SLC37A2, A Phosphorus-related Moleculeincreases in Smooth Muscle Cellsin The Calcified Aorta Ⅱ
Feb 04, 2024
Results
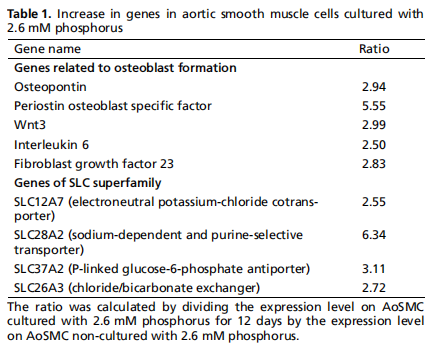
Search for candidate molecules in phosphate-inducedcalcified AoSMC.To search for molecules whose expression increases in calcified AoSMC, DNA microarray analysis of about 26,000 genes was performed using calcified AoSMC and noncalcified AoSMC mRNA. Figure 1 shows the HeatMap of different genes expressed between AoSMC not cultured with 2.6 mM And those cultured with 2.6 mM P for 12 days. A total of 3,066 genes increased more than two-fold on cultured vs non-cultured AoSMC.Genes with increased expression were molecules related to osteoblast formation such as osteopontin, periostin osteoblast-specific factor (POSTN), Wnt3, interleukin-6 (IL-6), and fibroblast growth factor 23 (FGF23) as well as several molecules in theSLC superfamily (Table 1). Among these molecules, we focused on SLC37A2for which mRNA increased about 3.1 times onAoSMC cultured with 2.6 mM P for 12 days compared to AoSMCnot cultured with 2.6 mM P. SLC37A2 has been reported to be related to P regulation, but its detailed function is unknown effect of inflammation on SLC37A2 expression. Vascular calcification has been reported to be promoted by inflammation so we examined whether the SLC37A2 mRNA is affected by inflammatory response. At 24 h after LPS treatment, IL-6 significantly increased compared to AoSMC not treated with LPS(Fig. 2A). These results were also seen 72 h after LPS treatment. There were no significant differences between groups in SLC37A2mRNA expression (Fig.2B).

CLICK HERE TO GET NATURAL ORGANIC CISTANCHE EXTRACT WITH 25% ECHINACOSIDE AND 9% ACTEOSIDE FOR KIDNEY FUNCTION
Supportive Service Of Wecistanche-The largest cistanche exporter in the China:
Email:wallence.suen@wecistanche.com
Whatsapp/Tel:+86 15292862950
Shop For More Specifications Details:
https://www.xjcistanche.com/cistanche-shop
Expression of SLC37A2 in phosphate-induced calcified AoSMC.To determine whether SLC37A2 is involved in calcification, we examined the expression of SLC37A2 mRNA on non-calcified and calcified AoSMC (Fig.3). Calcification was observed in AoSMC cultured with 2.6 mM P for 6 days, and more advanced calcification was observed in AoSMC cultured with2.6 mM P for 12 days (Fig. 3A). Runx2 mRNA significantly increased on calcified AoSMC compared to non-calcified AoSMC(Fig. 3B).SLC37A2 mRNA significantly began to increase on day 3 in calcified AoSMC, and on day 9, calcified AoSMCsignificantly increased about two-fold compared with noncalcified AoSMC (Fig. 3C). Expression of SLC37A2 protein significantly increased about two-fold on day 6 in calcified AoSMC compared with non-calcified AoSMC (Fig. 3D).
Expression of SLC37A2 in CKD model rats.
To reveal whether SLC37A2 is involved in vascular calcification in vivo, we examined SLC37A2 mRNA expression on vessels of adenine-induced CKD rats. Adenine-induced CKD rats were created according to the protocol shown in Fig. 4A. Table 2 shows body weight and biochemical data on the day of sacrifice. The body weight of adenine-induced CKD rats was significantly lower compared with non-CKD rats. Compared with the non-CKD group, CKD groups had higher plasma P and Cr levels, and lower plasma Calevels, and 24-hour urinary excretion of P, Ca, and Cr. Further, these results were more prominent in the CKD-HP group, as seen in previous studies.37,39) Thus, renal function decreased in the CKD group, particularly in the CKD-HP group. We examined mRNA expression in the aortic vessels of CKD rats(Fig. 4). Although there were no significant differences in the expression of Runx2 mRNA, a transcription factor that moves early in the mechanism of vascular calcification, between groups (Fig. 4B) expression of osteopontin mRNA significantly increased in a theCKD-HP group compared to the non-CKD group and the CKD-LPgroup (Fig. 4C).SLC37A2 mRNA expression was also significantly increased in the CK D-HP group compared to the non-CKD group and CKD-LP group (Fig. 4D). Because there were large individual differences in these rats, we examined the correlation between osteopontin and SLC37A2 mRNA and found a significant correlation between these molecules ( Fig. 4E). Histological examination of the thoracoabdominal aorta revealed medial calcification only in the aorta of the CKD-HPgroup (Fig. 5A). To examine the expression site of SLC37A2 on the aorta, we conducted immunostaining of the aorta of CKD-HPrats. SLC37A2 was strongly expressed at the area where vascular calcification occurred (Fig. 5B).

Fig. 1. The HeatMap of differently expressed genes between AoSMCnon-cultured with 2.6 mM P and AoSMC cultured with 2.6 mM P. Red represents genes with a high signal level, and green represents genes with a low signal level. AOSMC aortic smooth muscle cells. See the figure in the online version.


Discussion
We investigated candidate P-related molecules involved in vascular calcification. Our results showed that SLC37A2 is one of the molecules that increase with vascular calcification in Vitro and in vivo.
The SLC37 family belongs to the SLC superfamily and includes proteins: SLC37A 1-440) The SLC37 family contains transmembrane proteins in the endoplasmic reticulum membrane and has a sequence homologous to the bacterial organophosphatephosphate antiporter.(41) SLC37A4 is better known as the glucose6-phosphate transporter.(2-44 However, there are few reports about SLC37A2, and details about its function are not fully understood. SLC37A2, also known as SPX2, is abundantly expressed in murine macrophages, spleen, thymus, and white adipose tissue (WAT) of genetically obese mouse models and osteoclast-like cells derived from Raw264.7 cells.(45.6) TheSLC37A2 protein transports both glucose-6-phosphate/phosphateand phosphate/phosphate exchanges.(4) Recently, reports have shown an association of SLC37A2 with animal disease. (47-49) An SLC37A2 defect causes craniomandibular osteopathy (CMO), a self-limiting proliferative bone disease, in terrier breeds. (47) In dairy cattle, a SLC37A2 homozygous mutation is responsible for embryonic death. (8.9) In addition. SLC37A2 has been considered a target for vitamin D target genes. (so) The potential for the phosphor-Ser294 progesterone receptor target gene in the progression of breast cancer has also been considered(1)It has been reported that inflammation may both cause and exacerbate vascular calcification. (2s26) Furthermore. SLC37A2 is abundantly expressed in macrophages and WAT which are affected by macrophage infiltration. (s) Therefore. we considered that SLC37A2 might be a molecule expressed during inflammation. However, our findings showed no increase in SLC37A2 expression in AoSMC exposed to an inflammatory response, this molecule does not appear to be directly affected by inflammatory stimulation. SLC37A2 may be involved in vascular calcification through a mechanism other than inflammation.

It has been reported that SLC37A2 acts as a P-linked anti-porter. (41) In addition, SLC37A2 was reported to be a target gene for vitamin D, and the SLC37A2 gene contains a major vitamin Dreceptor-binding site(ss2) Systematically correlated changes in the expression of SLC37A2 genes and circulating forms of vitamin D.have has been seen in human peripheral blood mononuclear cells. (50)Vitamin D receptors bind to 1a, 25-dihydroxy vitamin D, which is a metabolite of vitamin D, and regulate gene expression. lo, 25dihydroxyvitamin D, functions in the absorption of Ca and P in the intestine, Ca mobilization in the bone, and reabsorption of Cain the kidneys. (3) There is a report that in adenine-induced CKD model rats, serum la, 25-dihydroxy vitamin D, was significantly higher in rats of high-p diet intake, with advanced vascular calcification, compared to rats of low-p diet intake.39) SLC37A2 may be related to P regulation via vitamin D. Further studies are needed to clarify that vitamin D affects SLC37A2 expression in vitro and Vivo.
It has been shown that the mechanism of vascular calcification is similar to that of bone formation. The deficiency of SLC37A2 caused by CMO is clinically comparable to human infantile cortical hyperostosis, also known as Caffey disease.7 In addition there are reports that SLC37A2 is expressed in bone marrow and osteoclasts.o Hytone et al.(7 hypothesized that a dysfunction of SLC37A2 would cause an imbalance in the function of osteoblastic and osteoclastic bone formation, and as a result, lead to hyperostosis.
Runx2 is an essential transcription factor for vascular calcification. It is also reported that Runx2 is an osteoblast differentiation transcription factor expressed in developing breast epithelial cells. (s435) Runx2 appears to be necessary for the regulation of phosphor-Ser294 progesterone target genes, which are associated with breast cancer progression. SLC37A2 is considered a candidate phosphor-Ser294 progesterone receptor target gene. (s1)Therefore, it is considered that Runx2 may regulate SLC37A2In the present study, the expression of SLC37A2 mRNA in the aorta of CkD rats was correlated with osteopontin mRNA, which targets the gene of Runx2.