Superiority Of Cystatin C Over Creatinine For Early Diagnosis Of Acute Kidney Injury in Pediatric Acute Lymphoblastic Leukemia/ Lymphoblastic Lymphoma
Jan 30, 2024
The exact incidence of acute kidney injury (AKI) during chemotherapy for acute lymphoblastic leukemia (ALL)/lymphoblastic lymphoma (LBL) is unknown. Furthermore, childhood cancer survivors are at risk of AKI-chronic kidney disease transition. Thus, early diagnosis of AKI is crucial. This study aimed to elucidate the incidence of AKI in patients undergoing chemotherapy for pediatric ALL/LBL and to compare the usefulness of serum cystatin C (CysC)- and creatinine (Cr)-based estimated glomerular filtration rate (eGFR) as diagnostic measures. Data from 16 patients with ALL/LBL treated with a total of 75 courses of chemotherapy were retrospectively analyzed. CysC- and Cr-based eGFR were measured before and three times per week during therapy. To calculate the eGFR, an equation for Japanese children was used. AKI was diagnosed when eGFR dropped by ≥ 25% from the highest eGFR value obtained during the last 2 weeks since the start of chemotherapy. AKI was graded based on the pediatric Risk, Injury, Failure, Loss, End Stage Renal Disease scale. All patients developed AKI during chemotherapy; however, more than 90% of the cases were mild and eventually recovered. No significant differences were found in the incidence of AKI between CysC- and Cr-based eGFR (p = 0.104). The median time to AKI diagnosis was significantly shorter in the CysC-based eGFR than in the Cr-based eGFR (8 vs. 17 days, p < 0.001). In this study, all patients with pediatric ALL/LBL could develop mild AKI during treatment. CysC-based eGFR is a more effective measure than Cr-based eGFR for the early diagnosis of AKI.
Keywords: acute kidney injury; acute lymphoblastic leukemia; chemotherapy; creatinine; cystatin C

CLICK HERE TO GET NATURAL ORGANIC CISTANCHE EXTRACT WITH 25% ECHINACOSIDE AND 9% ACTEOSIDE FOR KIDNEY FUNCTION
Supportive Service Of Wecistanche-The largest cistanche exporter in the China:
Email:wallence.suen@wecistanche.com
Whatsapp/Tel:+86 15292862950
Shop For More Specifications Details:
https://www.xjcistanche.com/cistanche-shop
Introduction
Acute kidney injury (AKI) is one of the serious comorbidities associated with the treatment of leukemia as it increases the mortality rate. In adult leukemia (acute myeloid leukemia and high-risk myelodysplastic syndrome), one-third of the patients undergoing chemotherapy develop AKI, and these patients demonstrate a five-fold increase in the mortality rate (Lahoti et al. 2010). However, in both adult and childhood acute lymphoblastic leukemia (ALL), the incidence of AKI during chemotherapy is unknown except for those related to the tumor lysis syndrome or associated with specific chemotherapeutic regimens such as methotrexate (Perazella 1999), cisplatin (Pablo and Dong 2008), and ifosfamide (Ciarimboli et al. 2011).
AKI is a risk factor for the future development of chronic kidney disease (CKD), a condition characterized by progressive loss of renal function. According to previous reports from the Childhood Cancer Survivor Study, the prevalence of CKD ranges from 2.4% to 32% among childhood cancer survivors (Kooijmans et al. 2019). Thus, early diagnosis and treatment of AKI during chemotherapy for ALL, which could reduce the risk of AKI-CKD transition, has clinical significance.
Although serum creatinine (Cr) is broadly used for the diagnosis of AKI, it is affected by several factors other than renal function, including sex, age, and muscle mass (Kellum et al. 2013). Cystatin C (CysC), which is not affected by the above-mentioned factors (Laterza et al. 2002), could potentially be more suitable and sensitive for the assessment of renal function in children (Lankisch et al. 2006; Nakhjavan-Shahraki et al. 2017).
In this study, we aimed to elucidate the incidence of AKI in pediatric ALL/lymphoblastic lymphoma (LBL) during chemotherapy and to compare the efficacy of the CysC-based estimated glomerular filtration rate (eGFR) with the Cr-based-eGFR in the diagnosis of AKI during ALL/LBL treatment.

Materials and Methods
Ethics approval and consent to participate
All procedures performed in this study were in accordance with the ethical standards of the institutional research committee at which the studies were conducted (Kansai Medical University; no. 2019288) and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The authors did not conduct experiments on animals in this study.
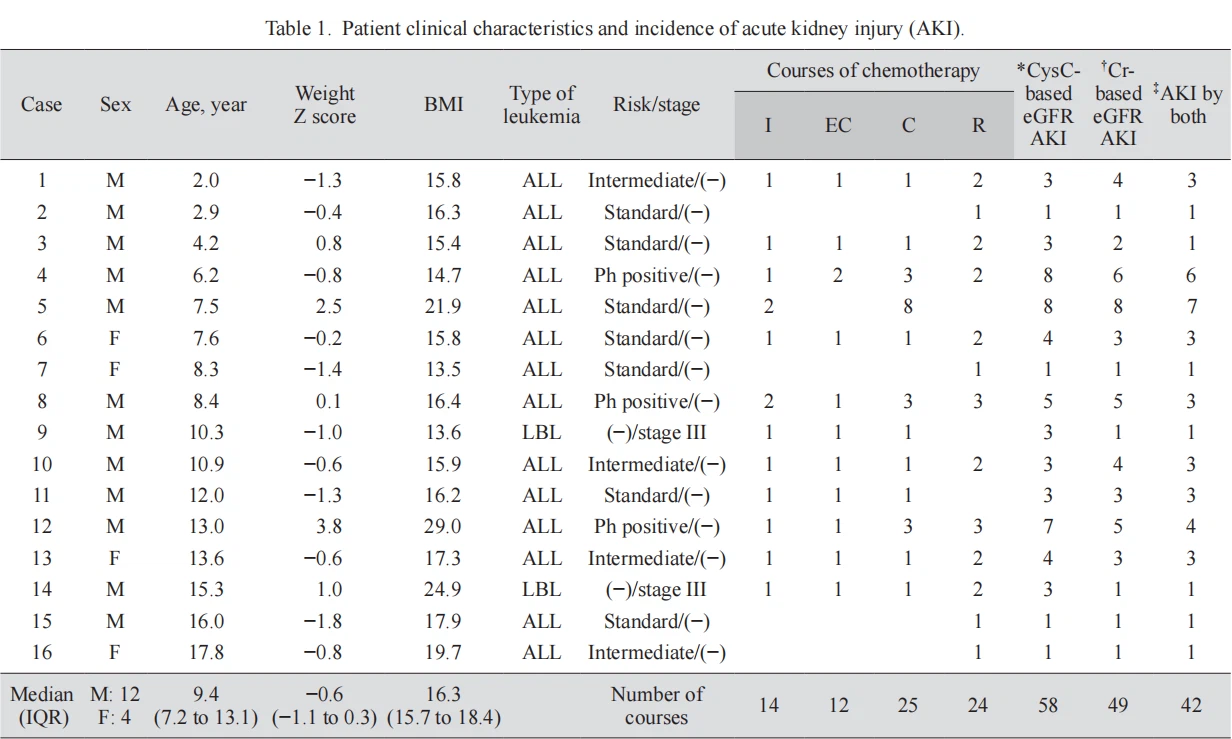
Participants and sample collection Fourteen ALL patients and two LBL patients aged 2-18 years treated at the Pediatric Department of Kansai Medical University Hospital from February 2016 to July 2020 were included. No participants showed elevated Cr or CysC due to tumor lysis syndrome or blast infiltration at diagnosis. The patients received a total of 75 courses of chemotherapy. Serum CysC and Cr levels were measured to calculate eGFR (CysC-based eGFR and Cr-based eGFR, respectively) (Uemura et al. 2014a, b), before and three times a week during each course of chemotherapy. There were 14 courses of induction, 12 courses of early consolidation, 25 courses of consolidation, and 24 courses of re-induction (Table 1). These chemotherapies included cyclophosphamide, cytarabine, dasatinib, daunorubicin, doxorubicin, etoposide, ifosfamide, L-asparaginase, mercaptopurine, methotrexate, pirarubicin, vincristine, and vindesine. The patients were treated according to the Japanese Pediatric Leukemia/ Lymphoma Study Group protocol.
Assessment of AKI AKI was diagnosed when eGFR dropped by 25% or more from the highest eGFR value obtained during the latest 2 weeks since the start of chemotherapy. AKI was graded based on the pediatric Risk, Injury, Failure, Loss, End Stage Renal Disease scale (pRIFLE) (Akcan-Arikan et al. 2007) as shown in Table 2, though the reduction in urine output was not applied in this study. AKI was diagnosed using the CysC- and Cr-based eGFR independently, and diagnoses were compared for AKI incidence rate, severity, and days until AKI diagnosis from the start of each treatment course.
Long-term follow-up
To assess long-term renal outcomes such as the development of CKD, CysC-based eGFR was followed until March 2021 with urine and blood tests. CKD was assessed primarily using the Kidney Disease: Improving Global Outcomes (KDIGO) definition: having either low eGFR (eGFR < 90 mL/min/1.73 m2 ) or proteinuria (protein ≥ 1+ by urine dipstick) (Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group 2013).

Statistical methods
Data on sex, age, type of leukemia, risk, number of courses, treatment dosage, and duration were extracted from the medical records. For data analyses, the Mann– Whitney U test or chi-square test were used, and p < 0.05 was considered significant. To verify the results, post hoc power analysis with the significance level α set to 0.05 was performed using G*Power version 3.1.9.4 (Heinrich-Heine University, Düsseldorf, Germany) (Faul et al. 2007).