The ‘other’ Big Complication: How Chronic Kidney Disease Impacts On Cancer Risks And Outcomes
Jan 25, 2024
CLICK HERE TO GET NATURAL ORGANIC CISTANCHE EXTRACT WITH 25% ECHINACOSIDE AND 9% ACTEOSIDE FOR KIDNEY FUNCTION
ABSTRACT
Cancer is the second leading cause of death in people with chronic kidney disease (CKD) after cardiovascular disease. The incidence of CKD in patients with cancer is higher than in the non-cancer population. Across various populations, CKD is associated with an elevated risk of cancer incidence and cancer death compared with people without CKD, although the risks are cancer-site-specific. Higher risk of cancer is detectable in mild CKD [estimated glomerular filtration rate (eGFR) 60–89 mL/min/1.73 m2], although this risk is more obvious if sensitive markers of kidney disease are used, such as cystatin C. Independent of eGFR, albuminuria is associated with an increased risk of site-specific cancer incidence and death. Here, we explore the potential mechanisms for the increased risk of cancer observed in CKD, including patient factors (shared risks such as cardiometabolic disease, obesity, smoking, diet, lifestyle, and environment), disease (genetic, inflammatory, and infective), and treatment factors. In particular, we discuss the ways in which renal adverse events associated with conventional chemotherapies and newer systemic anti-cancer therapies (including targeted and immunotherapies) may contribute to worse cancer outcomes in people with CKD. Finally, we review the potential benefits of acknowledging the increased risk of cancer in risk prediction tools used for the management of CKD.
Keywords: cancer, CKD, creatinine, cystatin C, GFR

RISK OF CANCER INCIDENCE AND DEATH IN CKD
Chronic kidney disease (CKD) is a significant and growing health problem, with CKD stage 3–5 affecting 11–13% of the general population globally. Understanding the relationship between CKD and multimorbidity is crucial to improve outcomes for patients. There is a clear overlap of the risk factors for CKD and cancer, but the incidence of CKD in patients with cancer is higher than in the general population and can have major impacts on treatment options and outcomes. In this review, we examine the potential processes underlying the multifactorial association between CKD and cancer.

Incident cancer-which eGFR equation?
The vast majority of studies report cancer risk using estimated glomerular filtration rate (eGFR) based on serum creatinine alone (eGFRcr) [1–11]. However, eGFR based on a combination of creatinine and cystatin C (eGFRcr-cys) is more accurate than eGFRcr compared to measured GFR [12]. eGFR based on cystatin C alone (eGFRcys) is more sensitive in detecting early changes in kidney function and increased risk of cardiovascular disease [13, 14].
Two studies have reported CKD cancer risk by eGFR measures incorporating cystatin C, with conflicting results. In the Atherosclerosis Risk in Communities (ARIC) cohort in the USA (n = 8935) [15], there was no clear association between lower eGFRcr-cys and overall cancer incidence after adjustment for other relevant baseline demographics. However, in 431 263 participants from the UK Biobank, cancer incidence and death were compared by eGFRcr, eGFRcys, and eGFRcr-cys. eGFR <90 mL/min/1.73 m2 by eGFRcys and eGFRcr-cys (but not eGFRcr) was associated with an increased risk of incident cancer, with a graded increase in risk with progressive decline in eGFR [16]. The risk was most strongly associated with eGFRcys {hazard ratio (HR) 1.04 [95% confidence interval (CI) 1.03–1.04]} [16]. There was also an increased risk in site-specific incidence of hematological, respiratory, and abdominal solid organ cancers in people with eGFR <90 mL/min/1.73 m2, but only when kidney function was estimated using eGFRcys, and not eGFRcr [16].
Incident cancer in all CKD
Amongst people diagnosed with cancer, CKD has increased prevalence. In a French observational study of 4684 patients (Renal Insufficiency and Cancer Medications: IRMA), more than half of participants diagnosed with cancer had mild reduction in kidney function [17] <90 mL/min/1.73 m2 [57% by creatinine clearance; 53% by Modified Diet in Renal Disease [18] (MDRD) eGFR], and 12–20% had creatinine clearance or MDRD eGFR <60 mL/min/1.73 m2.
Cancer event rates are higher among people with CKD than in the general population [16]. Population cohorts from Taiwan (n = 405 878 [1] and n = 123 717 [2]), China (n = 11 508) [4], Korea (n = 242 583) [5], Japan (n = 961) [6], Sweden (n = 719 033) [7], Australia (n = 4077) [8] and the UK (n = 431 263) [16] suggest that there is up to 108% increased risk of overall cancer in patients with moderate CKD (eGFRcr <60 mL/min/1.73 m2), even after accounting for shared risk factors for CKD and cancer.
Albuminuria is independently and additively associated with cancer risk, specifically a 9–66% increase in risk of incident cancer [1, 5, 15, 16]. This is observed whether proteinuria is identified semi-quantitatively on dipstick testing [1, 5, 9, 19] or quantitatively by urinary albumin-to-creatinine ratio (ACR) [15, 16, 20], demonstrating a dose–response relationship [1, 5, 16].

Incident cancer in non-dialysis CKD
In non-dialysis CKD, several population cohorts [3, 7, 9, 11, 21] and one meta-analysis [10] describe no difference in risk of overall cancer incidence or death, but do report associations between lower eGFRcr and elevated risk of specific cancer subtypes [7, 9, 10, 15, 19]. Associations between lower eGFRcr and risk of incident cancer exist for site-specific cancers of the renal tract (kidney, ureter, and bladder) [7, 10, 16], oropharyngeal [19], respiratory [4, 16], haematologic [7, 16] (including myeloma [9], leukemia [9]), skin [7] and abdominal solid organs [16]. Lower eGFR predominantly is not associated with increased risk of sex-specific prostate [7, 9, 10] and breast [7, 9, 10] cancers. The presence of albuminuria has been associated with site-specific incidence from cancers of the renal tract [16, 19, 20], lung [15, 16, 19, 20], stomach [19], abdominal solid organs [16] (including liver [19] and pancreas [19]) and hematological [16] (including myeloma [19] and non-myeloma [16]) malignancies, although not prostate [16, 20], breast [16, 19, 20] or digestive tract (including colorectal [19, 20]) cancers.
Incident cancer in end-stage kidney disease
It is well established that patients with end-stage kidney disease are at a higher risk of cancer than the general and CKD populations. Kidney transplant recipients have around 3-fold increased risk of developing cancer than the general population [22, 23], particularly in association with infections (especially viruses) [23]. Malignancy (21%) and infection (23.6%) are now the most common causes of death in kidney transplant recipients in the UK, outside of the first year after transplantation [24]. The risk of cancer increases with time post-transplantation [23, 24]. There is also reasonable data to support the increased risk of cancer incidence and cancer death in people on dialysis [10, 23]. In a meta-analysis including data from five clinical trials and one population cohort (n = 32 057 individuals), there was a trend toward increased risk of cancer incidence and a significant increase in cancer death across all cancer subtypes among patients on dialysis [10]. In particular, there was more than double the risk of incident urinary tract cancers, more than 11 times the risk of endocrine cancers, and more than twice the risk of death from digestive cancers. However, this was balanced by a relative reduction in risk of other site-specific cancers such as prostate.
Cancer outcomes
Perhaps more importantly than its association with incident cancer, CKD is associated with reduced cancer survival.
Death from cancer is common in people with CKD, accounting for 14–39% of deaths in people with eGFR <60 mL/min/1.73 m2 [19, 21, 25]. Across all cancer subtypes, CKD is associated with a 20–48% increase in cancer death [1, 2, 8, 16]. This is most pronounced when eGFR is estimated using cystatin C [HR for death from cancer 1.10 (95% CI 1.08–1.11) per 10 mL/min/1.73 m2 decline in eGFRcys] [16]. Relative to people who do not have CKD, people with moderate-to-severe CKD (G3–G5) have a 50–74% increase in the risk of cancer death for abdominal solid organs [16] including liver [2], more than 3 times the risk of death from the kidney [2] cancers, up to 7 times the risk of death from urinary tract cancers [2, 16] and 3– 7 times the risk of cancer death from hematological cancers including lymphoma.
Similarly, albuminuria is associated with a 17–53% increase in risk of cancer death overall [1, 16, 19], but a more pronounced association with cancer death in certain site-specific cancers. In particular, albuminuria is associated with 2–4 times the risk of death from urinary tract cancers [16, 19], nearly 4 times the risk of death from myeloma [19], and smaller increases in the risk of death from cancers of the abdominal solid organs [16] (including liver [19] and pancreas [19]) and digestive tract [16] (including stomach [19]). This association may be stronger in younger (<65 years) people [16], but remains for older (≥65 years) people, suggesting that it is not solely due to a confounding effect of age. Transient dipstick proteinuria does not appear to be associated with an increased risk of cancer death, although it is associated with an increased risk of cardiovascular death [26].
WHY MIGHT CKD BE ASSOCIATED WITH CANCER?
Patient, disease,e and treatment factors that may influence cancer risk in CKD are summarized in Figure 1.
Patient factors
In addition to shared risk factors including smoking, obesity, and cardiometabolic disease, unhealthy lifestyle factors, such as poor diet and sedentary behavior, are associated with an increased risk of cancer mortality in patients with and without CKD [27], and increased risk of both incidents [28] and progressive CKD [29]. The mechanisms behind the association between lifestyle and cancer have not been fully elucidated, but include a direct association with other cardiometabolic risk factors, increased inflammation leading to oxidative DNA damage, or ingestion of carcinogens (such as nitrites contained in many preserved foods) [30]. Similarly, deprivation (reducing access to healthcare), health literacy (reducing understanding of the significance of symptoms or how to engage with healthcare services), and occupational or environmental exposures (such as asbestos, pesticides, dust, hydrocarbons, and other pollutants) may also contribute. Inadequate adjustment in observational models for these confounders may partly explain the observed associations. Exploration of causal pathways and the impact of shared genetic risks-e.g. using Mendelian randomization-could be a means of improving our understanding of these mechanisms.
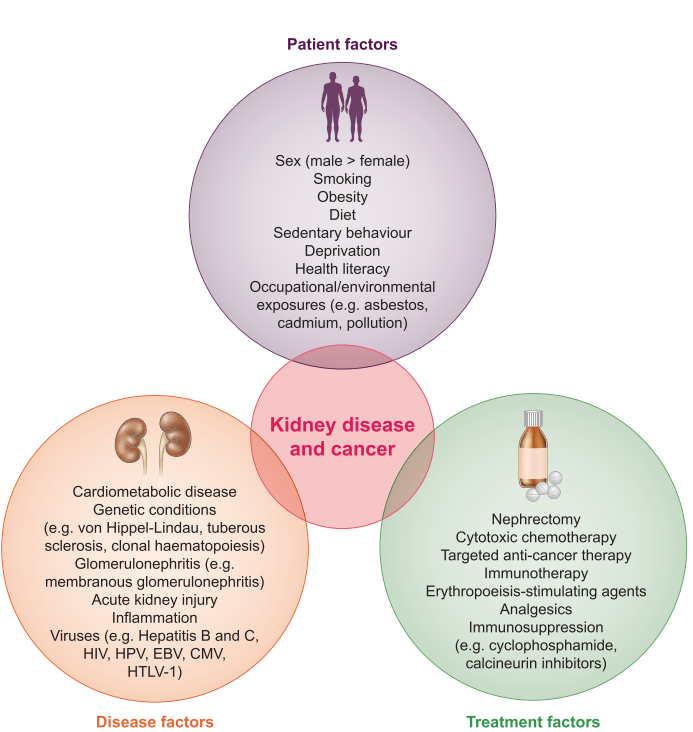
FIGURE 1: Patient, disease and treatment factors associated with kidney disease and cancer. HIV, human immunodeficiency virus; HPV, human papillomavirus; EBV, Epstein-Barr virus; CMV, cytomegalovirus; HTLV-1, human T-cell leukemia virus type 1.
Supportive Service Of Wecistanche-The largest cistanche exporter in the China:
Email:wallence.suen@wecistanche.com
Whatsapp/Tel:+86 15292862950
Shop For More Specifications Details:
https://www.xjcistanche.com/cistanche-shop