The Central Role Of Transforming Growth Factor-β (TGF-β) in Renal Fibrosis Development
Mar 26, 2022
Contact: Audrey Hu Whatsapp/hp: 0086 13880143964 Email: audrey.hu@wecistanche.com
PART Ⅰ: Role of Human Primary Renal Fibroblast in TGF-β1-Mediated Fibrosis-Mimicking Devices
Seong-Hye Hwang, Yun-Mi Lee & et al.
1. Introduction
Renal fibrosis, the final common pathway of innumerable progressive kidney diseases, is characterized by an increase in extracellular matrix(ECM) protein production, a decline in matrix degradation, dysfunction of cell-matrix interaction, the transformation of resident cells, and inflammatory cell infiltration. Transforming growth factor-β (TGF-β) is a multifunctional dimeric peptide that regulates biological processes such as cell proliferation, differentiation, and immunological responses [1]. Among the numerous fibrogenic factors, TGF-β(Transforming growth factor-β) plays a central role and has an anti-inflammatory effect. TGF-β1(Transforming growth factor-β)-deficient mice die of massive inflammation. Transgenic mice overexpressing TGF-β1(Transforming growth factor-β) are virtually protected from developing renal fibrotic pathology, mainly through its anti-inflammatory activity [2]. The profibrotic and anti-inflammatory properties of TGF-β(Transforming growth factor-β) are double-edged swords regarding the therapeutic application of TGF-β(Transforming growth factor-β) inhibition. Remarkable efforts have been made to obstruct TGF-β(Transforming growth factor-β) action to hinder the progression of renal fibrosis [3]. Although many approaches have been utilized to combat renal fibrosis, an experimental model to evaluate currently available drugs is not ideal.
cistanche herb prevent renal diseases
In general, animal studies play a pivotal role in preclinical tests in predicting pharmacokinetics and drug efficacy. However, various differences exist between animals and humans, which drives us to question the accuracy of animal-based drug efficacy and safety tests 4. Previous reports have depended on preclinical animal studies during drug development to assess the nephrotoxicity and safety of drugs. The renal functioning of animals is more than twice that of the human kidneys. Hence, drugs are metabolized more quickly in animals than in humans, making it difficult to elucidate the results of drug toxicity tests using animal studies. Furthermore, the results of animal experiments cannot be used to predict drug responses in humans, as animals have different physiological functions. Animal-based renal fibrosis models have difficulties in reproducing human renal fibrosis; hence, research is underway to overcome current obstacles and optimize drug-discovery platforms [5].
Organ-on-a-chip (OoC) technology has emerged as a novel concept to solve these issues. This platform was designed considering the current shortcomings, and it has shown great promise in the field of nephrology [6]. In addition, OoC models hold great potential as a replacement for experimental animal models, provide a solution for inter-species differences, and can help end animal cruelty and ethical debates [7]. There are few in vitro models that simultaneously use primary renal fibroblasts with endothelial and epithelial cells, which are important cells in kidney fibrosis. The role of each cell in fibrosis is being studied separately; however, there is limited knowledge concerning the interaction of cells.
In the present study, we developed fibrosis-mimicking devices using human primary fibroblasts. We confirmed that TGF-β1(Transforming growth factor-β) treatment has various effects on the renal fibrosis model. Notably, fibroblasts play a pivotal role as effector cells, promote the formation of a pro-fibrotic microenvironment, and secrete growth factors and cytokines. Therefore, based on this new method, fibroblasts may provide an ideal cell model system to study renal disease. We evaluated whether fibrotic renal disease-derived fibroblasts affect the integrity of cells cultured in three dimensions (3D). Finally, we present a proof-of-principle study that demonstrates the potential of human renal fibrosis-mimicking devices as a model for predicting the response to human renal fibrosis.
effects of cistanche: treat kidney infection
2. Results
2.1.Identification of Alpha Smooth Muscle Actin(a-SMA)and Keratin-8(KRT-8)by TGF-β1(Transforming growth factor-β) in HK-2 Cells
In the early phase, we examined known markers of fibrosis and the cytoskeleton in two-dimensional (2D)cultures. Immunofluorescent staining assays were performed to estimate the α-SMA expression of fibrosis and tubular epithelial to mesenchymal transition (EMT)marker and KRT8 levels as a cytoskeleton marker. Immunofluorescence staining showed that the expression of α-SMA was originally weak and KRT8 levels were high in the monolayer HK-2 epithelial cells. There was no significant alteration in fluorescence intensity by TGF-β1(Transforming growth factor-β) (5 ng/mL)or the specific inhibitor treatment of HK-2 epithelial cells for 24 h (Figure 1).


Figure 1. Immunofluorescence shows that the expression of alpha-SMA and keratin-8(KRT8) was maintained by treatment of TGF-β1(Transforming growth factor-β) or the inhibitor in HK-2 cells. (A)The expression of alpha-SMA and (B)CCK-8 had no effect on HK-2cells by TGF-β1(Transforming growth factor-β)(5ng/mL) or the inhibitor. Cells were originally plated at a density of 1×10° per well (a-c)untreated control and (d-f)stimulated with5ng/mL TGF-β1(Transforming growth factor-β) or(g-i)10 μM inhibitor(SB431542) for 24 h and fixated with 4% paraformaldehyde. The cells were then stained with anti-α-SMA and anti-cytokeratin-8 for 20 min. Co-staining with Hoechst dye H33342 to identify cell nuclei was performed. Scale bars in micrographs indicate 200 um.

cistanche powder improves kidney function
2.2. KRT8 Expression Decreased by TGF-β1(Transforming growth factor-β) on a Chip
Next, we evaluated the expression levels of α-SMA and KRT8 in 3D-cultured HK-2 and the total length of GFP-human umbilical-vein endothelial cells(HUVECs) treated with TGF-β1(Transforming growth factor-β) and a specific inhibitor. To accomplish this, we first established a tissue chip pattern comprising two cell types, HK-2 epithelial cells, and GFP-HUVECs. Following confirmation of the construct, we exposed the tissue chip with TGF-β1(Transforming growth factor-β) (5 ng/mL) or a specific inhibitor (10 um SB431542) for 24 h.
As shown in Figure 2A-C, the expression of α-SMA was not specifically changed by TGF-β1(Transforming growth factor-β) or the specific inhibitor treatment. However, the expression of KRT8 was reduced in the two-cell type co-cultured chip. Moreover, the addition of a TGF-ß1 inhibitor increased the expression of KRT8.
We examined the formation of a 3D tubular capillary-like network by TGF-β1(Transforming growth factor-β) treatment in HUVECs, as endothelial cells can spontaneously form a 3D tubular capillary-like network. In accordance with our cultural conditions, the formation of thick lines was significantly decreased. However, capillary-like thin lines were increased in the TGF-β1(Transforming growth factor-β) treated GFP-HUVECs. In the case of the TGF-β1(Transforming growth factor-β) inhibitor treatment on the 3D-cultured chip, the total length of the thick and thick lines showed a reverse tendency to TGF-β1(Transforming growth factor-β) treatment. An increase in the length of the thick line was observed compared to the untreated control group. A similar pattern was observed in the diameter of the thick line in GFP-HUVEC after TGF-β1(Transforming growth factor-β) treatment and inhibitor treatment. The diameter of the GFP-HUVEC was suppressed by the TGF-β1(Transforming growth factor-β) treatment but increased irrespective of the thickness of the line by the inhibitor compared to both the controls and TGF-β1(Transforming growth factor-β). The total length and diameter of the thick line were dramatically increased by TGF-β1 inhibitor treatment compared to the untreated control and TGF-β1 treatment. Notably, the obtained results indicated an increased density of tiny vessels in the TGF-β(Transforming growth factor-β)-treated group compared to their untreated counterparts. In contrast, the density of thick vessels in the TGF-β1(Transforming growth factor-β) treatment group was lower than that in the untreated control group (Figure 2D, E).



Figure 2. KRT8 expression in HK-2 and total length and diameter of the HUVECs.(A)Immunofluorescence shows that the expression of α-SMA was maintained and KRT8 expression was dramatically decreased by treatment of TGF-β1(Transforming growth factor-β) in HK-2. Cells were plated and stimulated with 5ng/mL TGF-β1(Transforming growth factor-β) or 10 μm inhibitor (SB 431542) for 24 h and fixated with 4%paraformaldehyde. The cells were stained with anti-α-SMA and anti-cytokeratin-8 for 20 min. Co-staining with Hoechst dye H33342 to identify cell nuclei was performed. The expression of α-SMA(a-c) had no alteration but KRT8(d-f) in HK-2 cells and GFP in HUVEC (g-i) expression were significantly changed by TGF-β1(Transforming growth factor-β)(5 ng/mL) and the inhibitor(B, C).In the total length of the HUVECs, the thin vessel was increased but the thick vessel was decreased by TGF-β1(Transforming growth factor-β). The diameter was increased in both the thin and thick vessels. These results were reversed by the inhibitor SB431542 (D, E). Scale bars in the micrographs indicate 100 μm.*p<0.05,**p<0.01,***p<0.001 versus the control group;#p<0.01,###p<0.001 versus the TGF-β1 group. Each value represents three technical replicates of each of the three biological replicates. Statistical significance of the length compared to the non-treated cells is represented in the graph. Thin vessels mean a length shorter than 50 um and thick vessels represent a length longer than 50 um.

what is cistanche used for: treating chronic kidney diseases
2.3. TGF-β1(Transforming growth factor-β) Affects Three Cell Types of the Chip with a Multi-Compartment Structure
To study the TGF-β1(Transforming growth factor-β) responses of the proposed 3D chip, we performed immunostaining on each compartment separately (Figure 3). Primary human renal fibroblasts were utilized in this analysis. The samples were analyzed using FACS and confirmed to be positive for the fibroblast marker antibody. Following incubation of the three cell types with TGF-β1(Transforming growth factor-β) for 24 h, immunostaining was performed to assess the expression of α-SMA and KRT8. The expression of a-SMA was upregulated by TGF-1 treatment, and opposite results were observed with TGF-β1(Transforming growth factor-β) inhibitor treatment. Conversely, the expression of KRT8 was downregulated by TGF-β1(Transforming growth factor-β) and showed an opposite response to the TGF-β(Transforming growth factor-β) inhibitor (Figure 4A-C).


Figure 3. Sample image with device schematic demonstration of the fabrication and experimental schedule. (A)The top two images show the layout. The left image shows an overall top-down photo of the device. The bottom images show the cross-section. Schematic view detailing the dimensions of the liquid guides. (B)This figure describes the experimental schedules for fibrosis-mimicking devices. The four-step loading process for each well.Location of each hydrogel patterning area, as well as the placement of the media in top-down and isometric cross-section. (1) A total of 1.5 uL of hydrogel 1 is spontaneously guided into the central channel. (2)Central channel is filled with5uL of hydrogel. (3)A total of 10uL of hydrogel 3 are patterned on the reservoir floor. (4)A total of 200μL of media are dispensed. Red scale bar=9 mm.
As depicted in Figure 4D-G, the length of the capillary-like thin line was significantly increased by TGF-β1(Transforming growth factor-β) treatment in the GFP-HUVECs. However, the thickness of the thick line was decreased in the TGF-β1(Transforming growth factor-β) treated HUVECs. The diameter was reduced on average for the thin and thick vessels in the HUVECs. In the case of the TGF-β1(Transforming growth factor-β) inhibitor treatment on the 3D-cultured chip, the total length of the thick and thick lines showed an opposite trend to the TGF-β1 treatment. Further, the length of the thick line was increased compared to the untreated control group.


Figure 4. Alteration of 3D-cultured HK-2 and HUVECs with primary renal fibroblasts. Total cells were plated at a density of 5×10° per 3D chip and stimulated with5 ng/mL TGF-β1(Transforming growth factor-β) or 10 μm inhibitor (SB 431542) for 24 h and fixated with 4%paraformaldehyde. (A) Cells in the 3D chip were stained with anti-oα-SMA and anti-cytokeratin-8. The expression of α-SMA (a-c) was increased and KRT8(d-f) expression was decreased by TGF-β1(5 ng/mL) significantly. The total length of the HUVECs; thin vessels were dramatically increased and thick vessels were decreased by TGF-ß1(Transforming growth factor-β)(D, E). The diameter was decreased for thin and thick vessels(F, G). These results were reversed by the inhibitor SB431542. Scale bars in micrographs indicate 100 μm.*p<0.05,**p<0.01,and ** p<0.001 versus the control group;#p<0.05,#p<0.01,and###tp<0.001 versus the TGF-β1 group. Each value represents three technical replicates of each of the three biological replicates. Thin vessels mean a length shorter than 50 um and thick vessels represent a length longer than 50 um.
2.4.TGF-81 Modulates Inflammatory Mediator and Growth Factors
Next, we investigated the inflammatory cytokines and growth factors in the supernatant; IL-1β, FGF-2, TGF-β2(Transforming growth factor-β), and TGF-β3(Transforming growth factor-β) protein secretion was dramatically higher in 3D-cultured chips than the 2D fibroblast cultured well, even though they had the same total cell count. However, TGF-β1(Transforming growth factor-β) levels were similar, as the 2D- or 3D-cultured models were treated with similar concentrations of TGF-β1(Transforming growth factor-β)(Figure 5A-E). Particularly, the release of FGF-2 from human primary renal fibroblasts was 20.9 ± 0.4 pg/day/5× 10° cells of the 2D mono-layered culture and 3094.8± 0.2 pg/day/5× 10° cells including the HUVECs and HK-2 of the 3D-cultured chip in the untreated control. The secretion of FGF-2 by TGF-β1(Transforming growth factor-β) stimulation was 31.0±0.2pg/day/5×10°cells for the 2D culture and 3683.9± 0.8 pg/day/5×10° cells for the 3D-cultured chip. This increase in FGF-2 protein production in the 2D monolayer culture was paralleled by an increase in the 3D-cultured chip model by TGF-β1(Transforming growth factor-β) treatment (Figure 5B).
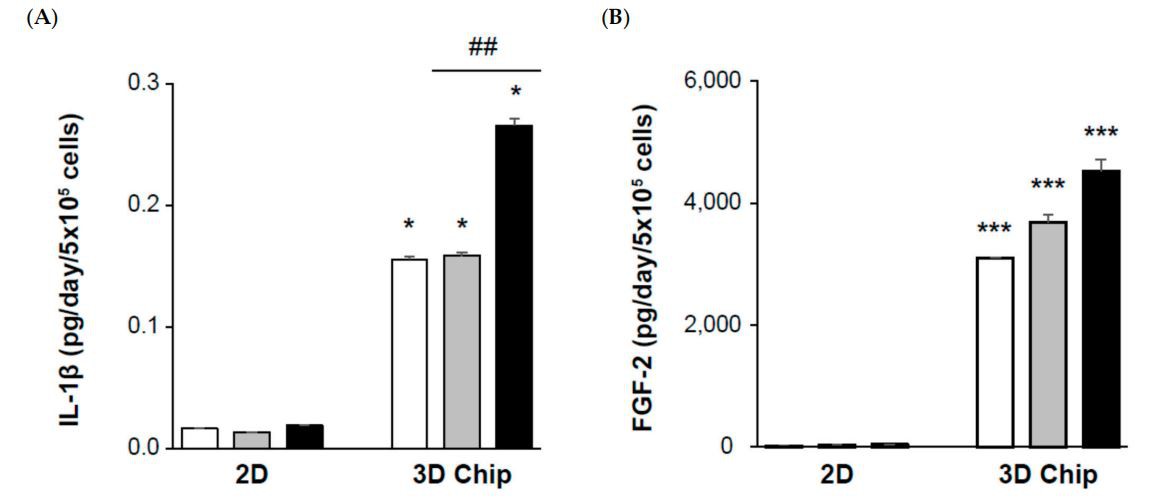


Figure 5. Comparison of 2D-and 3D-cultured models as renal fibrosis-mimicking platforms. Cells were plated at a density of 5×10°per 2D well or 3D chip and stimulated with5ng/mL TGF-β1(Transforming growth factor-β) or 10 um inhibitor (SB 431542) for 24 h. Primary human renal fibroblasts were used in the 2D-cultured and in the 3D-cultured model. Fibroblasts were utilized at a density of 8× 10* cultured with HUVECs and HK-2. The primary human renal fibroblast-to-HUVEC ratio, or F: H ratio, was 1:5 for assessing fibrosis.HK-2 cells were used at a density of 2×104. Detection of(A)IL-1β,(B)basic fibroblast growth factor (FGF-2), and(C-E)TGF-beta 1, 2, and 3 in the supernatant were performed by multiplex analysis of cytokines and multiplex bead immunoassay. Each bar represents the mean± SE.*p<0.05,*** p<0.001 versus 2D;##p<0.01,## p<0.001 versus TGF-β1(Transforming growth factor-β).

cistanche tubolosa benefits: treating kidney diseases
Furthermore, we estimated the mRNA expression of pro-inflammatory cytokines such as IL-1β, IL-6, IL-8, and TNF-α. The results were significantly downregulated by recombinant human TGF-β1(Transforming growth factor-β) treatment in the 3D-cultured chip as an anti-inflammatory property of TGF-β(Transforming growth factor-β). We found that mRNA expression was upregulated by the TGF-β1(Transforming growth factor-β) inhibitor (SB431542). Furthermore, inhibitor-mediated expression of IL-6, IL-8, and TNF-αwas specifically downregulated compared to in the untreated control (Figure 6A-D). The mRNA level of VEGF, a fibrotic factor, was significantly increased by TGF-β1(Transforming growth factor-β) treatment. The mRNA level of VEGF was decreased by the TGF-β1 inhibitor (Figure 6E), whereas the mRNA expression of IL-10 with anti-fibrotic effect was decreased in fibrotic conditions induced by TGF-β1. In contrast, I-10mRNA expression was increased by TGF-β1inhibitor treatment (Figure 6F).



Figure 6. Real-time PCR shows the overall mRNA expression of the 3D chip. Cells were plated at a density of5×10° per 3D chip and stimulated with5ng/mL TGF-β1(Transforming growth factor-β) or 10 um inhibitor(SB431542)for 24h. Total RNA extracted from the 3D-cultured chip. The mRNA expression of(A)IL-1 β,(B)TNF-α,(C)IL-6, and (D)IL-8 represents the anti-inflammatory effect of the TGF-β1(Transforming growth factor-β) treatment. (E)VEGF expression as an angiogenic factor was increased. (F)IL-10 expression as an anti-fibrotic factor was significantly decreased by TGF-β1.*p<0.05,**p<0.01,***p<0.001 versus control;#p<0.05,#p<0.01,##p<0.001 versus TGF-β1(Transforming growth factor-β).








