNV, ZIKV, and HIV-1 may possess veiled antagonistic mechanisms.
The mechanism by which the phosphorylation of SLFN11 by protein phosphatase 1 catalytic subunit G (PPP1CC) regulates type II tRNA cleavage ability has been reported [103]. Cellular protein activity is well known to be regulated by viral kinases [104]. No evidence has yet been found to support the hypothesis that viruses regulate the phosphorylation of SLFN11 through virus-encoded kinases, or indirectly through host cell kinases, such as PPP1CC. Further research is required to explore the possibility that viruses exploit protein phosphorylation to circumvent the antiviral activity of Schlafens, as has been observed for other host restriction factors [105–112].
8.2. Roles of SLFN13 during Virus Infection
Crystallographic analysis revealed that SLFN13 is a new class of tRNA/rRNA nucleases [24]. In addition, it was also reported that SLFN13 had an antiviral function against HIV and ZIKV by inhibiting protein synthesis through nucleolytic activity, similar to SLFN11. However, the key determinant of tRNA cleavage by SLFN13, which blocks protein synthesis, is the secondary structure of the tRNA and is not correlated with the anti-codon sequence [24], which appears to be different from the codon usage-based mechanism of SLFN11.
The sequence of the N-terminal domain of SLFN13, which is required for enzyme function, is conserved in other Schlafen proteins. However, specific positively charged amino acid residues are different. It was confirmed that certain family members, such as human SLFN5 and mouse Slfn1, are not involved in tRNA cleavage [24]. Thus, it is likely that the distribution of positively charged amino acid residues inside the N-terminal domain can determine the ability and selection tendencies of tRNA/rRNA cleavage as well as antiviral spectra for other Schlafens.
Influenza A (PR8) and B (Victoria) virus infections were observed to induce SLFN13 mRNA expression in human lung adenocarcinoma A549 cells [19]. This induction was more robust in viral NS1-deficient mutant infection, presumably due to the ability of NS1 to repress RIG-I-mediated activation of the IFN promoter [113]. Furthermore, the depletion of SLFN13 increased influenza A and B virus plaque development, implying that SLFN13 promotes antiviral responses to these viruses [19]. However, whether the SLFN13 antiviral function against the influenza virus is related to tRNA/rRNA cleavage is unknown. Therefore, there is a need to determine whether Schlafen nucleolytic activity is a common mechanism for Schlafen-mediated antiviral function. The absence of an antiviral effect of SLFN11 against a virus with a negative-sense single-stranded RNA genome [21] suggests the existence of a mechanism independent of the anti-influenza virus function of SLFN13.
8.3. Roles of SLFN14 during Virus Infection
Antiviral functions have also been reported for SLFN14, and expression is increased by influenza A infection [19]. Depletion of SLFN14 limited the upregulation of IP-10, a major ISG, following influenza infection. These results suggest a possible mechanism by which SLFN14 recognizes the viral RNA genome, enhances the activating RIG-I mediated signal, and inhibits influenza replication [19]. However, it is necessary to confirm whether SLFN14, the same as for helicases, such as DDX1 or RIG-I, truly detects the viral genome [114]. SLFN14 delays the nuclear translocation of nucleoprotein NP. Delayed nuclear translocation of NP may impair viral replication by impairing viral ribonucleoprotein nuclear transport.
In addition to its effects on RNA viruses, SLFN14 has also been demonstrated to possess antiviral activity against DNA viruses, such as the varicella-zoster virus (VZV). VZV infection induces SLFN14 expression and inhibits viral antigen production in cells overexpressing SLFN14 [19]. Although SLFN14’s antiviral mechanism against RNA viruses and DNA viruses is assumed to be distinct, additional research on the putative helicase domain of SLFN14 and the association of RIG-I-mediated IFN signaling is required for a more detailed mechanism analysis. In addition, since cell types expressing SLFN14 are very limited, or the expression level is low [115], the genuine function of SLFN14 in virus-infected cells remains to be evaluated.
SLFN14 has been found to have ribosome-associated endonuclease activity and can degrade tRNA, rRNA, and mRNA [90]. There is no sequence specificity or preferred structure specificity in RNA cleavage, and this enzymatic activity is strictly Mg2+- and Mn2+-dependent and ATP-independent [90]. However, only the C-terminally truncated short version of SLFN14 exhibited enzymatic activity, whereas full-length SLFN14 lacked endonuclease activity and did not bind to ribosomes [90]. This feature appears to be a way to maintain the integrity of cellular RNAs. Since the SLFN14 protein is present at low levels in most cells and occurs in the nucleus, an inactive precursor state similar to caspase may shield cellular RNAs against non-specific endonuclease activity. Viral infections induce SLFN14 expression like that of RNase L [116], and it may participate in the clearance of total cellular RNA to inhibit viral reproduction. However, it is still required to demonstrate that SLFN14 is processed into the active form after infection or in certain environments.
8.4. Roles of SLFN5 during Virus Infection
In human cells, SLFN5, along with SLFN11, is the most abundant Schlafen family protein [18]. SLFN5 is a nuclear member of the Schlafen family, which has been linked to immune cell proliferation and differentiation [28,117].
Studies report that influenza virus, WNV, and rhinovirus infection result in increased SLFN5 expression [37,118,119]. However, the function of SLFN5 against these viruses has not been investigated, and unlike SLFN11, it has been experimentally established that SLFN5 has no antiviral activity against HIV infection [18]. A recent investigation of SLFN5 revealed an antiviral action and mechanism against HSV-1, a virus with a double-stranded DNA genome [20]. In that study, host factors associated with HSV-1 DNA were isolated using a proteomics technique called Isolation of Proteins On Nascent DNA (iPOND), which identifies proteins accumulating on newly synthesized DNA [120–122]. When applied to HSV-1 infection with wild-type and mutant viruses, this technique revealed that SLFN5 undergoes proteasomal degradation as a result of accelerated ubiquitination by viral protein ICP0.
The HSV-1 immediate-early protein ICP0 facilitates viral gene transcription and virus reactivation from latency. ICP0 features a ubiquitin E3 ubiquitin ligase domain that antagonizes host defenses through the proteasomal degradation of intrinsic antiviral host factors [123,124]. The HSV-1 DNA has been found associated with several ICP0 degradation targets, which were also shown to inhibit the production of viral genes and/or the activation of antiviral cell signals [124]. Although previous studies identified ICP0 substrates as restriction factors, the mechanism for suppressing viral gene expression is not fully understood. In this recent study, it was confirmed biochemically that ICP0 specifically ubiquitinated and degraded SLFN5 via the proteasome [20].
The direct interaction between ICP0 and SLFN5 was found to occur via the extended C-terminal domain of SLFN5, a region that is absent in SLFN11, which was not targeted for degradation. The C-terminal region of SLFN5 contains an intrinsically disordered region, a frequent feature of cellular proteins bound by viral factors, such as ICP0 [125]. The antiviral effect of SLFN5 on HSV-1 is more clearly observed in mutant viruses lacking the E3 ligase activity of ICP0 than in wild-type viruses. The observations that HSV-1 targets SLFN5 [20] and HCMV targets SLFN11 [102] suggest that proteasome-mediated degradation may be a more common viral strategy used to antagonize Schlafen restriction.
The antiviral mechanism of SLFN5 has been proposed to bind to viral DNA and inhibit RNA polymerase II loading onto viral gene promoters [20]. Additionally, it was proven that, unlike other Schlafens, it did not affect mRNA degradation. Although putative helicase activity may be the mechanism by which SLFN5 represses viral DNA gene transcription, the Walker A helicase motif of SLFN5 does not affect its antiviral function [20]. SLFN5 does not appear to have any DNA sequence specificity. The examination of SLFN5 binding to promoter and gene body regions revealed no apparent preference; however, there was a noticeable tendency to bind more to viral DNA over cellular DNA [20]. A recent study of SLFN5 structure demonstrated a high affinity for double-stranded DNA and identified the residues involved in nucleic acid binding [31]. Although SLFN5 binds tRNA, it does not share the endoribonuclease activity reported for other Schlafens [31]. SLFN5 was also shown to have a preference for binding to free DNA over nucleosome-bound DNA [31]. Perhaps the ease of access to euchromatic viral DNA in the lytic infection environment may contribute to selective binding to viral genomic DNA [126].
The interaction between SLFN5 and viral DNA was detected for both incoming viral genomes and actively replicating viral genomes at viral replication compartments [20]. The host PML protein, a well-known HSV-1 restriction factor, as well as an ICP0 substrate, also accesses incoming viral DNA and inhibits viral gene transcription [127–130]. Although further biochemical studies are required, the ICP0-mediated degradation of SLFN5 appears to be less efficient than PML degradation [20]. Immediately upon infection, the majority of HSV-1 DNA is surrounded by PML protein; however, when ICP0 is expressed, PML is rapidly eliminated, and viral DNA is once again entrapped by SLFN5 protein [20].
This suggests that PML and SLFN5 may work cooperatively to create an unfavorable environment for viral gene expression. Therefore, the role of SLFN5 may be a second line of defense supporting the antiviral function of PML (Figure 3). The observation that SLFN5 regulates immune responses, and is also targeted by ICP0, suggests that it may form part of a ‘self-guarded’ immune pathway to monitor infection. The degradation of SLFN5 by ICP0 could thus trigger the activation of secondary immune responses. This guard hypothesis was recently suggested for MORC3, another target of the ICP0 [131]. Further studies are required to understand the connections between SLFN5 and other regulators of HSV-1 infection.

A recent analysis of the HSV-1 single-cell transcriptome revealed that β-catenin recruitment to the viral replication compartment is required for HSV-1 gene expression [132]. SLFN5 is known to inhibit cell migration and proliferation by inhibiting the expression of βcatenin [133,134], implying that SLFN5 could also indirectly affect HSV-1 gene expression.
Even though SLFN5 has no antiviral activity against retroviruses, it does have an antiviral effect against HSV-2, an alphaherpesvirinae close to HSV-1. Interestingly, the results for betaherpesvirinae HCMV differed depending on the stage of infection [20]. Within the first 24 h after infection, SLFN5 depletion promotes the expression of viral immediate early and early gene transcripts; however, this is reversed in the late phase, resulting in the reduced expression of these viral genes in the absence of SLFN5. As a result, HCMV replication yield is slightly decreased in SLFN5-deficient cells. One difference between HSV-1 and HCMV is the time course of infection, with HSV-1 replication being much faster than HCMV.
Since SLFN5 inhibits STAT1-mediated ISGs transcription [65], SLFN5 depletion may result in increased ISG signaling, which decreases HCMV replication. Indeed, knocking down SLFN5 resulted in higher levels of ISG15 expression, which increased further following HCMV infection [20]. As a result, SLFN5 is directly involved in reducing early viral gene expression, and it appears to have a distinct effect on HSV-1 at later stages. Another DNA virus, adenovirus, was unaffected by SLFN5, and viral infection did not result in SLFN5 protein degradation [20]. Together, these data suggest specificity to the antiviral activity across the Schlafen protein family, similar to what has been observed for other families of host restriction factors.
9. Conclusions and Future Perspectives
Continuous and in-depth research on the Schlafen family has made significant progress toward elucidating the roles of Schlafen proteins in recent years. Current studies have shown that Schlafen proteins play critical roles in regulating both the immune response and the cell cycle. Some of these proteins are associated with tumor treatment susceptibility and drug resistance [13–15,135]; thus, the biological function of Schlafen family proteins in tumor cells provides new methods and ideas for tumor detection and treatment. In addition, the Schlafen proteins exhibit a relatively broad inhibitory effect on retroviruses via RNA modulation to inhibit translation. Schlafen proteins have also been implicated in viral infection indirectly via interferon signaling. The discovery of a mechanism for the direct inhibition of viral gene expression through SLFN5 binding to viral DNA in the nucleus has highlighted the potential diversity in the antiviral mechanisms of the Schlafen family.
Numerous findings to date demonstrate that the Schlafen family has a role in a variety of cellular responses, including immune cell development and intrinsic/innate immunity. This protein family is, unquestionably, an important target for cancer treatment, as well as research into understanding and preventing viral infections. However, functional studies on the Schlafen proteins are still in their infancy, and many important questions remain to be solved. Although the Schlafen family shares several similar domains, they show functional differences. These distinctions imply that the Schlafen family members confer specificity to their antiviral activities, highlighting the importance of studying structural properties and functional mechanisms.
Fortunately, structures have been determined for rat Slfn13 [24] and human SLFN5 [31], providing insight for ongoing studies of Schlafen family proteins. Furthermore, the limitations of animal experiments for clinical application must be overcome. The Schlafen family showed a rapid evolutionary tendency in several rodents, and the degree of conservation between rodent and human Schlafen genes is not high. For example, SLFN5 and SLFN11 are the most abundant and highly studied in various cells, but the similarity between mice and humans of SLFN5 is only 59% based on the amino acid sequence identity, and there is no SLFN11 ortholog in mice. SLFN5 and SLFN14 are the only orthologs shared between mice and humans (Figure 1). Therefore, there is a need to develop a new platform, such as organoid models, that can substitute for in vivo studies.
Given the diverse functions of Schlafen family proteins, various binding partner proteins in the cell are expected to play roles in their regulation. Although no report on the results of a global proteomic approach to interactome has been published, it is critical to discover and study the role of binding partners as factors that differentiate the function and regulation of intracellular activity of Schlafen family proteins. The study of associations between Schlafen expression level and cancer prognosis can be applied to virus-mediated tumor research or treatment using viral vectors. Despite their name, the Schlafen field is far from a sleepy one. Ongoing studies will provide important insight into both virus and tumor biology and will suggest ways that their unique activities can be harnessed for therapeutic applications.

Author Contributions:
Conceptualization, E.T.K. and M.D.W.; writing—original draft preparation, E.T.K.; writing—review and editing, M.D.W.; visualization, E.T.K. All authors have read and agreed to the published version of the manuscript.
Funding:
This work was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2021R1A2C1010313). M.D.W. was supported in part by grants from the National Institutes of Health (AI115104 and NS082240) and funds from the Children’s Hospital of Philadelphia.
Acknowledgments:
We thank Joe Dybas for his comments on the manuscript.
Conflicts of Interest:
The authors declare no conflict of interest.
References
1. Schwarz, D.A.; Katayama, C.D.; Hedrick, S.M. Schlafen, a new family of growth regulatory genes that affect thymocyte development. Immunity 1998, 9, 657–668. [CrossRef]
2. Geserick, P.; Kaiser, F.; Klemm, U.; Kaufmann, S.H.E.; Zerrahn, J. Modulation of T cell development and activation by novel members of the Schlafen (slfn) gene family harboring an RNA helicase-like motif. Int. Immunol. 2004, 16, 1535–1548. [CrossRef] [PubMed]
3. Neumann, B.; Zhao, L.; Murphy, K.; Gonda, T.J. Subcellular localization of the Schlafen protein family. Biochem. Biophys. Res. Commun. 2008, 370, 62–66. [CrossRef] [PubMed]
4. Kuang, C.; Yang, T.; Zhang, Y.; Zhang, L.; Wu, Q. Schlafen 1 inhibits the proliferation and tube formation of endothelial progenitor cells. PLoS One 2014, 9, e109711. [CrossRef] [PubMed]
5. Brady, G.; Boggan, L.; Bowie, A.; O’Neill, L.A.J. Schlafen-1 Causes a Cell Cycle Arrest by Inhibiting Induction of Cyclin D1. J. Biol. Chem. 2005, 280, 30723–30734. [CrossRef]
6. Oh, P.-S.; Patel, V.B.; Sanders, M.A.; Kanwar, S.S.; Yu, Y.; Nautiyal, J.; Patel, B.B.; Majumdar, A.P.N. Schlafen-3 decreases cancer stem cell marker expression and autocrine/juxtacrine signaling in FOLFOX-resistant colon cancer cells. Am. J. Physiol. Gastrointest. Liver Physiol. 2011, 301, G347–G355. [CrossRef]
7. Patel, V.B.; Yu, Y.; Das, J.K.; Patel, B.B.; Majumdar, A.P.N. Schlafen-3: A novel regulator of intestinal differentiation. Biochem. Biophys. Res. Commun. 2009, 388, 752–756. [CrossRef]
8. Sassano, A.; Mavrommatis, E.; Arslan, A.D.; Kroczynska, B.; Beauchamp, E.M.; Khuon, S.; Chew, T.-L.; Green, K.J.; Munshi, H.G.; Verma, A.K.; et al. Human Schlafen 5 (SLFN5) Is a Regulator of Motility and Invasiveness of Renal Cell Carcinoma Cells. Mol. Cell. Biol. 2015, 35, 2684–2698. [CrossRef]
9. Al-Marsoummi, S.; Vomhof-DeKrey, E.; Basson, M.D. Schlafen12 Reduces the Aggressiveness of Triple Negative Breast Cancer through Post-Transcriptional Regulation of ZEB1 That Drives Stem Cell Differentiation. Cell. Physiol. Biochem. 2019, 53, 999–1014. [CrossRef]
10. Al-Marsoummi, S.; Pacella, J.; Dockter, K.; Soderberg, M.; Singhal, S.K.; Vomhof-Dekrey, E.E.; Basson, M.D. Schlafen 12 Is Prognostically Favorable and Reduces C-Myc and Proliferation in Lung Adenocarcinoma but Not in Lung Squamous Cell Carcinoma. Cancers 2020, 12, 2738. [CrossRef]
11. Companioni Nápoles, O.; Tsao, A.C.; Sanz-Anquela, J.M.; Sala, N.; Bonet, C.; Pardo, M.L.; Ding, L.; Simo, O.; Saqui-Salces, M.; Blanco, V.P.; et al. SCHLAFEN 5 expression correlates with intestinal metaplasia that progresses to gastric cancer. J. Gastroenterol. 2016, 52, 39–49. [CrossRef] [PubMed]
12. Zoppoli, G.; Regairaz, M.; Leo, E.; Reinhold, W.C.; Varma, S.; Ballestrero, A.; Doroshow, J.H.; Pommier, Y. Putative DNA/RNA helicase Schlafen-11 (SLFN11) sensitizes cancer cells to DNA-damaging agents. Proc. Natl. Acad. Sci. USA 2012, 109, 15030–15035. [CrossRef] [PubMed]
13. Tian, L.; Song, S.; Liu, X.; Wang, Y.; Xu, X.; Hu, Y.; Xu, J. Schlafen-11 sensitizes colorectal carcinoma cells to irinotecan. Anticancer. Drugs 2014, 25, 1175–1181. [CrossRef] [PubMed]
14. Nogales, V.; Reinhold, W.C.; Varma, S.; Martinez-Cardus, A.; Moutinho, C.; Moran, S.; Heyn, H.; Sebio, A.; Barnadas, A.; Pommier, Y.; et al. Epigenetic inactivation of the putative DNA/RNA helicase SLFN11 in human cancer confers resistance to platinum drugs. Oncotarget 2015, 7, 3084–3097. [CrossRef] [PubMed]
15. Stewart, C.A.; Tong, P.; Cardnell, R.J.; Sen, T.; Li, L.; Gay, C.M.; Masrorpour, F.; Fan, Y.; Bara, R.O.; Feng, Y.; et al. Dynamic variations in epithelial-to-mesenchymal transition (EMT), ATM, and SLFN11 govern response to PARP inhibitors and cisplatin in small cell lung cancer. Oncotarget 2017, 8, 28575–28587. [CrossRef] [PubMed]
16. Shee, K.; Wells, J.D.; Jiang, A.; Miller, T.W. Integrated pan-cancer gene expression and drug sensitivity analysis reveals SLFN11 mRNA as a solid tumor biomarker predictive of sensitivity to DNA-damaging chemotherapy. PLoS One 2019, 14, e0224267. [CrossRef]
17. Winkler, C.; Armenia, J.; Jones, G.N.; Tobalina, L.; Sale, M.J.; Petreus, T.; Baird, T.; Serra, V.; Wang, A.T.; Lau, A.; et al. SLFN11 informs on the standard of care and novel treatments in a wide range of cancer models. Br. J. Cancer 2021, 124, 951–962. [CrossRef]
18. Li, M.; Kao, E.; Gao, X.; Sandig, H.; Limmer, K.; Pavon-Eternod, M.; Jones, T.E.; Landry, S.; Pan, T.; Weitzman, M.D.; et al. Codon-usage-based inhibition of HIV protein synthesis by human schlafen 11. Nature 2012, 491, 125–128. [CrossRef]
19. Seong, R.-K.; Seo, S.; Kim, J.-A.; Fletcher, S.J.; Morgan, N.V.; Kumar, M.; Choi, Y.-K.; Shin, O.S. Schlafen 14 (SLFN14) is a novel antiviral factor involved in the control of viral replication. Immunobiology 2017, 222, 979–988. [CrossRef]
20. Kim, E.T.; Dybas, J.M.; Kulej, K.; Reyes, E.D.; Price, A.M.; Akhtar, L.N.; Orr, A.; Garcia, B.A.; Boutell, C.; Weitzman, M.D. Comparative proteomics identifies Schlafen 5 (SLFN5) as a herpes simplex virus restriction factor that suppresses viral transcription. Nat. Microbiol. 2021, 6, 234–245. [CrossRef]
For more information:1950477648nn@gmail.com






